Advanced Heavy Metal Free Synthesis Of Aminoketones For Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking robust synthetic routes that balance high purity with environmental sustainability, and patent CN1032001C presents a compelling solution for the production of aminoketones used as central muscle relaxants. This intellectual property discloses a novel preparation method that fundamentally alters the traditional approach to synthesizing these critical pharmacophores by eliminating the reliance on hazardous heavy metal oxidants. Historically, the oxidation of secondary alcohols to ketones, a prerequisite step for aminoketone synthesis, often depended on chromium-based reagents which pose significant toxicity and disposal challenges. The disclosed innovation replaces these dangerous oxidants with a hypohalite-based system catalyzed by pyridine, ensuring that the resulting intermediates are free from heavy metal contamination. This shift is not merely a chemical substitution but a strategic enhancement of the safety profile and regulatory compliance of the manufacturing process. By integrating this heavy-metal-free oxidation with a streamlined aminomethylation sequence, the technology offers a pathway to produce high-purity pharmaceutical intermediates with reduced environmental impact. 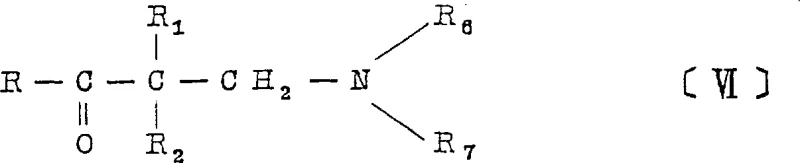
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for preparing aminoketones have long been plagued by inefficiencies related to purification complexity and toxicological concerns. Conventional wisdom dictated the use of strong, stoichiometric oxidants such as chromic acid to convert the precursor alcohols into the necessary ketone intermediates. While chemically effective, the use of chromium introduces severe complications regarding waste stream management and product safety, as residual heavy metals must be rigorously removed to meet pharmacopeial standards. Furthermore, subsequent steps in classical routes often involve the formation of amine hydrochloride salts, necessitating an additional neutralization step using alkaline agents like sodium hydroxide or ammonia to liberate the free base. This extra unit operation increases solvent consumption, extends cycle times, and introduces potential impurities from the neutralization reagents. The cumulative effect of these drawbacks results in a manufacturing process that is costly, environmentally burdensome, and operationally cumbersome for large-scale production facilities aiming for lean manufacturing principles.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a hypohalite oxidant in conjunction with a pyridine catalyst to achieve the alcohol-to-ketone transformation under mild conditions. This chemical strategy effectively bypasses the need for toxic heavy metals, thereby simplifying the downstream purification process and significantly lowering the risk of metal contamination in the final active pharmaceutical ingredient. Moreover, the process is designed to facilitate the direct formation of the free base aminoketone without passing through a stable salt intermediate. By reacting the in-situ generated ketone with an amine and formaldehyde in a controlled manner, the synthesis avoids the laborious salt-formation and neutralization cycle entirely. This telescoping of reactions not only enhances the overall yield by minimizing material loss during isolation steps but also drastically reduces the volume of solvents and reagents required. The result is a cleaner, more efficient synthetic route that aligns perfectly with modern green chemistry initiatives and cost-reduction mandates in the fine chemical sector.
Mechanistic Insights into Hypohalite-Pyridine Catalyzed Oxidation
The core of this technological advancement lies in the mechanistic interplay between the hypohalite species and the pyridine catalyst during the oxidation phase. The reaction proceeds through the generation of an active oxidizing species, likely an N-halopyridinium intermediate, which facilitates the selective abstraction of hydrogen from the alpha-carbon of the alcohol substrate. This mechanism ensures high chemoselectivity, preventing over-oxidation or degradation of sensitive functional groups that might be present on the complex aromatic or heterocyclic rings typical of muscle relaxant intermediates. The use of pyridine or its acid addition salts serves to buffer the reaction medium and stabilize the transition state, allowing the oxidation to proceed efficiently at temperatures ranging from -30°C to 40°C. This mild thermal window is crucial for maintaining the integrity of the starting materials and preventing exothermic runaways, which is a key consideration for process safety during scale-up. The ability to tune the reaction by selecting specific pyridine derivatives further allows chemists to optimize kinetics for different substrate classes, providing a versatile platform for synthesizing a wide library of aminoketone analogues.
