Advanced Biocatalytic Synthesis of (R)-Phenylethylene Glycol for Commercial Scale-Up
Advanced Biocatalytic Synthesis of (R)-Phenylethylene Glycol for Commercial Scale-Up
The global demand for optically pure chiral building blocks has surged dramatically, driven by the stringent regulatory requirements of the pharmaceutical industry and the high-performance specifications of the liquid crystal display sector. Patent CN101230363B represents a significant technological breakthrough in this domain, disclosing a highly efficient method for preparing (R)-phenylethylene glycol through the asymmetric transformation of a specifically engineered recombinant strain. This innovation moves beyond traditional chemical synthesis, leveraging the power of metabolic engineering to achieve superior stereocontrol and environmental sustainability. By utilizing a recombinant Escherichia coli strain harboring the (R)-specific alcohol dehydrogenase gene rcr, the process converts 2-hydroxyacetophenone into the target diol with remarkable precision. The following analysis dissects the technical merits of this patent, offering critical insights for R&D directors seeking robust synthetic routes and procurement managers aiming for cost-effective supply chains. 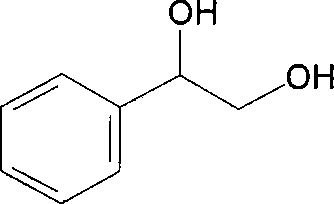
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure phenylethylene glycol has relied heavily on chemical resolution or non-specific reduction methods that suffer from inherent inefficiencies and environmental drawbacks. Traditional chemical synthesis often necessitates the use of stoichiometric amounts of expensive chiral catalysts or resolving agents, which not only drives up the raw material costs but also generates substantial quantities of hazardous waste streams requiring complex disposal protocols. Furthermore, because the phenylethylene glycol structure contains two hydroxyl groups, conventional chemical approaches frequently require selective protection and deprotection steps to prevent side reactions, thereby elongating the synthetic sequence and diminishing the overall atom economy. The use of heavy metal catalysts in some chemical reductions poses severe risks of residual metal contamination, a critical failure point for pharmaceutical intermediates destined for active drug substance manufacturing where impurity profiles are strictly regulated. Additionally, many existing biological methods utilizing wild-type microorganisms exhibit low substrate tolerance, often limiting the substrate concentration to negligible levels such as 0.6 g/L, which renders them economically unviable for large-scale industrial production due to massive solvent and downstream processing volumes.
The Novel Approach
The methodology outlined in CN101230363B fundamentally disrupts these legacy constraints by introducing a tailored recombinant biocatalyst designed for high-efficiency asymmetric reduction. By cloning the rcr gene from Candida parapsilosis into the pET21c vector and expressing it in E. coli BL21, the inventors have created a cellular factory capable of highly stereoselective ketone reduction. A pivotal innovation in this approach is the implementation of a substrate-coupled cofactor regeneration system using isopropanol as an auxiliary substrate. This strategy effectively recycles the necessary nicotinamide cofactors in situ, drastically reducing the requirement for expensive external cofactors and allowing for a significant reduction in the biomass loading needed for the reaction. The process operates under mild physiological conditions, typically around 30°C and pH 8.0, which minimizes energy consumption and prevents the thermal degradation of sensitive intermediates. Most importantly, this recombinant system demonstrates the ability to handle higher substrate concentrations compared to prior art wild-type strains, achieving yields between 70% and 100% with optical purities reaching up to 100% e.e., thereby establishing a viable pathway for the reliable pharmaceutical intermediates supplier market.
