Advanced Synthesis of Pyridine Quinazoline Intermediates for Scalable Agrochemical Manufacturing
Advanced Synthesis of Pyridine Quinazoline Intermediates for Scalable Agrochemical Manufacturing
The global demand for high-efficiency insecticides continues to drive innovation in the synthesis of complex heterocyclic intermediates, particularly within the quinazoline class. Patent CN111533701B, published in October 2020, introduces a groundbreaking synthetic methodology for producing 3-amino-6-(perfluoropropane-2-yl)-3,4-dihydroquinazolin-2(1H)-one, a critical precursor for pyridine quinazoline insecticides. This intermediate represents a substantial portion of the final active ingredient's molecular mass and accounts for over 80% of the raw material cost, making its efficient production paramount for the agrochemical industry. The disclosed technology shifts the paradigm from expensive aromatic amine starting materials to a more economical nitrobenzene-based route, addressing long-standing challenges in yield optimization and impurity control. By leveraging a novel one-pot reduction and substitution strategy, this process not only enhances chemical efficiency but also aligns with modern green chemistry principles, offering a compelling value proposition for manufacturers seeking to optimize their supply chains.
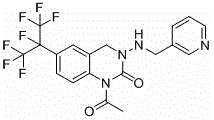
Understanding the structural complexity of the target molecule is essential for appreciating the synthetic challenges involved. The presence of the perfluoropropane-2-yl group introduces significant steric and electronic effects that complicate traditional electrophilic substitutions. As illustrated in the molecular structure, the integration of the fluorinated moiety requires precise control to prevent defluorination side reactions, which can severely impact the purity profile of the final agrochemical active. The patent highlights that the molar mass of this specific intermediate constitutes approximately 72% of the final pyridine quinazoline insecticide, underscoring its role as the dominant cost driver in the manufacturing value chain. Consequently, any improvement in the synthesis of this core scaffold directly translates to enhanced competitiveness in the global pesticide market, providing a strategic advantage for producers who can master this specific chemical architecture.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key intermediate has relied heavily on o-toluidine or 2-methylaniline derivatives as starting materials, a approach fraught with economic and technical inefficiencies. Traditional routes, such as those disclosed in WO2005123695 or JP2006036758A, typically involve free radical substitution reactions followed by amino protection and benzyl chlorination. These methods suffer from inherently low yields during the benzyl chloride formation step, leading to significant material loss and the generation of complex impurity profiles that are difficult to purge. Furthermore, the reliance on o-toluidine presents a severe cost disadvantage, as this raw material is considerably more expensive than commodity chemicals like nitrobenzene. From an environmental and safety perspective, conventional processes often necessitate the use of elemental chlorine gas or phosgene for ring closure and chlorination, posing acute toxicity risks to plant personnel and requiring extensive, costly abatement systems to manage hazardous emissions and waste streams.
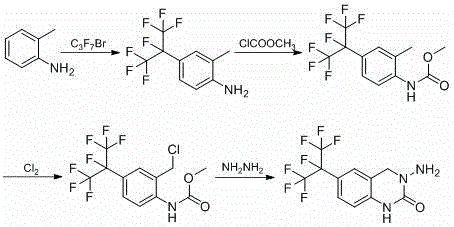
The Novel Approach
In stark contrast, the methodology detailed in CN111533701B revolutionizes the production landscape by utilizing nitrobenzene and heptafluoro bromopropane as the foundational building blocks. This innovative route employs a sophisticated one-pot technique that simultaneously achieves benzene ring nitro reduction and substitution, effectively streamlining the initial stages of synthesis. A critical breakthrough in this approach is the observation that the presence of nitrobenzene during the substitution reaction actively inhibits the formation of defluorinated impurities, a common failure mode in fluorinated chemistry that previously plagued yield rates. By replacing hazardous chlorine gas with a controlled chloromethylation reaction using paraformaldehyde and hydrochloric acid, the new process eliminates the risks associated with handling toxic gases while improving selectivity. This shift not only simplifies the operational workflow but also drastically reduces the environmental footprint, making it an ideal candidate for large-scale industrial adoption where safety and sustainability are non-negotiable priorities.
