Advanced Isosorbide Synthesis: Scalable Bio-Based Routes for Global Supply Chains
Advanced Isosorbide Synthesis: Scalable Bio-Based Routes for Global Supply Chains
The global demand for sustainable, bio-based platform chemicals is driving a paradigm shift in how fine chemical manufacturers approach synthesis, particularly for versatile molecules like isosorbide. Patent CN109134485B introduces a transformative methodology that leverages biomass-derived 1,4-sorbitan as a primary feedstock, utilizing a novel solid acid-catalyzed ketalization and intramolecular etherification sequence to achieve yields as high as 97%. This technological breakthrough addresses critical bottlenecks in traditional dehydration processes, offering a pathway that is not only chemically efficient but also operationally robust for large-scale manufacturing. By shifting away from corrosive liquid acids and extreme vacuum conditions, this process aligns perfectly with the modern imperatives of green chemistry and supply chain resilience, providing a reliable foundation for producing high-purity pharmaceutical intermediates and polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of isosorbide has relied heavily on the two-step intramolecular dehydration of sorbitol, a process fraught with significant technical and economic challenges that hinder optimal production efficiency. Conventional methods typically require harsh reaction environments, such as high vacuum degrees or elevated temperatures exceeding 200°C, to drive the equilibrium towards dehydration, which places immense stress on reactor materials and increases energy consumption drastically. Furthermore, the use of strong inorganic liquid acids like concentrated sulfuric acid creates severe corrosion issues, necessitating expensive specialized equipment and generating substantial quantities of hazardous waste acid that require complex neutralization and disposal protocols. The presence of multiple active hydroxyl groups in sorbitol and its intermediates often leads to non-selective side reactions, including polymerization and the formation of dark-colored humins, which severely compromise product purity and complicate downstream purification efforts.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a strategic one-pot synthesis where 1,4-sorbitan reacts with aliphatic ketones in the presence of reusable solid acid catalysts, fundamentally altering the reaction landscape to favor selectivity and ease of operation. This approach effectively mitigates the inhibitory effect of water by employing ketones that act both as reactants and reaction media, thereby stabilizing the intermediate species and preventing the catalyst deactivation commonly seen in aqueous systems. The use of heterogeneous catalysts such as strong acid cation exchange resins or hydrogen-type zeolites allows for simple separation via filtration or centrifugation, eliminating the need for energy-intensive distillation steps to remove liquid acids from the product stream. Consequently, this novel route not only simplifies the operational workflow but also significantly enhances the overall atom economy and environmental profile of the manufacturing process, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Solid Acid-Catalyzed Ketalization and Etherification
The core innovation of this synthesis lies in the dual-function mechanism where aliphatic ketones first protect the hydroxyl groups of 1,4-sorbitan through ketalization, forming a stable five-membered ring intermediate that shields the molecule from uncontrolled polymerization. This protective step is crucial because it lowers the activation energy required for the subsequent intramolecular etherification, allowing the reaction to proceed efficiently at moderate temperatures between 100°C and 180°C rather than the extreme conditions required for direct dehydration. The solid acid catalyst provides the necessary proton source to activate the carbonyl group of the ketone and the hydroxyl groups of the substrate, facilitating the nucleophilic attack that forms the ketal linkage without introducing soluble metal contaminants that could compromise the purity of the final API intermediate. This mechanistic pathway ensures that the chiral centers of the biomass-derived feedstock are preserved while systematically removing water equivalents through the formation of the ketal, thus driving the equilibrium forward without the need for continuous vacuum stripping.
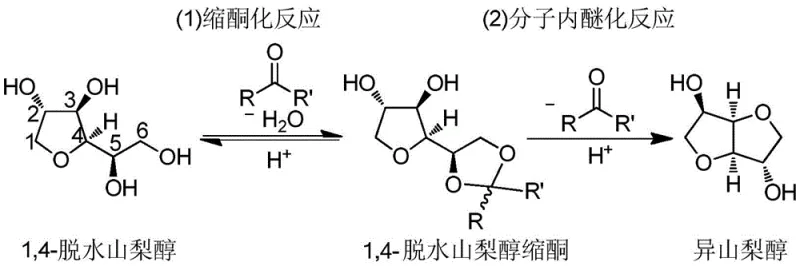
Following the initial ketal formation, the reaction system seamlessly transitions into the intramolecular etherification phase under the same catalytic conditions, where the protected intermediate undergoes cyclization to form the rigid bicyclic structure characteristic of isosorbide. The solid acid sites continue to promote the elimination of the ketal protecting group in situ, releasing the ketone back into the medium for potential recycling while yielding the target diether product with high stereochemical integrity. This tandem reaction sequence minimizes the residence time of reactive intermediates in the bulk solution, thereby reducing the probability of intermolecular condensation reactions that lead to oligomeric byproducts. For R&D directors focused on impurity profiles, this mechanism offers a distinct advantage by inherently suppressing the formation of colored bodies and polymeric tars, resulting in a crude product that requires less aggressive purification steps to meet stringent pharmacopeial standards.