Mechanistic Insights into Recombinant Strain Asymmetric Transformation
The core of this technology lies in the precise molecular recognition capabilities of the expressed (R)-specific alcohol dehydrogenase (ADH) within the recombinant E. coli host. The enzyme's active site provides a chiral environment that strictly favors the pro-R hydride transfer from the reduced cofactor to the carbonyl carbon of the 2-hydroxyacetophenone substrate. This enzymatic specificity ensures that the resulting alcohol is formed almost exclusively in the (R)-configuration, bypassing the formation of the unwanted (S)-enantiomer that typically complicates chemical syntheses. The genetic engineering aspect involves the optimization of the expression cassette to ensure high-level production of the soluble, active enzyme, which is critical for maintaining high volumetric productivity in the bioreactor. The use of the T7 promoter system in the pET21c vector allows for tight control over gene expression, preventing the formation of inclusion bodies and ensuring that the biocatalyst remains functional throughout the extended reaction period of 48 hours. This level of control over the biocatalyst's physiology is what enables the consistent production of high-purity OLED material precursors and pharmaceutical intermediates with minimal batch-to-batch variability.
Furthermore, the integration of the isopropanol-driven cofactor regeneration cycle is a masterstroke of process chemistry that enhances the thermodynamic favorability of the reduction. In typical biocatalytic reductions, the stoichiometric consumption of NADPH would make the process prohibitively expensive; however, the ADH utilized in this patent possesses the dual capability of oxidizing isopropanol to acetone while reducing the ketone substrate. This coupling creates a closed loop where the oxidized cofactor generated during the product formation is immediately reduced back to its active form by the oxidation of the cheap and abundant isopropanol. This mechanism not only lowers the cost reduction in chiral building blocks manufacturing but also shifts the reaction equilibrium towards the product side, driving the conversion to near completion. The result is a process that achieves optical purities of 80-100% e.e. and yields of 70-100%, demonstrating that the impurity profile is tightly controlled by the enzyme's intrinsic selectivity rather than downstream purification efforts.
How to Synthesize (R)-Phenylethylene Glycol Efficiently
The practical implementation of this patented technology requires a disciplined approach to strain maintenance and reaction parameter optimization to ensure maximum efficiency. The process begins with the rigorous cultivation of the recombinant E. coli BL21/pET-RCR strain, where induction timing and temperature are critical variables that dictate the final enzyme activity per gram of wet cell weight. Following the harvest of the biocatalyst, the asymmetric transformation is conducted in a buffered aqueous system, typically Tris-HCl at pH 8.0, which provides the optimal ionic environment for enzyme stability. The addition of isopropanol at concentrations between 2.5% and 10% is essential to sustain the cofactor cycle, while the substrate loading can be tuned between 0.5 g/L and 5 g/L depending on the specific purity requirements of the final application. Detailed standardized synthesis steps see the guide below.
- Construct the recombinant plasmid pET-RCR by inserting the rcr gene into vector pET21c and transform into E. coli BL21.
- Culture the recombinant strain in LB medium with ampicillin, induce expression with IPTG at 30-37°C until OD600 reaches 0.6-0.8.
- Perform asymmetric transformation in Tris-HCl buffer (pH 8.0) with 2-hydroxyacetophenone substrate and isopropanol for cofactor regeneration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chemical resolution to this recombinant biocatalytic process offers profound strategic advantages that extend far beyond simple unit cost calculations. The elimination of toxic heavy metal catalysts and hazardous organic solvents significantly simplifies the waste treatment infrastructure required at the manufacturing site, leading to substantial cost savings in environmental compliance and disposal fees. Moreover, the high stereoselectivity of the enzymatic process means that the crude product contains significantly fewer isomeric impurities, which reduces the burden on downstream purification units such as chromatography or crystallization, thereby shortening the overall production cycle time. This streamlined workflow enhances the reliability of supply by reducing the number of potential failure points in the manufacturing process, ensuring that delivery schedules for critical pharmaceutical intermediates are met with greater consistency. The ability to operate at moderate temperatures and atmospheric pressure also reduces the energy intensity of the process, contributing to a lower carbon footprint and aligning with the sustainability goals of modern multinational corporations.
