Advanced Dinuclear Aryl Ruthenium Complexes: Revolutionizing Anticancer Drug Intermediates Manufacturing
Advanced Dinuclear Aryl Ruthenium Complexes: Revolutionizing Anticancer Drug Intermediates Manufacturing
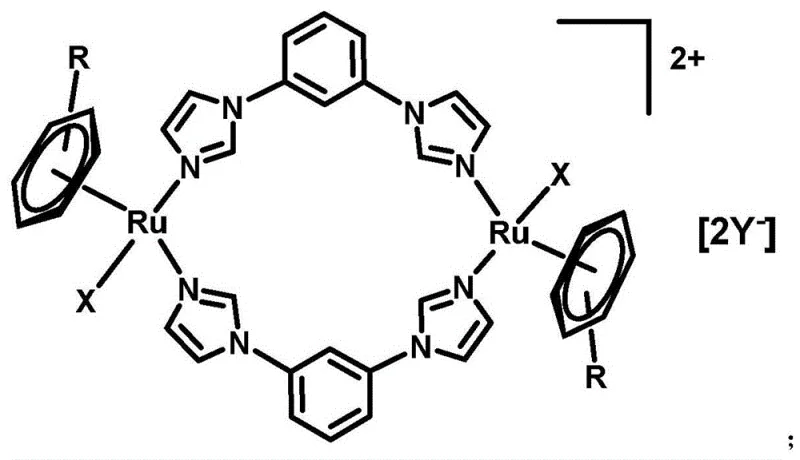
The pharmaceutical industry is constantly seeking next-generation metallodrugs that can overcome the limitations of traditional platinum-based therapies. Patent CN107286199B introduces a breakthrough class of dinuclear aryl ruthenium metal complexes that demonstrate exceptional promise as anticancer agents. These complexes are characterized by a unique bowl-shaped structural motif that facilitates selective interaction with biological targets while minimizing off-target toxicity. The core innovation lies in the coordination of ruthenium centers with 1,3-bis(1H-imidazol-1-yl)benzene ligands, creating a stable yet reactive framework capable of disrupting cancer cell proliferation. As a reliable pharmaceutical intermediate supplier, understanding the structural nuances of these compounds is critical for developing effective therapeutic formulations. The versatility of this chemical platform allows for the modulation of physicochemical properties, particularly water solubility, through the strategic selection of counter-anions, thereby addressing key formulation challenges in drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional anticancer metallodrugs, predominantly based on platinum, have long been the cornerstone of chemotherapy regimens; however, their clinical utility is increasingly compromised by significant drawbacks. The primary concern is the severe systemic toxicity associated with platinum agents, manifesting as nephrotoxicity, neurotoxicity, and hematological disorders such as thrombocytopenia. Furthermore, the development of drug resistance in tumor cells limits the long-term efficacy of these treatments, necessitating higher dosages that exacerbate side effects. From a manufacturing perspective, the synthesis of complex organometallic anticancer agents often involves multi-step procedures with harsh reaction conditions, leading to lower overall yields and higher production costs. The inability to finely tune the solubility and reactivity of these conventional drugs restricts their bioavailability and targeting precision, creating a pressing need for innovative alternatives that offer improved safety profiles and synthetic efficiency.
The Novel Approach
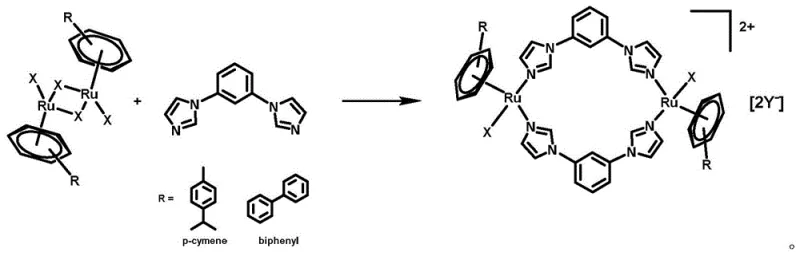
The synthesis method disclosed in the patent represents a paradigm shift towards more efficient and selective organometallic drug design. By utilizing a dinuclear ruthenium architecture bridged by rigid imidazole-based ligands, the new approach achieves a balance between stability and biological activity that is superior to mononuclear counterparts. The process is remarkably streamlined, involving the direct reaction of aryl ruthenium dimers with the 1,3-bib ligand in common organic solvents under mild inert conditions. This simplicity not only enhances operational safety but also significantly improves the scalability of the production process. The resulting complexes exhibit a "bowl-shaped" conformation that is conducive to specific DNA binding interactions, as evidenced by circular dichroism studies. This structural advantage translates into high selectivity for cancer cells, effectively sparing normal healthy tissue and reducing the therapeutic index concerns that plague older generations of metallodrugs.
Mechanistic Insights into DNA Interaction and Selective Toxicity
The biological efficacy of these dinuclear aryl ruthenium complexes is rooted in their sophisticated mechanism of action, which differs fundamentally from simple alkylation. Spectroscopic analysis, specifically circular dichroism (CD), reveals that these complexes interact with CT-DNA through an intercalation mode, where the extended aromatic systems of the ligands insert themselves between DNA base pairs. This interaction induces conformational changes in the DNA helix, evidenced by the gradual decrease in the intensity of positive and negative CD signals without significant peak shifting as the complex concentration increases. Such binding disrupts essential cellular processes like replication and transcription, leading to apoptosis in rapidly dividing cancer cells. Crucially, the ruthenium center offers multiple valence states (RuII, RuIII, RuIV) in vivo, which may facilitate redox activation mechanisms that are less prone to the resistance pathways developed against platinum drugs. This multi-target capability ensures that the drug remains effective even in resistant tumor lines.