Following the oxidation, the mechanistic pathway transitions seamlessly into the aminomethylation step, commonly known as the Mannich reaction. In this phase, the newly formed ketone reacts with a secondary amine and formaldehyde in an aqueous or alcoholic medium. The absence of strong acids typically required for Mannich reactions in older methods is notable here, as the reaction conditions are sufficiently mild to proceed without degrading the sensitive ketone functionality. The mechanism involves the formation of an iminium ion intermediate from the amine and formaldehyde, which then undergoes nucleophilic attack by the enol form of the ketone. Crucially, the process conditions are optimized to yield the free base directly, rather than precipitating as a hydrochloride salt. This eliminates the thermodynamic and kinetic barriers associated with salt cracking, ensuring that the final product is obtained in a high state of purity with minimal effort. 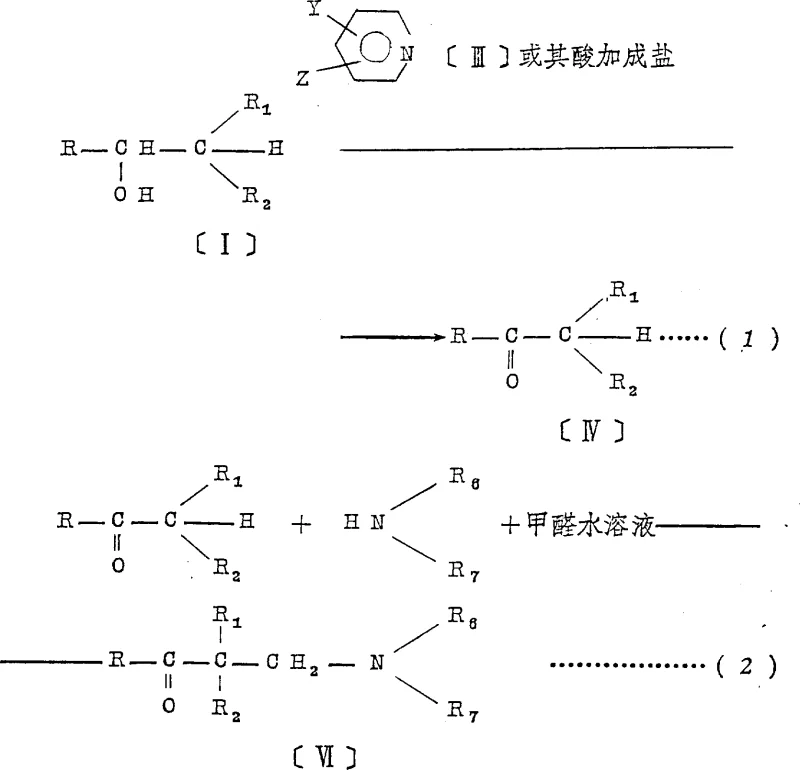
How to Synthesize Aminoketones Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent addition rates and temperature control to maximize efficiency and safety. The process begins with the dissolution of the starting alcohol in a suitable organic solvent such as dichloromethane or ethyl acetate, followed by the controlled addition of the hypohalite oxidant and the pyridine catalyst. Maintaining the reaction temperature within the specified range is critical to prevent side reactions and ensure complete conversion to the ketone intermediate. Once the oxidation is complete, the amine and formaldehyde solution are introduced to the same reaction vessel, leveraging the one-pot capability of the method to save time and resources. Detailed standardized synthesis steps see the guide below.
- Oxidize the starting alcohol intermediate using a hypohalite oxidant in the presence of a pyridine catalyst to form the ketone intermediate.
- Without isolating the ketone, react the mixture with an amine and aqueous formaldehyde to perform the aminomethylation.
