Strategic Manufacturing of Doxylamine Succinate via Optimized Grignard Route
The pharmaceutical landscape for antihistamines continues to evolve, driven by the demand for safer, more efficient manufacturing processes for key active ingredients like Doxylamine Succinate. A pivotal advancement in this domain is detailed in patent CN103524403A, which outlines a robust preparation method that fundamentally re-engineers the synthetic pathway for this critical sedative and antihistaminic agent. Unlike traditional methods that rely on harsh Lewis acid catalysts and problematic distillation steps, this novel approach leverages the superior reactivity of iodobenzene in a Grignard reaction to generate the core alcohol intermediate. This strategic shift not only streamlines the reaction conditions but also addresses long-standing purity challenges associated with the low melting point of the intermediate species. For global procurement teams and R&D directors, understanding this technological pivot is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials consistently.
Furthermore, the integration of low-temperature recrystallization techniques represents a significant leap forward in process control, ensuring that the final API meets stringent regulatory standards without the yield losses typical of thermal separation methods. By optimizing the stoichiometry of the Grignard reagent and refining the workup procedures, the patented process achieves a balance between high reaction efficiency and operational simplicity. This makes it particularly attractive for cost reduction in API manufacturing, where every step of solvent recovery and waste treatment impacts the bottom line. As we delve deeper into the technical specifics, it becomes clear that this methodology offers a scalable solution for the commercial scale-up of complex pharmaceutical intermediates, positioning manufacturers to meet the rigorous demands of the international market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
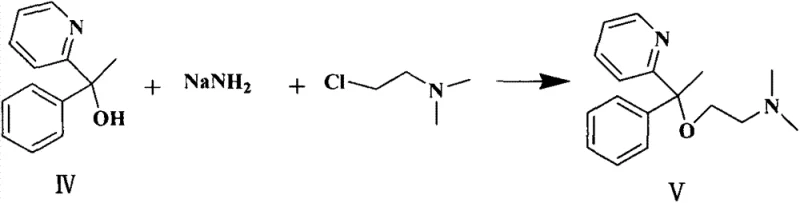
Historically, the synthesis of Doxylamine Succinate has been plagued by inefficiencies inherent in older synthetic routes, particularly those disclosed in prior art such as CN201210587388.0 and CN102108059A. These conventional methods typically rely on the use of bromobenzene to generate the Grignard reagent, which often necessitates the addition of aggressive Lewis acid catalysts like boron trifluoride or tin tetrachloride to drive the reaction to completion. The reliance on these corrosive and toxic catalysts introduces significant safety hazards and environmental burdens, requiring specialized equipment and complex neutralization protocols that inflate operational costs. Moreover, the purification of the key intermediate, 2-pyridyl phenyl methyl alcohol, has traditionally been attempted through vacuum distillation. However, this physical property presents a major bottleneck; the intermediate has a melting point of merely 33~34.5 ℃, making it prone to solidification in condenser lines and leading to substantial product loss and inconsistent purity profiles during thermal processing.
The Novel Approach
In stark contrast, the novel approach described in the patent data circumvents these pitfalls by substituting bromobenzene with iodobenzene, a reagent with significantly higher reactivity that facilitates Grignard formation without the need for auxiliary Lewis acids. This elimination of catalysts simplifies the reaction matrix, reducing the impurity load from the outset and minimizing the generation of hazardous waste streams. Instead of relying on the precarious process of distillation for the low-melting intermediate, the new method employs a sophisticated low-temperature recrystallization technique using specific solvent systems like n-hexane and ethyl acetate at temperatures ≤-15 ℃. This phase-change purification strategy effectively isolates the desired alcohol in high purity (up to 99.5%) while avoiding the thermal degradation and mechanical losses associated with distillation. Consequently, this approach not only enhances the overall yield but also establishes a more robust foundation for the subsequent etherification step, ensuring a smoother transition to the final Doxylamine base.
Mechanistic Insights into Iodobenzene-Mediated Grignard Addition
The core of this synthetic innovation lies in the mechanistic advantages conferred by the use of iodobenzene in the initial Grignard formation. Iodobenzene possesses a weaker carbon-iodine bond compared to the carbon-bromine bond in bromobenzene, which lowers the activation energy required for the insertion of magnesium. This heightened reactivity ensures a more rapid and complete formation of the phenylmagnesium iodide species, which then acts as a potent nucleophile attacking the carbonyl carbon of 2-acetylpyridine. The absence of Lewis acid catalysts is particularly noteworthy; in traditional pathways, these acids are required to activate the carbonyl group, but they often lead to side reactions and difficult-to-remove metal residues. By relying solely on the intrinsic reactivity of the iodine-based Grignard reagent, the process achieves a cleaner reaction profile, significantly reducing the formation of biphenyl byproducts and unreacted ketone residues that typically complicate downstream purification.
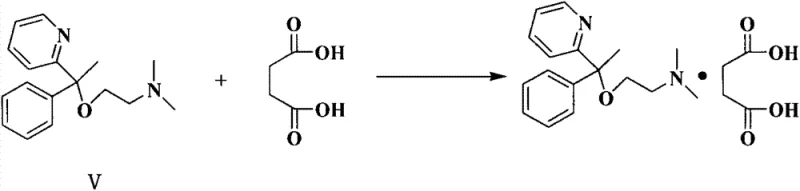
Following the formation of the alcohol intermediate, the process transitions to an etherification reaction mediated by sodium amide (NaNH2) in a xylene solvent system. In this step, the hydroxyl group of the 2-pyridyl phenyl methyl alcohol is deprotonated by the strong base to form an alkoxide, which subsequently undergoes a nucleophilic substitution with 2-dimethylamino chloroethane. The choice of xylene as a high-boiling solvent allows the reaction to proceed at reflux temperatures (around 140 ℃), providing the necessary thermal energy to overcome the activation barrier for the substitution without decomposing the sensitive pyridine ring. The mechanistic precision here is critical for controlling the impurity profile, as excessive basicity or temperature fluctuations could lead to elimination side products. The resulting Doxylamine base is then isolated through a pH-controlled extraction sequence, ensuring that basic impurities are separated before the final salt formation.