- Cost Reduction in Manufacturing: The implementation of the substrate-coupled cofactor regeneration system using isopropanol eliminates the need for purchasing expensive stoichiometric reducing agents or external cofactors, which are traditionally major cost drivers in fine chemical synthesis. By recycling the catalytic equivalents in situ, the process drastically reduces the raw material bill, allowing for a more competitive pricing structure for the final chiral diol. Additionally, the reduction in cell mass requirement due to improved catalytic efficiency means that less fermentation capacity is needed to produce the same amount of product, optimizing capital utilization rates. The avoidance of complex protection and deprotection chemistry further trims the number of synthetic steps, directly correlating to lower labor costs and reduced consumption of auxiliary chemicals. These factors combine to create a manufacturing protocol that is inherently leaner and more cost-effective than legacy chemical routes.
- Enhanced Supply Chain Reliability: Utilizing a robust recombinant E. coli host ensures a stable and scalable source of the biocatalyst, mitigating the risks associated with the variability of wild-type microbial strains or the supply volatility of rare chemical catalysts. The fermentation process for producing the enzyme is well-established and easily scalable from laboratory shake flasks to industrial fermenters, ensuring that the supply of the biocatalyst can be ramped up quickly to meet surges in market demand. Because the process relies on readily available bulk chemicals like glucose, isopropanol, and standard buffer salts, it is less susceptible to the geopolitical supply chain disruptions that often affect specialized reagents. This resilience makes the technology an ideal choice for securing the long-term supply of high-purity pharmaceutical intermediates, providing peace of mind to procurement teams managing critical inventory levels.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium and the absence of volatile organic compounds (VOCs) in the primary transformation step simplify the engineering controls required for scale-up, facilitating a smoother transition from pilot plant to commercial production. The biodegradable nature of the biological waste streams contrasts sharply with the persistent toxic waste generated by chemical resolution, making it easier to obtain environmental permits and maintain good standing with regulatory agencies. The process parameters, such as pH and temperature, are mild and easy to control even in large vessels, reducing the risk of thermal runaways or safety incidents that could halt production. Consequently, this technology supports the commercial scale-up of complex polymer additives and pharmaceutical ingredients while adhering to the strictest global environmental, health, and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology, derived directly from the patent specifications and industry best practices. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific capabilities of the recombinant strain E.coli BL21/pET-RCR and the optimized reaction conditions described in the intellectual property.
Q: What is the optical purity achievable with this recombinant strain method?
A: According to patent CN101230363B, the optimized process yields (R)-phenylethylene glycol with an optical purity ranging from 80% to 100% e.e., with specific examples demonstrating up to 95.5% e.e.
Q: Why is isopropanol added during the biocatalytic reaction?
A: Isopropanol serves as an auxiliary substrate for coenzyme regeneration. It allows the costly cofactor (NADPH/NADH) to be recycled in situ, significantly reducing the amount of recombinant cells required and improving overall conversion efficiency.
Q: How does this biological method compare to traditional chemical resolution?
A: Traditional chemical methods often require toxic chiral catalysts and protective group strategies which generate hazardous waste. This biocatalytic approach operates under mild conditions (30°C, pH 8.0), eliminates heavy metal contamination, and offers superior stereoselectivity without complex protection-deprotection steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Phenylethylene Glycol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced chiral intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality that spans the entire value chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of CN101230363B can be translated into a robust industrial reality. We understand that for applications in liquid crystal materials and active pharmaceutical ingredients, meeting stringent purity specifications is non-negotiable, which is why our rigorous QC labs employ state-of-the-art chiral HPLC and GC-MS instrumentation to verify every batch. Our facility is equipped to handle the fermentation and downstream processing required for this biocatalytic route, guaranteeing a consistent supply of high-quality material that meets the exacting standards of global innovators.
We invite you to collaborate with us to unlock the full potential of this green synthesis technology for your specific product needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this biocatalytic route for your specific volume requirements. We encourage you to contact us today to discuss your project specifics,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of (R)-phenylethylene glycol that strengthens your competitive position in the global market.
