Advanced Synthesis of Dinuclear Aryl Ruthenium Complexes for Next-Generation Anticancer Therapeutics
Advanced Synthesis of Dinuclear Aryl Ruthenium Complexes for Next-Generation Anticancer Therapeutics
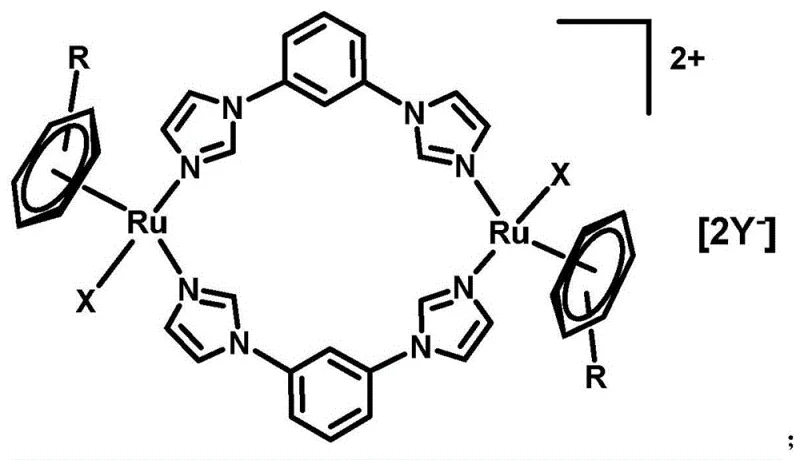
The pharmaceutical landscape is witnessing a paradigm shift away from traditional platinum-based chemotherapies towards more selective and less toxic metal-based alternatives, driven by the urgent need to overcome drug resistance and severe systemic toxicity. Patent CN107286199B introduces a groundbreaking class of dinuclear aryl ruthenium metal complexes that offer a compelling solution to these longstanding challenges in oncology. These novel compounds feature a unique bowl-shaped architecture where two ruthenium centers are bridged by 1,3-bis(1H-imidazol-1-yl)benzene ligands, creating a robust structure capable of interacting effectively with biological targets. The innovation lies not just in the molecular design but in the remarkably efficient synthesis pathway that allows for the precise tuning of physicochemical properties, such as water solubility, through simple anion exchange. This technological breakthrough positions these complexes as prime candidates for the next generation of anticancer agents, offering a reliable pharmaceutical intermediate supplier with the capability to deliver high-purity materials for clinical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the oncology sector has relied heavily on platinum-based drugs like cisplatin, carboplatin, and oxaliplatin, which function by cross-linking DNA to inhibit cell division. However, the clinical utility of these agents is severely compromised by their lack of selectivity, leading to devastating side effects such as nephrotoxicity, neurotoxicity, and myelosuppression in patients. Furthermore, the rapid development of tumor resistance to platinum agents has rendered many treatments ineffective, necessitating higher doses that exacerbate toxicity without improving survival rates. From a manufacturing perspective, the purification of platinum drugs often involves complex downstream processing to remove trace heavy metals, adding significant cost and environmental burden to the supply chain. The rigid coordination chemistry of platinum also limits the structural diversity available to medicinal chemists, restricting the ability to fine-tune pharmacokinetic profiles for specific cancer types.
The Novel Approach
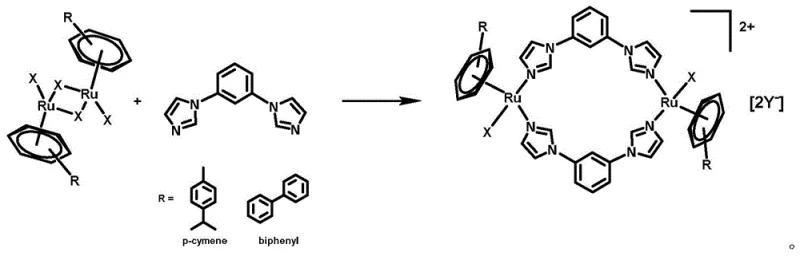
The synthesis method disclosed in the patent represents a significant departure from these conventional limitations by leveraging the versatile coordination chemistry of ruthenium. The process involves a straightforward ligand substitution reaction where an aryl ruthenium dimer reacts with a bis-imidazole ligand under mild conditions, typically between 25°C and 80°C. This approach eliminates the need for harsh reaction conditions or expensive transition metal catalysts often required in C-H activation strategies. Crucially, the method allows for the modular introduction of different anions (such as Cl-, Br-, I-, PF6-, BF4-) in a final step, enabling manufacturers to tailor the water solubility and lipophilicity of the final drug substance without altering the core synthetic route. This flexibility is a game-changer for cost reduction in pharmaceutical intermediate manufacturing, as it streamlines the production of multiple analogues from a common precursor.
Mechanistic Insights into Dinuclear Ruthenium Coordination and DNA Binding
The efficacy of these dinuclear complexes stems from their unique ability to interact with DNA through mechanisms distinct from platinum drugs. The ruthenium centers, stabilized by the arene ligands (p-cymene or biphenyl) and the nitrogen donors from the imidazole rings, create a cationic species that is attracted to the negatively charged phosphate backbone of DNA. Unlike the covalent binding typical of platinum, these ruthenium complexes often engage in non-covalent interactions, including intercalation of the aromatic ligands between DNA base pairs. This mode of binding induces conformational changes in the DNA helix, as evidenced by circular dichroism studies which show a decrease in signal intensity without peak shifting upon complex addition. Such interactions disrupt the replication machinery of cancer cells while sparing normal tissue, attributed to the faster ligand exchange kinetics of ruthenium compared to platinum which allows for better control over reactivity in the biological milieu.