How to Synthesize Isosorbide Efficiently
The practical implementation of this synthesis route involves a straightforward one-pot procedure where biomass-derived 1,4-sorbitan is mixed with a selected aliphatic ketone reagent, such as 2-pentanone or methyl isobutyl ketone, alongside a measured quantity of solid acid catalyst. The reaction mixture is heated to a controlled temperature range, typically optimized between 150°C and 170°C, and maintained under stirring for a duration sufficient to ensure complete conversion of the starting material, usually within 60 to 180 minutes depending on the specific catalyst activity. Upon completion, the heterogeneous catalyst is removed through simple physical separation methods, and the excess ketone solvent is recovered via distillation, leaving behind the high-purity isosorbide product ready for final crystallization or drying. The detailed standardized synthesis steps, including specific molar ratios and catalyst loading optimizations, are outlined in the guide below.
- Mix biomass-derived 1,4-sorbitan with an aliphatic ketone reagent (e.g., 2-pentanone) and a solid acid catalyst like Amberlyst-15 or H-Beta zeolite in a reactor.
- Heat the mixture to a temperature between 100°C and 180°C under magnetic stirring to facilitate ketal formation and subsequent etherification.
- After reaction completion (30-180 mins), filter to recover the solid catalyst and remove the solvent under reduced pressure to isolate high-purity isosorbide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this solid acid-catalyzed process presents a compelling value proposition centered on operational simplicity, raw material flexibility, and significant reductions in total cost of ownership. By eliminating the reliance on corrosive liquid mineral acids, manufacturers can utilize standard stainless steel reactors rather than exotic alloys, leading to substantial capital expenditure savings and reduced maintenance downtime associated with equipment corrosion. The ability to recycle both the solid catalyst and the aliphatic ketone solvent creates a closed-loop system that minimizes raw material consumption and waste generation, directly translating to lower variable costs per kilogram of produced isosorbide. Furthermore, the mild reaction conditions reduce energy intensity compared to high-temperature vacuum processes, contributing to a smaller carbon footprint and aligning with the sustainability goals increasingly demanded by downstream customers in the polymer and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The transition to heterogeneous catalysis removes the complex and costly neutralization and washing steps typically required to remove liquid acid residues, streamlining the downstream processing workflow. This simplification reduces the consumption of auxiliary chemicals like bases and water, while also decreasing the volume of wastewater that requires treatment, leading to significant operational expenditure efficiencies. Additionally, the high selectivity of the reaction minimizes the loss of valuable biomass feedstock to tar and polymer byproducts, maximizing the yield of saleable product from every ton of 1,4-sorbitan input.
- Enhanced Supply Chain Reliability: Utilizing widely available aliphatic ketones and robust solid acid catalysts mitigates the risk of supply disruptions often associated with specialized or hazardous reagents. The stability of the solid catalysts allows for easier storage and handling compared to corrosive liquids, reducing safety risks and regulatory burdens in the warehouse and during transportation. This robustness ensures consistent production schedules and reliable delivery timelines, which is critical for maintaining the continuity of supply for key customers in the medical device and biodegradable polymer industries.
- Scalability and Environmental Compliance: The one-pot nature of the reaction simplifies scale-up from pilot to commercial production, as there are no intermediate isolation steps that could introduce variability or yield losses at larger volumes. The process inherently generates less hazardous waste and avoids the emission of acidic fumes, making it easier to comply with increasingly stringent environmental regulations regarding air and water quality. This environmental compatibility facilitates faster permitting for new production lines and enhances the corporate social responsibility profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced isosorbide synthesis technology, derived directly from the experimental data and beneficial effects described in the patent literature. These insights are intended to clarify the operational parameters and strategic advantages for stakeholders evaluating this technology for integration into their existing manufacturing portfolios. Understanding these nuances is essential for making informed decisions about process adoption and supply chain optimization.
Q: Why is the ketalization strategy superior to direct dehydration for isosorbide production?
A: Direct dehydration of sorbitol often suffers from polymerization and humin formation due to harsh conditions. The ketalization route protects active hydroxyl groups, suppressing side reactions and enabling milder conditions with solid acid catalysts.
Q: Can the solid acid catalysts be reused in this process?
A: Yes, the patent highlights that solid acid catalysts such as cation exchange resins and zeolites can be separated via simple filtration or centrifugation and recycled, significantly reducing waste and operational costs.
Q: What represents the rate-determining step in traditional isosorbide synthesis?
A: The conversion of 1,4-sorbitan to isosorbide is the rate-determining step. Traditional methods struggle here due to water inhibition and catalyst deactivation, which this new solvent-mediated approach effectively overcomes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isosorbide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this solid acid-catalyzed route for producing high-purity isosorbide and are fully equipped to support its commercialization at any scale. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs that utilize advanced analytical techniques to verify the absence of residual catalysts and organic impurities, guaranteeing a product that meets the exacting standards of the global pharmaceutical and specialty polymer markets.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific application needs, whether for bio-based plastics, renewable solvents, or active pharmaceutical ingredients. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements, and ask for specific COA data and route feasibility assessments to see how we can optimize your supply chain today.
