Revolutionizing Quinoline Production: A Cost-Effective Iron-Catalyzed Aerobic Oxidation Strategy for Global Supply Chains
The global demand for high-quality nitrogen heterocycles continues to surge, driven by their indispensable role in the development of next-generation therapeutics and agrochemicals. Among these scaffolds, the quinoline nucleus stands out as a privileged structure found in numerous bioactive molecules, ranging from antimalarials to kinase inhibitors. However, the industrial realization of complex quinoline derivatives has historically been hindered by synthetic bottlenecks, including harsh reaction conditions, expensive catalysts, and poor atom economy. A groundbreaking solution to these challenges is presented in Chinese Patent CN107628996B, which discloses a highly efficient, one-pot synthesis method for polysubstituted quinolines. This innovative approach leverages the catalytic power of abundant iron salts and utilizes molecular oxygen as a green oxidant, marking a significant departure from traditional methodologies that rely on precious metals or corrosive reagents.
For R&D directors and process chemists, the implications of this technology are profound. By enabling the direct oxidative cyclization of readily available starting materials—specifically acetophenone, aniline derivatives, and dimethyl sulfoxide (DMSO)—this method streamlines the synthetic route considerably. The patent details a robust protocol where the reaction proceeds smoothly under mild thermal conditions (100-140°C) in an oxygen-containing atmosphere. This not only simplifies the operational workflow but also aligns perfectly with the principles of green chemistry, offering a sustainable pathway for the commercial scale-up of complex pharmaceutical intermediates. As we delve deeper into the technical specifics, it becomes evident that this iron-catalyzed strategy represents a paradigm shift in how we approach the construction of the quinoline core.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline derivatives has been dominated by classical methods such as the Skraup synthesis, which, while effective, suffers from severe drawbacks including the use of concentrated sulfuric acid, high temperatures, and the generation of substantial toxic waste. Furthermore, modern variations often rely on transition metal catalysts that are either prohibitively expensive or environmentally hazardous. For instance, prior art methods frequently employ rare earth catalysts to facilitate the condensation of substituted anilines with α,β-unsaturated carbonyl compounds.
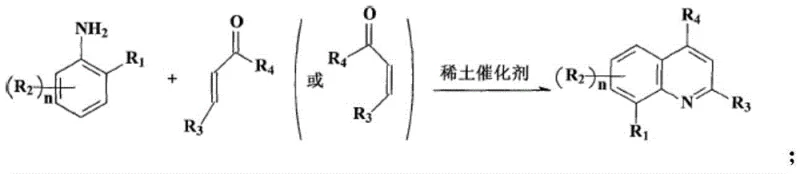
As illustrated in the reaction scheme above, these conventional routes often necessitate the pre-preparation of specialized substrates like α,β-unsaturated aldehydes or ketones, which adds steps, cost, and potential safety risks to the overall process. Additionally, methods utilizing acidic ionic liquids or corrosive triflic acid create significant challenges for equipment maintenance and waste disposal, making them less attractive for large-scale cost reduction in pharmaceutical intermediate manufacturing. The reliance on sealed systems for unstable imine substrates further complicates the engineering requirements, limiting the flexibility and scalability of these traditional approaches.
The Novel Approach
In stark contrast, the methodology disclosed in CN107628996B offers a streamlined, atom-economical alternative that bypasses these historical limitations. The core innovation lies in the direct utilization of acetophenone, aniline, and DMSO in a single reaction vessel, catalyzed by inexpensive ferric or ferrous salts. This one-pot strategy eliminates the need for pre-functionalized substrates and harsh acidic environments, replacing them with a mild, aerobic oxidation process.
The reaction mechanism involves the oxidative dehydrogenation and condensation of the acetyl group from acetophenone, the amino group from the aniline, and remarkably, a methyl group from the DMSO solvent itself to construct the pyridine ring. This unique integration of the solvent into the product skeleton not only enhances atom economy but also simplifies the purification process. By operating under an oxygen atmosphere (preferably air) at moderate temperatures (optimized around 120°C), this method achieves impressive yields (up to 65% in optimized examples) while maintaining a safety profile that is far superior to high-pressure or strong-acid alternatives. This represents a tangible leap forward for reliable pharmaceutical intermediate suppliers seeking to optimize their production portfolios.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Cyclization
The success of this transformation hinges on the dual functionality of the iron catalyst and the oxidant. Mechanistically, the ferric salt (such as FeCl3·6H2O) acts as a Lewis acid to activate the carbonyl group of the acetophenone, facilitating nucleophilic attack by the aniline nitrogen. Simultaneously, the iron species mediates the electron transfer processes required for oxidative dehydrogenation. The presence of molecular oxygen is critical; it serves as the terminal electron acceptor, regenerating the active catalytic species and driving the equilibrium towards the fully aromatic quinoline system. Experimental data from the patent indicates that without the iron catalyst or with alternative metals like cobalt or nickel, the reaction efficiency drops precipitously, underscoring the specific electronic properties of iron that are essential for this cycle.