Furthermore, the presence of two metal centers in close proximity within the bowl-shaped structure facilitates multi-point binding to the DNA double helix, potentially enhancing the stability of the drug-DNA adduct and overcoming resistance mechanisms that rely on DNA repair enzymes. The variation of the 'R' group (p-cymene vs. biphenyl) and the 'X' halide ligands allows for fine-tuning of the electronic environment around the metal center, directly influencing the redox potential and thus the activation of the prodrug within the hypoxic environment of solid tumors. This sophisticated level of control over the mechanistic action ensures that the resulting pharmaceutical intermediates possess the high purity and specific activity required for rigorous preclinical and clinical evaluation.
How to Synthesize Dinuclear Aryl Ruthenium Complex Efficiently
The patented synthesis route offers a robust and reproducible method for producing these high-value intermediates, characterized by high yields and operational simplicity. The process begins with the dissolution of the aryl ruthenium dimer precursor in a polar organic solvent such as acetonitrile or methanol under an inert argon atmosphere to prevent oxidation. The key ligand, 1,3-bis(1H-imidazol-1-yl)benzene, is then introduced, and the mixture is heated to facilitate the coordination of the imidazole nitrogens to the ruthenium centers. Following the formation of the cationic intermediate, a metathesis reaction is performed by adding a specific anionic salt to precipitate or stabilize the desired counter-ion. The crude product is subsequently purified via column chromatography, yielding the final complex as a stable yellow powder with yields often exceeding 80%, demonstrating the efficiency of this protocol for commercial scale-up of complex organometallic intermediates.
- Dissolve aryl ruthenium dimer in organic solvent (e.g., acetonitrile, methanol) under inert gas to form an initial solution.
- Add 1,3-bis(1H-imidazol-1-yl)benzene (1,3-bib) ligand to the solution and react at 25-80°C for 1-80 hours.
- Add anionic salt (e.g., AgCF3SO3, NaCl), stir at room temperature, filter, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis technology offers substantial strategic benefits beyond mere technical performance. The reliance on readily available starting materials, such as commercially sourced aryl ruthenium dimers and simple imidazole derivatives, mitigates the risk of raw material shortages that often plague the supply of exotic catalytic intermediates. The reaction conditions are mild and do not require specialized high-pressure equipment or cryogenic cooling, allowing for production in standard glass-lined reactors found in most multipurpose pharmaceutical facilities. This compatibility with existing infrastructure drastically simplifies the technology transfer process and reduces the capital expenditure required for new production lines. Moreover, the high yields reported in the patent examples indicate a material-efficient process that minimizes waste generation, aligning with modern green chemistry principles and reducing the costs associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts (other than the ruthenium substrate itself which is incorporated into the product) and the use of common solvents like ethanol and methanol significantly lower the bill of materials. The high conversion rates mean less raw material is wasted, and the simplified purification steps reduce the consumption of silica and eluents. This streamlined workflow translates to a lower cost of goods sold (COGS), providing a competitive pricing advantage for the final API or intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The synthetic route avoids the use of unstable reagents or those with long lead times, ensuring a consistent and predictable production schedule. The ability to tune the final product properties by simply changing the anion salt in the last step allows manufacturers to respond quickly to changing formulation requirements from downstream partners. This agility is crucial for maintaining supply continuity in the fast-paced oncology drug market, where delays in intermediate supply can halt entire clinical trials or commercial launches.
- Scalability and Environmental Compliance: The process generates minimal hazardous byproducts, primarily consisting of inorganic salts that are easy to treat and dispose of in accordance with environmental regulations. The absence of toxic heavy metal catalysts that require stringent removal steps (like scavengers) simplifies the regulatory filing process and reduces the environmental footprint of the manufacturing site. This makes the technology highly attractive for large-scale production, ensuring that the supply chain can meet global demand for these promising anticancer agents sustainably.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dinuclear ruthenium complexes. Understanding these details is essential for R&D teams evaluating the feasibility of incorporating these intermediates into their drug discovery pipelines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of this dinuclear ruthenium complex over traditional platinum drugs?
A: Unlike platinum drugs which suffer from severe side effects like nephrotoxicity and drug resistance, this dinuclear aryl ruthenium complex exhibits high selectivity for cancer cells over normal cells, significantly reducing toxic side effects while maintaining potent anticancer activity through DNA binding mechanisms.
Q: How can the water solubility of the complex be adjusted for different pharmaceutical formulations?
A: The water solubility is tunable by varying the counter-anions (Y) in the complex structure. By selecting different anionic salts such as Cl-, Br-, CF3SO3-, or PF6- during the final synthesis step, manufacturers can optimize the physicochemical properties for specific drug delivery requirements.
Q: Is the synthesis process scalable for commercial production of anticancer intermediates?
A: Yes, the process is highly scalable. It utilizes standard organic solvents like methanol and acetonitrile, operates at moderate temperatures (25-80°C), and achieves high yields (up to 92.1%) without requiring exotic catalysts, making it suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dinuclear Aryl Ruthenium Complex Supplier
As the demand for novel metal-based anticancer agents continues to rise, partnering with an experienced CDMO is critical for navigating the complexities of organometallic synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle air-sensitive reactions and rigorous purification protocols, guaranteeing stringent purity specifications for every batch of dinuclear ruthenium complex we produce. With our rigorous QC labs and commitment to quality, we provide the reliability and consistency that global pharmaceutical companies require for their critical supply chains.
We invite you to collaborate with us to leverage this advanced synthesis technology for your oncology portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your development timelines and optimize your production costs.