Beyond mere DNA binding, the selectivity of these complexes is a defining feature that addresses the toxicity issues of conventional chemotherapy. In vitro cytotoxicity assays using the MTT method demonstrate that specific complexes, such as Complex 2, exhibit potent antiproliferative activity against various cancer cell lines including Hela, HepG2, MCF-7, and A549. More importantly, these complexes show significantly lower cytotoxicity towards normal human liver cells (L02), with selectivity factors far exceeding those of cisplatin. For instance, Complex 2 displays an IC50 value greater than 215.9 μM on normal L02 cells while maintaining moderate activity against A549 lung cancer cells. This differential toxicity suggests that the complex exploits specific metabolic or transport differences between malignant and normal cells, potentially offering a safer therapeutic window. The ability to modulate this selectivity by changing the R group (p-cymene or biphenyl) and the anion Y provides medicinal chemists with a powerful toolkit for optimizing drug candidates.
How to Synthesize Dinuclear Aryl Ruthenium Complex Efficiently
The patented synthesis route offers a robust and reproducible methodology for producing these high-value intermediates, making it highly attractive for commercial scale-up. The process begins with the dissolution of an aryl ruthenium dimer precursor in a suitable organic solvent such as acetonitrile, methanol, or dichloromethane under an inert argon atmosphere to prevent oxidation. The 1,3-bis(1H-imidazol-1-yl)benzene ligand is then introduced, and the mixture is heated to temperatures ranging from 25°C to 80°C for a duration of 1 to 80 hours, depending on the specific reactivity of the precursors. Following the coordination step, an anion exchange is performed by adding salts like AgCF3SO3 or NaCl to tailor the solubility profile of the final product. The crude material is subsequently purified via column chromatography to yield the target complex as a high-purity yellow powder. Detailed standardized synthesis steps for specific analogues are provided in the guide below.
- Dissolve the aryl ruthenium dimer precursor in an organic solvent such as acetonitrile or dichloromethane under an inert gas atmosphere to form the initial solution.
- Add the 1,3-bis(1H-imidazol-1-yl)benzene (1,3-bib) ligand to the initial solution and react at temperatures between 25°C and 80°C for 1 to 80 hours.
- Introduce the desired anion salt (e.g., AgCF3SO3, NaCl) to the mixture, stir at room temperature, filter, concentrate the filtrate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis technology presents compelling opportunities for cost optimization and risk mitigation. The streamlined nature of the reaction sequence eliminates the need for exotic catalysts or extreme pressure conditions, which are often bottlenecks in fine chemical manufacturing. By relying on commercially available starting materials like aryl ruthenium dimers and simple imidazole derivatives, the supply chain becomes more resilient to raw material fluctuations. The high yields reported in the patent examples, reaching over 90% in optimized conditions, directly correlate to reduced waste generation and lower cost of goods sold (COGS). Furthermore, the flexibility in solvent choice allows manufacturers to utilize existing infrastructure without significant capital expenditure on new reactor types. This adaptability ensures a consistent supply of high-quality intermediates, crucial for maintaining uninterrupted drug development pipelines.
- Cost Reduction in Manufacturing: The synthesis protocol drastically simplifies the production workflow by combining ligand coordination and anion exchange into a seamless one-pot or two-step sequence. This reduction in unit operations minimizes labor costs, energy consumption, and solvent usage, leading to substantial economic benefits. The elimination of complex purification steps beyond standard column chromatography further enhances the cost-efficiency profile, making these ruthenium complexes competitive with traditional small molecule intermediates.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable precursors and common organic solvents mitigates the risks associated with sourcing hazardous or unstable reagents. The inert atmosphere requirement is a standard industrial practice that does not impose undue burdens on facility capabilities. Consequently, manufacturers can secure long-term contracts with confidence, knowing that the production process is robust against minor variations in environmental conditions, ensuring timely delivery of critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, transitioning smoothly from gram-scale laboratory synthesis to kilogram or ton-scale commercial production. The use of recyclable solvents and the generation of minimal heavy metal waste align with modern green chemistry principles and stringent environmental regulations. This compliance reduces the regulatory burden and potential fines, while the high atom economy of the coordination reaction supports sustainable manufacturing goals increasingly demanded by global stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dinuclear ruthenium complexes. The answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for integrating this technology into existing drug discovery and development workflows.
Q: What is the primary advantage of this dinuclear ruthenium complex over traditional platinum drugs?
A: Unlike platinum drugs which suffer from severe side effects like nephrotoxicity and drug resistance, this dinuclear aryl ruthenium complex exhibits high selectivity for cancer cells over normal cells, significantly reducing toxic side effects while maintaining potent anticancer activity through DNA interaction.
Q: How does the synthesis method ensure high purity and yield?
A: The patented process utilizes a straightforward ligand substitution reaction under inert conditions followed by anion exchange. The use of standard organic solvents and column chromatography purification allows for the isolation of high-purity yellow powder complexes with yields reaching up to 92.1% in optimized examples.
Q: Can the water solubility of the complex be adjusted for different drug formulations?
A: Yes, the complex features a bowl-shaped structure where the water solubility can be precisely tuned by varying the counter-anions (Y), such as Cl-, BF4-, PF6-, or CF3SO3-, allowing manufacturers to tailor the physicochemical properties for specific pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dinuclear Aryl Ruthenium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organometallic therapeutics in the fight against cancer. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom anion variants or specific isotopic labeling for mechanistic studies, our flexible manufacturing capabilities allow us to tailor our services to your exact project requirements.
We invite you to collaborate with us to accelerate your anticancer drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis for your specific target molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis of dinuclear aryl ruthenium complexes can enhance your project's value proposition and speed to market.