Furthermore, the role of DMSO extends beyond that of a mere solvent. It participates actively in the bond-forming events, likely undergoing activation by the iron center to provide the necessary carbon fragment for ring closure. This intricate interplay between the catalyst, the oxidant, and the solvent ensures high selectivity for the desired polysubstituted quinoline product while minimizing the formation of polymeric byproducts or over-oxidized species. For process chemists, understanding this mechanism is key to controlling the impurity profile. The mild nature of the oxidation prevents the degradation of sensitive functional groups (such as halogens or alkyl chains) on the aniline ring, ensuring that the final high-purity quinoline derivatives retain the structural integrity required for downstream biological testing and drug development.
How to Synthesize Polysubstituted Quinoline Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometric ratios and reaction parameters identified during the patent's optimization phase. The process is designed to be operationally simple, requiring standard glassware and heating equipment without the need for specialized high-pressure reactors. The following guide outlines the generalized procedure derived from the patent's most successful experimental embodiments, providing a roadmap for reproducing these high-yield results.
- Dissolve acetophenone, aniline compound, and ferric salt catalyst (e.g., FeCl3·6H2O) in dimethyl sulfoxide (DMSO) within a reaction vessel.
- Heat the mixture to a temperature range of 100-130°C under an oxygen-containing atmosphere (air or pure oxygen) with stirring for 18-30 hours.
- Upon completion, cool the reaction, dilute with ethyl acetate, wash with water, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iron-catalyzed aerobic oxidation method offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The shift from precious metal catalysts to commodity iron salts fundamentally alters the cost structure of quinoline production. Iron salts are among the most abundant and inexpensive transition metals available globally, ensuring a stable and predictable supply chain that is immune to the geopolitical volatility often associated with rare earth elements or noble metals like palladium and platinum.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the use of air as a free oxidant drastically lowers the variable costs associated with each batch. Furthermore, the one-pot nature of the reaction reduces solvent consumption and labor hours by removing intermediate isolation and purification steps. This streamlined workflow translates into substantial operational savings, allowing manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates without compromising on quality margins.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis—acetophenone, aniline derivatives, and DMSO—are bulk commodity chemicals produced at massive scales worldwide. This ubiquity ensures that supply disruptions are minimal, providing a secure foundation for long-term production planning. Unlike methods relying on custom-synthesized unsaturated ketones or unstable imines, this route utilizes shelf-stable reagents that can be sourced from multiple vendors, thereby reducing lead time for high-purity quinoline derivatives and mitigating the risk of single-source dependency.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of corrosive mineral acids or toxic heavy metals simplify the engineering requirements for scale-up. Waste streams are significantly cleaner, primarily consisting of iron salts and organic residues that are easier to treat than the acidic sludge generated by Skraup synthesis. This environmental compatibility facilitates regulatory approval and reduces the overhead costs associated with waste disposal and environmental compliance, making it an ideal candidate for sustainable, large-volume manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aimed at clarifying the practical benefits for industrial partners.
Q: What are the primary cost advantages of this iron-catalyzed method compared to traditional Skraup synthesis?
A: Unlike the traditional Skraup synthesis which requires concentrated sulfuric acid and harsh high-temperature conditions, this method utilizes inexpensive and abundant iron salts (FeCl3) as catalysts and molecular oxygen (air) as the terminal oxidant. This eliminates the need for expensive rare earth metals or corrosive acidic environments, significantly reducing raw material costs and waste treatment expenses.
Q: How does the use of DMSO in this reaction impact the process safety and scalability?
A: In this novel pathway, dimethyl sulfoxide (DMSO) serves a dual role as both the solvent and a carbon source (providing the methyl group for ring closure). This one-pot design simplifies the operational procedure by removing the need for pre-synthesis of unstable intermediates like imines or α,β-unsaturated ketones, thereby enhancing process safety and making large-scale commercial production more feasible.
Q: Is this method suitable for synthesizing quinolines with diverse substitution patterns?
A: Yes, the patent demonstrates high tolerance for various substituents on the aniline ring, including alkyl, alkoxy, halogen (chloro, bromo), and aryl groups. This versatility allows for the efficient production of a wide library of polysubstituted quinoline derivatives, which are critical scaffolds for developing new pharmaceutical agents and agrochemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Quinoline Supplier
The technological breakthroughs detailed in CN107628996B underscore the immense potential of iron-catalyzed oxidative cyclization in modern organic synthesis. At NINGBO INNO PHARMCHEM, we recognize the strategic value of such innovations and have integrated advanced catalytic methodologies into our CDMO capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch.
We invite forward-thinking partners to collaborate with us on leveraging this efficient synthetic route for your specific quinoline-based projects. Whether you require custom synthesis of novel analogs or reliable supply of established intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this cost-effective technology can enhance your supply chain resilience and accelerate your time-to-market.