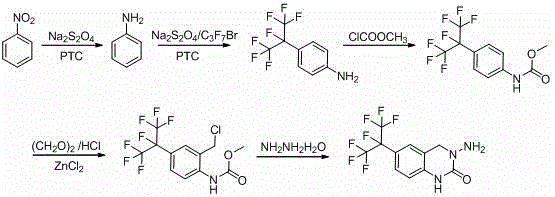
Mechanistic Insights into One-Pot Nitro Reduction and Substitution
The core of this technological advancement lies in the intricate mechanistic interplay during the initial transformation of nitrobenzene. The process utilizes sodium hydrosulfite as a reducing agent in conjunction with a phase transfer catalyst, specifically tetrabutylammonium bisulfate, to facilitate the reaction between the organic and aqueous phases. Under mild conditions ranging from 25°C to 30°C, the nitro group is reduced to an amine in situ, which immediately undergoes nucleophilic substitution with heptafluoro bromopropane. The presence of the nitro group prior to full reduction appears to exert an electron-withdrawing effect that stabilizes the aromatic ring against unwanted side reactions, thereby suppressing the elimination of fluorine atoms that typically occurs under basic or thermal stress. This dual-function reaction step is pivotal, as it consolidates two distinct chemical transformations into a single operational unit, minimizing solvent usage and intermediate isolation steps that often contribute to yield erosion in multi-step syntheses.
Following the formation of the fluorinated aniline, the synthesis proceeds through a carefully orchestrated sequence of protection and cyclization events designed to maximize purity. The amino group is protected as a methyl carbamate using methyl chloroformate, which serves to moderate the reactivity of the nitrogen atom during subsequent steps. The crucial chloromethylation is then catalyzed by zinc chloride, promoting the electrophilic attack of the chloromethyl species onto the aromatic ring with high regioselectivity. Finally, the ring closure is achieved through a nucleophilic attack by hydrazine hydrate on the chloromethyl carbamate intermediate. This cyclization step is conducted in methanol at controlled temperatures to ensure complete conversion while preventing the degradation of the sensitive perfluoroalkyl chain. The result is a robust process capable of delivering the target quinazolinone scaffold with a purity exceeding 95%, demonstrating superior control over the impurity spectrum compared to legacy methods.
How to Synthesize 3-amino-6-(perfluoropropane-2-yl)-3,4-dihydroquinazolin-2(1H)-one Efficiently
The implementation of this synthesis requires precise adherence to the reaction parameters outlined in the patent to ensure reproducibility and optimal yield. The process begins with the preparation of the reaction mixture containing nitrobenzene, sodium hydrosulfite, and the phase transfer catalyst in a biphasic solvent system of methyl tert-butyl ether and water. The addition of heptafluoro bromopropane must be controlled over a period of six hours to manage the exotherm and ensure complete conversion. Following the one-pot reduction and substitution, the organic layer is separated and directly utilized for the protection step without extensive purification, leveraging the compatibility of the solvent system to reduce waste. The subsequent chloromethylation and cyclization steps require strict temperature control and stoichiometric balance to avoid over-reaction or decomposition. For a comprehensive, step-by-step technical guide including exact reagent quantities and workup procedures, please refer to the standardized protocol below.
- Perform a one-pot nitro reduction and benzene ring substitution using nitrobenzene, sodium hydrosulfite, and heptafluoro bromopropane with a phase transfer catalyst to obtain 4-(perfluoropropane-2-yl) aniline.
- Protect the amino group by reacting the aniline derivative with methyl chloroformate and sodium carbonate to form 4-(perfluoropropane-2-yl) phenyl methyl carbamate.
- Execute a zinc chloride-catalyzed chloromethylation reaction using paraformaldehyde and hydrochloric acid to introduce the chloromethyl group safely without chlorine gas.
