Revolutionizing 2,3-Dichloropyridine Production via Safe Catalytic Transfer Hydrogenation Technology
The global demand for high-performance agrochemical intermediates continues to surge, driven by the need for efficient crop protection solutions like diamide insecticides. At the heart of this supply chain lies 2,3-dichloropyridine, a critical building block whose production efficiency directly impacts downstream manufacturing costs and safety profiles. Historically, the synthesis of this key intermediate has been plagued by hazardous reaction conditions and suboptimal yields, creating significant bottlenecks for procurement and supply chain teams. However, a pivotal technological breakthrough documented in patent CN103145609A introduces a transformative approach utilizing Catalytic Transfer Hydrogenation (CTH). This innovation replaces traditional high-pressure gaseous hydrogen protocols with a safer, ambient-pressure system using organic hydrogen donors. For R&D directors and technical procurement officers, this shift represents not merely a chemical optimization but a strategic advantage in risk management and operational stability. By leveraging this advanced methodology, manufacturers can achieve superior selectivity while eliminating the severe safety liabilities associated with explosive hydrogen gas, thereby securing a more resilient supply chain for essential agrochemical precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 2,3-dichloropyridine have long been constrained by inherent safety risks and economic inefficiencies that hinder large-scale industrial adoption. Early methodologies, such as the Gatterman reaction starting from 3-amino-2-chloropyridine, suffered from notoriously low yields that rendered them commercially non-viable for mass production. Subsequent attempts involving liquid-phase chlorination of pyridine or 3-chloropyridine resulted in complex isomer mixtures with poor selectivity, necessitating expensive and energy-intensive purification steps to isolate the target molecule. Furthermore, the most common industrial precedent, disclosed in patents like JP1193246, relies on the use of gaseous hydrogen in the presence of a palladium catalyst. While chemically effective, this approach mandates the use of high-pressure reactors capable of containing flammable and explosive hydrogen gas, imposing stringent safety regulations and substantial capital expenditure on infrastructure. Additionally, alternative post-treatment methods involving concentrated hydrochloric acid extraction generate massive volumes of corrosive waste acid, creating severe environmental compliance burdens and disposal costs that erode profit margins for chemical manufacturers.
The Novel Approach
The novel methodology presented in patent CN103145609A fundamentally reengineers the production landscape by substituting hazardous gaseous hydrogen with stable, solid organic hydrogen donors such as ammonium formate or sodium formate. This Catalytic Transfer Hydrogenation (CTH) strategy operates effectively under atmospheric pressure or slight reflux conditions, completely obviating the need for specialized high-pressure containment equipment and drastically reducing the potential for catastrophic safety incidents. The process utilizes a palladium on carbon (Pd/C) catalyst which, according to the patent data, can be recovered, regenerated, and continuously reused across multiple batches, significantly lowering the consumption of precious metals. Moreover, the reaction solvent system employs common C1-C4 alcohols like ethanol, which are inexpensive, readily available, and easily recovered through rectification for reuse in subsequent cycles. This holistic improvement in process safety, combined with the ability to fully utilize raw materials and convert by-products into economically valuable co-products, establishes a new benchmark for sustainable and cost-effective manufacturing of chlorinated pyridine derivatives.
Mechanistic Insights into Pd/C-Catalyzed Dechlorination
The core of this technological advancement lies in the sophisticated interaction between the palladium catalyst surface and the organic hydrogen donor within the alcoholic solvent medium. In this CTH system, the formate ion acts as a hydride source, adsorbing onto the active sites of the Pd/C catalyst where it undergoes decomposition to release reactive hydrogen species in situ. Unlike gaseous hydrogen which requires high pressure to force dissolution and reaction, these surface-bound hydrogen atoms are immediately available for the selective hydrodechlorination of the 2,3,6-trichloropyridine substrate. The reaction mechanism favors the removal of the chlorine atom at the 6-position of the pyridine ring due to electronic and steric factors facilitated by the catalyst, yielding the desired 2,3-dichloropyridine with high specificity. This controlled release of hydrogen prevents over-reduction of the pyridine ring itself, a common side reaction in aggressive hydrogenation processes, thereby maintaining the structural integrity of the aromatic system which is crucial for downstream coupling reactions in pesticide synthesis.
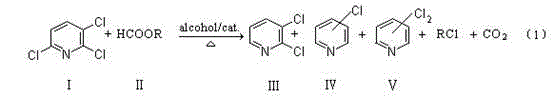
Impurity control is meticulously managed through the precise regulation of reaction temperature and the batch-wise addition of the hydrogen donor. The patent specifies maintaining a micro-reflux condition between 40°C and 60°C, preferably around 50-55°C, which provides sufficient thermal energy to drive the catalytic cycle without promoting thermal degradation or non-selective side reactions. By adding the hydrogen donor in batches over a period of 3 to 8 hours, the concentration of reactive hydrogen species is kept at an optimal level, preventing local excesses that could lead to over-hydrogenation or the formation of fully dechlorinated pyridine. The resulting by-product profile consists mainly of other chloropyridines such as 3-chloropyridine and 2,5-dichloropyridine, which are not waste but rather valuable intermediates in their own right. This high level of selectivity minimizes the formation of tarry residues or unidentifiable impurities, simplifying the downstream purification process and ensuring that the final product meets the stringent purity specifications required by global agrochemical companies for registration and formulation.
