Advanced Nickel-Catalyzed Thioether Synthesis for Scalable Pharmaceutical Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for carbon-sulfur bond construction, a critical structural motif found in numerous bioactive molecules. Patent CN114957058A discloses a groundbreaking synthetic method for thioether compounds that addresses long-standing challenges in efficiency and operational safety. This technology leverages a nickel-catalyzed cross-coupling strategy between iodobenzene derivatives and electrophilic sulfur reagents, specifically N-(phenylthio)phthalimide or 1-(p-toluenethio)pyrroline-2,5-dione. Unlike traditional methods requiring harsh conditions, this process operates under remarkably mild temperatures ranging from 20°C to 60°C, utilizing zinc powder as a reductant. The significance of this innovation is particularly evident in the synthesis of complex pharmaceutical intermediates, such as those required for the antidepressant drug Vortioxetine, where functional group tolerance is paramount.
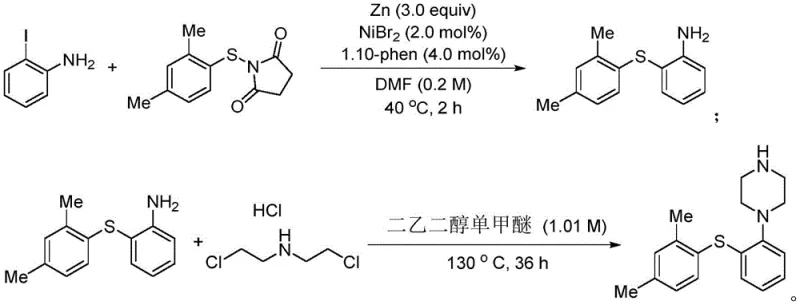
For procurement managers and supply chain heads, the implication is clear: this technology offers a pathway to cost reduction in pharmaceutical intermediate manufacturing by simplifying process requirements and enhancing safety profiles. The ability to synthesize high-purity thioethers without the need for malodorous thiols or strong bases represents a significant leap forward in green chemistry practices. As a reliable pharmaceutical intermediate supplier, understanding these underlying technological shifts is crucial for maintaining competitive advantage in the global market. The following analysis delves into the technical specifics, comparing this novel approach against conventional limitations to highlight its commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-S bonds has relied heavily on the nucleophilic substitution of aryl halides with thiophenols or the use of organometallic reagents. However, these conventional pathways are fraught with significant operational and chemical drawbacks that hinder large-scale production. Thiophenols, while effective nucleophiles, are notoriously toxic and possess an extremely unpleasant odor, creating severe safety and environmental hazards in manufacturing facilities. Furthermore, the sulfur atoms in thiophenols have a high affinity for transition metals, often leading to catalyst poisoning which necessitates high catalyst loadings to drive the reaction to completion. To overcome this deactivation, traditional methods frequently require elevated temperatures and strong bases, which severely limit the tolerance of sensitive functional groups such as esters, aldehydes, or ketones. Additionally, alternative strategies involving aryllithium reagents, as reported by groups like Hosoya, suffer from poor stability and苛刻 preparation conditions, making them unsuitable for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
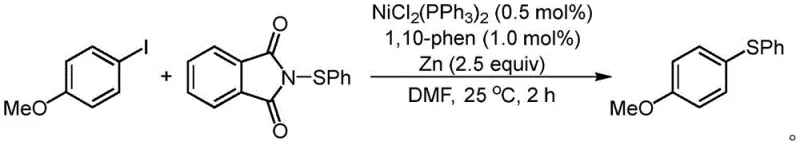
In stark contrast, the methodology described in CN114957058A introduces a paradigm shift by utilizing electrophilic sulfur reagents instead of nucleophilic thiols. This approach effectively bypasses the issues of catalyst poisoning and odor. The reaction employs a nickel catalyst system, specifically NiCl2(PPh3)2 or NiBr2, paired with 1,10-phenanthroline as a ligand, operating at ambient to moderately warm temperatures (25°C to 40°C in many examples). This mildness allows for the preservation of delicate functional groups that would otherwise decompose under basic or high-thermal stress. The use of zinc powder as a stoichiometric reductant facilitates the catalytic cycle without generating hazardous waste streams associated with other reductants. As illustrated in the representative reaction scheme below, the coupling of p-iodoanisole with N-(phenylthio)phthalimide proceeds efficiently to yield the corresponding thioether in high yields, demonstrating the practicality of this system for diverse substrate scopes.
Mechanistic Insights into Nickel-Catalyzed C-S Bond Formation
The success of this synthetic route lies in the intricate interplay between the nickel catalyst and the electrophilic sulfur source. Mechanistically, the reaction is believed to proceed through a Ni(0)/Ni(II) catalytic cycle initiated by the reduction of the nickel precatalyst by zinc powder. The active Ni(0) species undergoes oxidative addition with the aryl iodide derivative to form an aryl-nickel(II) intermediate. Subsequently, the electrophilic sulfur reagent, such as N-(phenylthio)phthalimide shown in the structure below, interacts with this intermediate. The unique electronic nature of the N-S bond in these reagents allows for a transmetallation-like step or radical pathway that transfers the sulfur moiety to the nickel center without the strong coordination that typically deactivates the catalyst in thiol-based reactions.