- Complete the synthesis via hydrazine hydrate cyclization in methanol to yield the final 3-amino-6-(perfluoropropane-2-yl)-3,4-dihydroquinazolin-2(1H)-one with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nitrobenzene-based synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. The fundamental shift from specialty aromatic amines to commodity-grade nitrobenzene fundamentally alters the cost structure of the intermediate, decoupling production economics from the volatile pricing of niche starting materials. By eliminating the need for hazardous chlorine gas and phosgene, facilities can significantly reduce their expenditure on safety infrastructure, personal protective equipment, and regulatory compliance measures associated with handling Schedule 1 chemicals. Furthermore, the simplification of the process flow, particularly the combination of reduction and substitution into a single pot, reduces the overall cycle time and labor intensity required for production. These operational efficiencies translate into a more resilient supply chain capable of responding rapidly to market demand fluctuations without the bottlenecks typical of complex, multi-step batch processes.
- Cost Reduction in Manufacturing: The replacement of expensive o-toluidine with nitrobenzene results in a drastic reduction in raw material expenditure, as nitrobenzene is a widely produced commodity chemical with a stable and low market price. Additionally, the consolidation of reaction steps minimizes solvent consumption and energy usage for heating and cooling cycles, further driving down the variable cost per kilogram of the final intermediate. The avoidance of expensive transition metal catalysts or specialized reagents in favor of inexpensive zinc chloride and sodium hydrosulfite ensures that the cost of goods sold remains competitive even at scale.
- Enhanced Supply Chain Reliability: Sourcing nitrobenzene and heptafluoro bromopropane is significantly more reliable than procuring specialized fluorinated anilines, which often have limited global suppliers and long lead times. The robustness of the one-pot reaction reduces the risk of batch failures due to intermediate instability, ensuring a consistent output of high-quality material. This reliability allows supply chain planners to maintain lower safety stock levels while still meeting delivery commitments, optimizing working capital and reducing inventory holding costs across the logistics network.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-hundred-gram batches with linear scalability to tonnage production without significant re-engineering. The elimination of toxic chlorine gas and the reduction of halogenated waste streams simplify wastewater treatment and exhaust gas scrubbing requirements, facilitating easier permitting and ongoing environmental compliance. This green chemistry profile not only mitigates regulatory risk but also aligns with the sustainability goals of major agrochemical customers who increasingly prioritize eco-friendly manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety protocols, and scalability factors. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the strategic fit of this intermediate within their product portfolio. The answers reflect the specific advantages of the nitrobenzene route over traditional methodologies, highlighting the practical implications for industrial production.
Q: What are the primary cost advantages of the nitrobenzene-based route compared to traditional o-toluidine methods?
A: The nitrobenzene-based route significantly lowers raw material costs because nitrobenzene is substantially cheaper and more readily available than o-toluidine. Additionally, the one-pot reduction and substitution step simplifies the process, reducing operational expenses and waste treatment costs associated with multi-step traditional routes.
Q: How does this new synthesis method improve safety regarding hazardous reagents?
A: This method eliminates the need for highly toxic chlorine gas (Cl2) and phosgene, which are required in conventional chlorination and ring-closure steps. Instead, it utilizes a safer chloromethylation reaction with paraformaldehyde and hydrochloric acid, drastically reducing industrial safety risks and environmental hazards.
Q: What is the expected purity and yield for the final intermediate using this patented process?
A: According to the patent data, the process achieves a total yield of 61.8% to 65.2% calculated from nitrobenzene. The final product content is consistently high, ranging between 95.0% and 97.0% as determined by liquid chromatography, ensuring suitability for downstream pharmaceutical or agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-amino-6-(perfluoropropane-2-yl)-3,4-dihydroquinazolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation insecticides depends on the secure and efficient supply of high-quality intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and risk-mitigated. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3-amino-6-(perfluoropropane-2-yl)-3,4-dihydroquinazolin-2(1H)-one meets the exacting standards required for downstream synthesis. Our facility is equipped to handle fluorinated chemistry safely and efficiently, leveraging the latest process intensification techniques to maximize yield and minimize environmental impact.
We invite global agrochemical manufacturers and R&D organizations to collaborate with us to leverage this advanced synthetic technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and pilot batch samples. Let us help you secure a competitive edge in the agrochemical market through superior chemistry and reliable supply chain execution.
