Advanced Grignard-Based Synthesis of Doxylamine Succinate for Commercial Pharmaceutical Manufacturing
Advanced Grignard-Based Synthesis of Doxylamine Succinate for Commercial Pharmaceutical Manufacturing
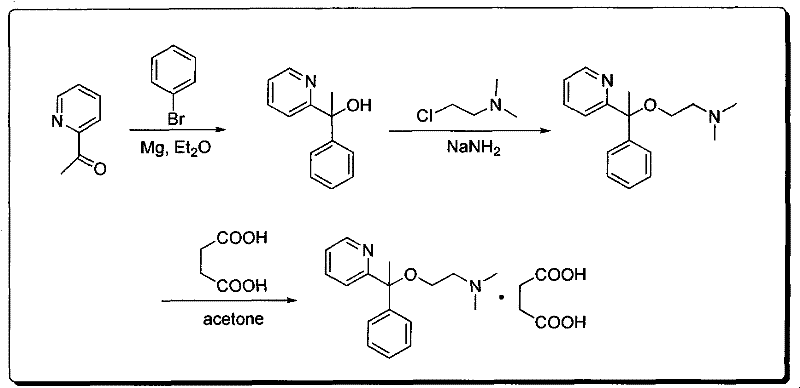
The pharmaceutical landscape for over-the-counter (OTC) antihistamines continues to demand robust, scalable, and cost-effective synthetic routes for key active ingredients. A pivotal development in this domain is detailed in patent CN102108059B, which discloses a novel and highly efficient method for the synthesis of Doxylamine Succinate (CAS 562-10-7). This ethanolamine-class antihistamine is critical for treating allergic conditions and insomnia, necessitating a supply chain capable of delivering high-purity material consistently. The patented methodology leverages a strategic Grignard reaction to construct the core carbon skeleton, followed by a precise etherification and salt formation sequence. For R&D directors and procurement specialists, understanding the nuances of this specific pathway is essential for evaluating supplier capabilities and ensuring long-term supply continuity for this high-volume OTC medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex ethanolamine derivatives like Doxylamine has posed significant challenges regarding regioselectivity and impurity profiles. Conventional approaches often struggle with the formation of the diphenyl-methyl backbone, frequently resulting in complex mixtures of isomers or over-alkylated by-products that are difficult to separate. In many traditional pathways, the introduction of the dimethylaminoethyl side chain can lead to quaternary ammonium salt formation or elimination reactions, drastically reducing overall yield and complicating downstream purification. Furthermore, older methods may rely on harsh conditions or expensive catalysts that are not economically viable for large-scale generic drug production. The lack of a directional reaction mechanism in these conventional routes often necessitates extensive chromatographic purification, which is a bottleneck for commercial scalability and significantly drives up the cost of goods sold (COGS) for the final API.
The Novel Approach
The methodology outlined in patent CN102108059B presents a breakthrough by utilizing a directional Grignard addition to establish the core structure with high fidelity. By reacting 2-acetylpyridine with a phenylmagnesium bromide Grignard reagent, the process ensures the precise formation of the tertiary alcohol intermediate, 2-pyridylphenylmethylcarbinol, with minimal side reactions. This is followed by a controlled etherification step using sodium amide and 2-dimethylaminochloroethane, which effectively installs the amine functionality without the polyalkylation issues common in other nucleophilic substitutions. The patent explicitly highlights that this synthetic route is directional and generates no significant side products, a claim that translates directly to simplified work-up procedures and higher throughput. This streamlined approach not only enhances the chemical purity of the intermediate but also facilitates a more predictable and manageable manufacturing process for industrial partners.
Mechanistic Insights into Grignard-Mediated Carbon Skeleton Construction
The cornerstone of this synthesis is the initial Grignard reaction, where the nucleophilic phenyl group attacks the electrophilic carbonyl carbon of 2-acetylpyridine. This step is critical as it forms the quaternary carbon center bearing both the pyridyl and phenyl rings, which defines the steric and electronic properties of the final drug molecule. The reaction is typically conducted in anhydrous ether or tetrahydrofuran (THF) to stabilize the organomagnesium species, requiring strict moisture control to prevent protonation of the Grignard reagent. Following the addition, the reaction mixture is quenched with an ammonium chloride ice-water solution, a standard procedure that safely decomposes the magnesium alkoxide complex to release the free alcohol. The patent specifies a reaction time of approximately 20 hours, allowing for complete conversion and minimizing the presence of unreacted ketone, which could otherwise carry through as a critical impurity in subsequent steps.
Subsequent transformation involves the deprotonation of the tertiary alcohol using sodium amide (NaNH2) in xylene to generate the corresponding alkoxide nucleophile. This alkoxide then undergoes an SN2-type substitution with 2-dimethylaminochloroethane to form the ether linkage. The choice of xylene as a solvent allows for high-temperature reflux (around 150°C), providing the necessary activation energy for this sterically hindered substitution. A key mechanistic advantage here is the recovery of unreacted 2-pyridylphenylmethylcarbinol via silica gel chromatography, as described in the patent. This ability to recycle the valuable intermediate back into the process loop is a significant factor in improving the overall atom economy and reducing waste, demonstrating a sophisticated understanding of process chemistry that goes beyond simple laboratory synthesis to true manufacturing optimization.