Following the formation of the aryl-nickel-sulfur species, reductive elimination occurs to release the desired thioether product and regenerate the Ni(0) catalyst. This mechanism is highly advantageous for impurity control because it avoids the formation of disulfide byproducts common in oxidative thiol couplings. The mild conditions also minimize side reactions such as homocoupling of the aryl iodide or decomposition of the sulfur reagent. For R&D directors, this mechanistic clarity ensures that the process is robust and predictable, allowing for the rational design of analogues. The broad substrate scope demonstrated in the patent, including substrates with trifluoromethyl, ester, aldehyde, and halide substituents, confirms that the catalytic cycle is resilient against electronic variations, making it a versatile tool for high-purity pharmaceutical intermediate synthesis.
How to Synthesize Thioether Compounds Efficiently
Implementing this nickel-catalyzed protocol requires careful attention to reaction conditions to maximize yield and purity. The process is designed to be operationally simple, utilizing standard laboratory equipment such as sealed tubes and inert atmosphere techniques. The key to success lies in maintaining anhydrous conditions and the correct stoichiometric ratio of zinc powder to ensure complete reduction of the catalyst and driving force for the reaction. Detailed standard operating procedures regarding reagent addition, temperature control, and workup protocols are essential for reproducibility. For those looking to adopt this technology, the following guide outlines the standardized synthesis steps derived directly from the patent examples.
- Under dry nitrogen protection, combine iodobenzene derivative, N-(phenylthio)phthalimide, zinc powder, NiCl2(PPh3)2, and 1,10-phenanthroline in a sealed tube.
- Add dry DMF solvent and stir the mixture at 20-30°C for 1 to 6 hours to allow the catalytic cycle to proceed.
- Dilute with saturated NaCl and ethyl acetate, filter through diatomaceous earth, separate layers, extract, dry over sodium sulfate, and purify via silica gel column.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers tangible benefits that extend beyond mere chemical efficiency. For procurement managers, the elimination of thiophenols translates directly into reduced safety compliance costs and simplified waste management protocols. The absence of strong bases and high-temperature requirements means that standard stainless steel reactors can be used without the need for specialized linings or extensive cooling systems, thereby lowering capital expenditure for process scaling. Furthermore, the low catalyst loading (as low as 0.5 mol%) significantly reduces the cost of goods sold (COGS) related to precious or transition metals, while also simplifying the downstream purification process to meet stringent heavy metal limits required for API production.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the simplification of the reaction workflow. By avoiding the need for cryogenic conditions or expensive organometallic reagents, the overall energy consumption is drastically lowered. The use of inexpensive zinc powder as a reductant and commercially available nickel salts further drives down raw material costs. Additionally, the high yields reported across various substrates mean less raw material is wasted, improving the overall atom economy of the process. This efficiency allows for substantial cost savings in the production of key intermediates, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of hazardous or specialized reagents. This method relies on stable, shelf-stable starting materials like iodobenzene derivatives and solid sulfur reagents, which are readily sourced from multiple suppliers globally. The robustness of the reaction conditions ensures that batch-to-batch variability is minimized, reducing the risk of production delays due to failed runs. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug development timelines are met without interruption caused by raw material shortages or quality issues.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this methodology mitigates those concerns. The mild temperature range (20-60°C) makes heat dissipation manageable even in large-scale reactors, preventing thermal runaways. Moreover, the avoidance of volatile and toxic thiols improves the working environment for operators and reduces the burden on scrubber systems. The waste stream, primarily consisting of zinc salts and organic solvents, is easier to treat compared to waste containing heavy sulfur residues or strong alkalis. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the scope and limitations of this nickel-catalyzed method. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development decisions. Understanding these nuances helps in assessing the feasibility of adapting this route for new molecular entities.
Q: What are the advantages of this method over traditional thiophenol coupling?
A: This method avoids the use of toxic and odorous thiophenols which often poison transition metal catalysts. It operates under mild conditions (20-60°C) without strong bases, ensuring better functional group tolerance.
Q: What is the typical catalyst loading required for this reaction?
A: The process utilizes very low catalyst loadings, typically ranging from 0.5 mol% to 2.0 mol% of nickel catalyst, which significantly reduces heavy metal residue concerns and cost.
Q: Can this method be applied to complex drug intermediates?
A: Yes, the patent explicitly demonstrates the application of this methodology in the synthesis of Vortioxetine drug molecules, proving its viability for complex pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioether Compounds Supplier
The technological advancements detailed in CN114957058A represent a significant opportunity for optimizing the supply chain of sulfur-containing pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such innovative academic and patent literature into robust, commercial-grade manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this mild nickel-catalyzed method can be realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of thioether intermediate meets the exacting standards required for drug substance synthesis.
We invite potential partners to engage with our technical team to explore how this methodology can be applied to your specific portfolio. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this safer, more efficient route. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition from development to commercial supply.