- Isolate the final free base aminoketone product directly, bypassing the need for salt formation and subsequent neutralization steps.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that resonate deeply with procurement managers and supply chain directors focused on cost optimization and reliability. The elimination of heavy metal oxidants translates directly into reduced costs associated with hazardous waste disposal and specialized scrubbing systems, which are often significant line items in the operational budget of a chemical manufacturing plant. Furthermore, the simplification of the purification workflow means that fewer unit operations are required, leading to lower utility consumption and reduced labor hours per batch. The use of commodity chemicals like sodium hypochlorite and pyridine ensures that the raw material supply chain is robust and less susceptible to the volatility seen with specialized reagents. This stability in sourcing contributes to a more predictable production schedule and enhances the overall reliability of supply for downstream pharmaceutical customers who depend on consistent quality and delivery.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of expensive and hazardous reagents along with the associated waste treatment costs. By avoiding the use of chromium-based oxidants, manufacturers eliminate the need for complex heavy metal scavenging steps and the regulatory burden of handling toxic waste streams. Additionally, the direct isolation of the free base aminoketone removes the necessity for purchasing neutralization agents and the energy costs associated with additional filtration and drying steps. These cumulative savings result in a significantly lower cost of goods sold (COGS), allowing for more competitive pricing in the global market while maintaining healthy profit margins. The streamlined nature of the process also reduces the capital expenditure required for equipment, as fewer reactors and separation units are needed to achieve the same output volume.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, commodity-grade raw materials rather than niche specialty chemicals. Sodium hypochlorite and pyridine are produced on a massive industrial scale globally, ensuring that supply disruptions are minimal compared to proprietary catalysts or complex organometallic reagents. This accessibility allows for flexible sourcing strategies, where procurement teams can negotiate better terms by leveraging multiple suppliers. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures that could disrupt delivery schedules. This reliability is paramount for maintaining long-term contracts with major pharmaceutical companies that require guaranteed continuity of supply for their critical drug pipelines.
- Scalability and Environmental Compliance: Scaling this process from kilogram to multi-ton production is facilitated by the inherent safety and simplicity of the chemistry involved. The mild reaction temperatures and the use of non-explosive reagents reduce the engineering controls required for large-scale reactors, making it easier to transfer technology from R&D to commercial manufacturing. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding heavy metal discharge and solvent emissions. By generating a cleaner waste profile, manufacturers can more easily obtain and maintain the necessary environmental permits, avoiding potential fines or shutdowns. This compliance advantage not only protects the company's reputation but also future-proofs the manufacturing asset against tightening environmental legislation, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heavy-metal-free synthesis route. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on how this method compares to industry standards. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for their specific product portfolios. The insights provided here aim to bridge the gap between theoretical patent claims and practical industrial application, ensuring that stakeholders have a realistic view of the technology's capabilities.
Q: Why is the hypohalite oxidation method preferred over chromic acid for pharmaceutical intermediates?
A: The hypohalite method eliminates the use of toxic heavy metals like chromium, which simplifies waste treatment and ensures the final product meets stringent residual metal specifications required for pharmaceutical applications.
Q: How does this process reduce manufacturing costs compared to traditional methods?
A: By avoiding the formation of hydrochloride salts during the aminomethylation step, the process removes the need for a separate neutralization and purification stage, thereby reducing solvent consumption and processing time.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the use of readily available reagents like sodium hypochlorite and the ability to perform reactions in common organic solvents makes this route highly scalable and robust for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminoketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical intermediate market. Our team of expert chemists has thoroughly evaluated the heavy-metal-free oxidation route described in patent CN1032001C and confirmed its potential for delivering high-purity aminoketones at a commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our rigorous QC labs and stringent purity specifications guarantee that every batch of aminoketone intermediate meets the exacting standards required for central muscle relaxant formulations. By leveraging this innovative chemistry, we can offer our partners a superior product profile that minimizes impurity risks and maximizes process efficiency.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this optimized synthesis can enhance your project's economics. Whether you are looking to secure a reliable source for existing programs or develop new analogues using this versatile platform, NINGBO INNO PHARMCHEM is equipped to support your goals with speed and precision. Contact us today to discuss how we can collaborate to bring safer, more cost-effective muscle relaxant intermediates to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