How to Synthesize Doxylamine Succinate Efficiently
Executing this synthesis requires precise control over reaction parameters, particularly during the exothermic Grignard formation and the high-temperature etherification stages. The process begins with the preparation of the Grignard reagent from bromobenzene and magnesium, followed by the slow addition of 2-acetylpyridine to manage heat evolution. After isolation of the carbinol intermediate via vacuum distillation, the second stage involves refluxing with sodium amide and the chloro-amine derivative in xylene for an extended period to ensure complete conversion. The final step is a straightforward acid-base salt formation between the purified Doxylamine base and succinic acid in acetone, yielding the stable crystalline succinate salt. For detailed operational parameters and safety protocols, please refer to the standardized guide below.
- Preparation of 2-Pyridylphenylmethylcarbinol via Grignard reaction between 2-acetylpyridine and phenylmagnesium bromide in ether or THF.
- Etherification of the carbinol intermediate using sodium amide and 2-dimethylaminochloroethane in xylene under reflux conditions.
- Salt formation by reacting purified Doxylamine base with succinic acid in acetone, followed by crystallization and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthetic route offers substantial strategic benefits, primarily driven by the availability of starting materials and the robustness of the unit operations. The primary raw materials, 2-acetylpyridine and bromobenzene, are commodity chemicals produced on a massive global scale, ensuring a stable and competitive pricing structure that shields manufacturers from volatile raw material markets. Unlike routes relying on exotic catalysts or bio-engineered precursors, this chemistry utilizes standard organic synthesis infrastructure found in most multipurpose chemical plants, reducing the barrier to entry for contract manufacturing organizations (CMOs) and enhancing supply chain resilience against disruptions.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the directional nature of the reaction, which minimizes the formation of difficult-to-remove impurities. By avoiding complex purification trains or expensive chiral resolutions, the overall processing time and solvent consumption are drastically reduced. Furthermore, the patent describes a specific mechanism for recovering unreacted intermediates during the chromatography step, allowing for their recycling back into the reactor. This closed-loop material flow significantly lowers the effective cost per kilogram of the final API, providing a distinct margin advantage for suppliers who implement this technology compared to those using less efficient linear syntheses.
- Enhanced Supply Chain Reliability: The reliance on common solvents such as diethyl ether, xylene, and acetone further bolsters supply chain security. These solvents are readily available from multiple global vendors, eliminating single-source dependencies that often plague specialty chemical supply chains. Additionally, the reaction conditions, while requiring anhydrous environments, do not demand extreme pressures or cryogenic temperatures that would necessitate specialized, high-capital equipment. This operational simplicity means that production can be easily scaled or shifted between different manufacturing sites without significant requalification efforts, ensuring consistent delivery schedules for downstream pharmaceutical formulators even during periods of high market demand.
- Scalability and Environmental Compliance: Scalability is inherently supported by the batch-wise nature of the Grignard and reflux steps, which are well-understood processes in the fine chemical industry. The ability to control exotherms through dropwise addition and the use of standard distillation for purification makes the transition from pilot plant to commercial tonnage straightforward. From an environmental standpoint, the process avoids the use of heavy metal catalysts, thereby simplifying waste stream treatment and reducing the burden of residual metal testing in the final drug product. The recycling of solvents and intermediates, as mandated by the economic logic of the process, aligns with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality of Doxylamine Succinate synthesized via this route. These insights are derived directly from the technical specifications and experimental data provided in the underlying patent documentation, offering transparency for potential partners evaluating this technology for their supply chain.
Q: What is the primary advantage of the Grignard route for Doxylamine Succinate?
A: The patented Grignard route offers a directional reaction pathway that minimizes side products during the carbon-carbon bond formation step, ensuring a cleaner intermediate profile compared to non-directional alkylation methods.
Q: How is the purity of the final Doxylamine Succinate ensured?
A: Purity is maintained through rigorous purification steps including vacuum distillation of the carbinol intermediate, silica gel chromatography to recover unreacted starting materials, and final recrystallization of the succinate salt from acetone.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes standard industrial solvents like ether, xylene, and acetone, and relies on well-established unit operations such as Grignard formation and reflux, making it highly adaptable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doxylamine Succinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of safe and effective OTC medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of the Grignard-based synthesis are managed with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Doxylamine Succinate meets the exacting standards required by global regulatory bodies. Our commitment to process optimization allows us to deliver a product that is not only chemically pure but also economically competitive.
We invite pharmaceutical manufacturers and procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized implementation of this patented route, we can help you reduce overall manufacturing costs while securing a reliable supply of this essential antihistamine. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical expertise can become a strategic asset to your supply chain.