How to Synthesize 2,3-Dichloropyridine Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding solvent ratios, catalyst loading, and thermal profiles to ensure maximum conversion and yield. The process begins by dissolving the starting material, 2,3,6-trichloropyridine, in a C1-C4 alcohol solvent such as ethanol, followed by the addition of a portion of the hydrogen donor to initiate the system. Once the mixture is heated to the induction temperature, the Pd/C catalyst is introduced, triggering the evolution of gas and the onset of the reduction reaction. To maintain control over the reaction kinetics and heat generation, the remaining hydrogen donor is added gradually over several hours while maintaining micro-reflux. Upon completion, the catalyst is filtered off while hot to prevent product crystallization on the filter cake, and the solvent is recovered for reuse, leaving a residue that is washed to remove salts before final distillation.
- Dissolve 2,3,6-trichloropyridine and a portion of the hydrogen donor in a C1-C4 alcohol solvent, heating to 40-60 degrees Celsius.
- Introduce the Pd/C metal catalyst and raise the temperature to micro-reflux to initiate the dechlorination reaction.
- Batch add the remaining hydrogen donor over 3-8 hours, then filter the catalyst hot and purify the product via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this CTH-based manufacturing process offers profound strategic benefits that extend far beyond simple chemical yield improvements. The elimination of gaseous hydrogen removes the requirement for expensive high-pressure reactors and the associated safety infrastructure, leading to significant capital expenditure savings and reduced insurance premiums for production facilities. Furthermore, the ability to recycle the Pd/C catalyst multiple times without significant loss of activity drastically reduces the consumption of precious metals, which are subject to volatile market pricing, thereby stabilizing long-term production costs. The use of common alcohol solvents and solid formate donors ensures a robust supply chain less susceptible to disruptions compared to specialized gases or hazardous reagents, guaranteeing consistent availability of raw materials for continuous manufacturing operations. Additionally, the simplified post-treatment process avoids the generation of large volumes of waste acid, reducing environmental compliance costs and waste disposal fees while aligning with increasingly strict global sustainability mandates for chemical production.
- Cost Reduction in Manufacturing: The substitution of high-pressure hydrogen gas with solid formate donors eliminates the need for costly compression equipment and specialized safety monitoring systems, resulting in substantial operational expenditure reductions. By enabling the continuous regeneration and reuse of the palladium catalyst, the process minimizes the consumption of expensive precious metals, directly lowering the variable cost per kilogram of the final product. The recovery and recycling of alcohol solvents further contribute to cost efficiency by reducing raw material procurement needs and minimizing waste treatment expenses associated with solvent disposal. These cumulative efficiencies create a leaner cost structure that allows for more competitive pricing in the global agrochemical intermediate market without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: Utilizing stable, solid hydrogen donors like ammonium formate mitigates the logistical risks associated with transporting and storing flammable compressed gases, ensuring uninterrupted production schedules even during supply chain disruptions. The robustness of the catalyst system, which tolerates repeated recycling cycles, reduces dependency on frequent catalyst replenishment shipments, thereby simplifying inventory management and reducing lead times for production runs. Moreover, the compatibility of this method with standard glass-lined or stainless steel reactors commonly found in fine chemical plants facilitates rapid technology transfer and scale-up without the need for bespoke equipment fabrication. This flexibility ensures that manufacturing partners can respond swiftly to fluctuations in market demand, securing a reliable supply of high-purity intermediates for downstream pesticide formulation.
- Scalability and Environmental Compliance: The atmospheric pressure operation of this CTH process inherently scales more safely than high-pressure hydrogenation, allowing for larger batch sizes without exponential increases in safety risks or engineering complexity. The reduction in hazardous waste generation, particularly the avoidance of waste acid streams from extraction processes, simplifies environmental permitting and reduces the burden on wastewater treatment facilities. By converting potential waste by-products into commercially viable chloropyridine co-products, the process maximizes atom economy and supports circular economy principles within the chemical manufacturing sector. This alignment with green chemistry principles enhances the corporate sustainability profile of manufacturers, meeting the rigorous environmental, social, and governance (ESG) criteria demanded by multinational agrochemical corporations.
Frequently Asked Questions (FAQ)
The following questions address critical technical and operational concerns regarding the implementation of this CTH technology for 2,3-dichloropyridine production. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for evaluating process feasibility. Understanding these details is essential for technical teams assessing the viability of adopting this safer and more efficient synthetic route for commercial scale-up.
Q: Why is Catalytic Transfer Hydrogenation safer than gaseous hydrogen reduction?
A: Traditional methods utilize flammable and explosive gaseous hydrogen under pressure, requiring specialized high-risk equipment. The CTH method employs stable organic formate salts as hydrogen donors under atmospheric pressure, significantly mitigating safety hazards and equipment costs.
Q: Can the Pd/C catalyst be reused in this process?
A: Yes, the patent explicitly states that the Pd/C catalyst can be recovered by hot filtration, washed to remove salts, and regenerated for continuous use in subsequent batches, enhancing process economics.
Q: What are the main by-products formed during this synthesis?
A: The reaction produces minor amounts of other chloropyridines such as 3-chloropyridine and 2,5-dichloropyridine. These by-products possess independent economic value and can be separated and sold, ensuring full utilization of raw materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dichloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced agrochemical intermediates requires more than just laboratory success; it demands proven industrial scalability and unwavering quality control. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the CTH method for 2,3-dichloropyridine are translated into reliable supply chains. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying every batch against the highest international standards, guaranteeing that the impurity profiles meet the exacting requirements of global pesticide registrars. We understand the critical nature of supply continuity in the agrochemical sector and have optimized our operations to deliver consistent quality and volume, mitigating the risks associated with single-source dependencies.
We invite forward-thinking procurement leaders to collaborate with us on optimizing their supply chains through advanced manufacturing technologies. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your project milestones. Let us demonstrate how our commitment to safety, efficiency, and quality can become a cornerstone of your sourcing strategy for high-value chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