How to Synthesize Doxylamine Succinate Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the benefits of the patented route. The process begins with the careful activation of magnesium chips, often facilitated by a catalytic amount of iodine, to ensure the smooth initiation of the Grignard reaction with iodobenzene in an ether or THF solvent. Once the Grignard reagent is established, 2-acetylpyridine is added under controlled conditions to manage the exotherm, followed by a quenching step with aqueous ammonium chloride to liberate the alcohol intermediate. The critical purification stage involves dissolving the crude oil in a specific solvent mixture and cooling it to sub-zero temperatures to induce crystallization, a step that is far superior to distillation for this specific molecule. Finally, the purified alcohol is reacted with sodium amide and the chloroamine derivative in xylene, followed by a multi-stage extraction workup to isolate the free base, which is then converted to the succinate salt.
- Generate Grignard reagent from iodobenzene and magnesium, then react with 2-acetylpyridine to form 2-pyridyl phenyl methyl alcohol.
- Purify the alcohol intermediate via low-temperature recrystallization instead of distillation to ensure high purity.
- React the purified alcohol with sodium amide and 2-dimethylamino chloroethane in xylene, followed by salt formation with succinic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages that extend beyond mere chemical yield. The primary value driver is the significant simplification of the manufacturing infrastructure required. By eliminating the need for corrosive Lewis acid catalysts, manufacturers can utilize standard glass-lined or stainless steel reactors without the need for exotic alloys or extensive corrosion monitoring, directly reducing capital expenditure (CapEx). Furthermore, the removal of these hazardous reagents simplifies the waste treatment protocol, as there is no longer a need to neutralize and dispose of heavy metal salts like tin or boron complexes. This reduction in hazardous waste volume aligns perfectly with increasingly stringent environmental regulations, mitigating the risk of production shutdowns due to compliance issues and ensuring a more stable supply continuity for downstream customers.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive and toxic catalysts and the improvement in overall material throughput. Traditional methods often suffer from yield erosion during the distillation of the low-melting intermediate, whereas the recrystallization method recovers a much higher percentage of the theoretical yield. Additionally, the simplified workup procedure reduces the consumption of solvents and reagents required for neutralization and cleaning, leading to substantial cost savings in raw material procurement. The ability to produce high-purity intermediates without complex chromatographic purification or multiple distillation passes further lowers the operational expenditure (OpEx), making the final Doxylamine Succinate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the availability of raw materials. Iodobenzene and 2-acetylpyridine are commodity chemicals with stable global supply lines, reducing the risk of bottlenecks associated with specialty catalysts. Moreover, the process tolerance is improved; the recrystallization step is less sensitive to minor fluctuations in reaction conversion compared to distillation, meaning that batch-to-batch variability is minimized. This consistency is crucial for maintaining long-term contracts with pharmaceutical clients who require strict adherence to quality specifications. The reduced complexity of the process also shortens the production cycle time, allowing for faster turnaround on orders and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is inherently safer and more manageable due to the absence of highly corrosive reagents and the avoidance of vacuum distillation for waxy solids. Vacuum distillation of low-melting compounds often presents engineering challenges at large scales, such as fouling of columns and pumps, which can halt production. The switch to crystallization avoids these mechanical failures entirely. From an environmental perspective, the process generates less hazardous waste, simplifying the permitting process for new manufacturing facilities. This 'green chemistry' aspect is increasingly valued by multinational corporations seeking to reduce their carbon footprint and ensure that their supply chain partners adhere to sustainable manufacturing practices, thereby future-proofing the supply agreement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Doxylamine Succinate using this optimized pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on why this method represents a superior choice for modern pharmaceutical manufacturing. Understanding these nuances helps stakeholders make informed decisions regarding vendor selection and process validation.
Q: Why is iodobenzene preferred over bromobenzene in this Grignard reaction?
A: Iodobenzene exhibits higher reactivity than bromobenzene, allowing the Grignard reagent to form more readily without the need for additional Lewis acid catalysts like boron trifluoride or tin tetrachloride, thereby simplifying the process and reducing toxicity.
Q: How does the new method improve the purity of the intermediate?
A: The method replaces difficult vacuum distillation with low-temperature recrystallization (≤-15°C). Since the intermediate has a low melting point (33–34.5°C), distillation often leads to product loss and decomposition, whereas recrystallization effectively removes impurities while maintaining structural integrity.
Q: What are the scalability benefits of eliminating Lewis acid catalysts?
A: Removing corrosive Lewis acids eliminates the need for specialized corrosion-resistant reactor linings and complex neutralization waste streams, significantly lowering capital expenditure (CapEx) and operational complexity for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doxylamine Succinate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensively analyzed the pathway described in CN103524403A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that the successful implementation of this Grignard-based route requires precise control over moisture levels during the organometallic step and rigorous temperature management during the recrystallization phase. Our state-of-the-art facilities are equipped with the necessary cryogenic capabilities and corrosion-resistant infrastructure to execute this chemistry safely and efficiently, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards required by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage this advanced synthetic route for your supply chain needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that details exactly how switching to this catalyst-free method can optimize your budget without compromising quality. We encourage potential partners to contact us directly to request specific COA data from our recent pilot runs and to discuss route feasibility assessments tailored to your specific volume requirements. Let us demonstrate how our commitment to process innovation can secure your supply of high-purity Doxylamine Succinate for the long term.