Advanced Metal-Free Synthesis of 1,5-Disubstituted 1,2,3-Triazoles for Commercial Pharmaceutical Applications
Advanced Metal-Free Synthesis of 1,5-Disubstituted 1,2,3-Triazoles for Commercial Pharmaceutical Applications
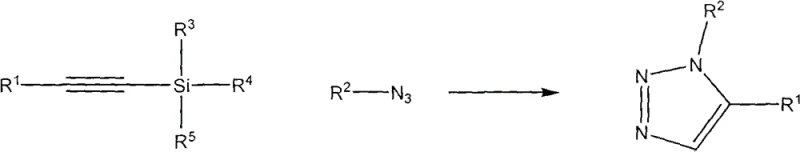
The pharmaceutical and agrochemical industries continuously seek robust, scalable, and environmentally benign synthetic routes for constructing nitrogen-containing heterocycles, particularly 1,2,3-triazoles, which serve as privileged scaffolds in drug discovery. Patent CN102336719B introduces a transformative methodology for synthesizing 1,5-disubstituted 1,2,3-triazole compounds utilizing silicon-based alkyne precursors. This innovation addresses critical bottlenecks in traditional heterocycle synthesis by eliminating the reliance on toxic transition metal catalysts and严苛 reaction conditions. By leveraging a base-mediated desilylation strategy, this process achieves high regioselectivity and yield under mild thermal conditions, positioning it as a superior alternative for the manufacturing of high-purity pharmaceutical intermediates. The technology represents a significant leap forward in process chemistry, offering a streamlined pathway that aligns with modern green chemistry principles while maintaining the rigorous quality standards required for active pharmaceutical ingredient (API) production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazoles has been dominated by the Huisgen 1,3-dipolar cycloaddition, which typically suffers from poor regioselectivity, yielding mixtures of 1,4- and 1,5-disubstituted isomers that are difficult and costly to separate. While the advent of Copper(I)-catalyzed Azide-Alkyne Cycloaddition (CuAAC) resolved the regioselectivity issue for 1,4-isomers, it introduced new challenges regarding residual heavy metal contamination, necessitating expensive and complex purification steps to meet stringent pharmaceutical limits. Furthermore, accessing the 1,5-disubstituted isomer traditionally required Ruthenium catalysts, which are not only prohibitively expensive for large-scale operations but also pose significant supply chain risks due to the scarcity of precious metals. These conventional methods often demand rigorous exclusion of moisture and oxygen, requiring specialized reactor setups and inert atmosphere handling that drastically increase capital expenditure and operational overhead for chemical manufacturers.
The Novel Approach
In stark contrast to metal-dependent methodologies, the novel approach detailed in the patent utilizes readily available silyl-alkyne compounds as stable surrogates for terminal alkynes, enabling a highly selective construction of the 1,5-disubstituted triazole core without any transition metal involvement. This metal-free protocol operates effectively in common organic solvents such as dimethyl sulfoxide (DMSO) or tetrahydrofuran (THF) at moderate temperatures ranging from 10°C to 100°C, significantly reducing energy consumption compared to high-temperature thermal cycloadditions. The use of inexpensive inorganic or organic bases as desilylation reagents replaces costly catalysts, fundamentally altering the cost structure of the synthesis by removing the need for metal scavengers and complex waste treatment associated with heavy metals. This streamlined process not only simplifies the reaction setup by removing the need for anaerobic protection but also enhances the overall safety profile of the manufacturing operation, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking efficiency.
Mechanistic Insights into Base-Mediated Desilylative Cycloaddition
The core mechanistic advantage of this technology lies in the unique reactivity of the silicon-carbon triple bond, which serves as a masked acetylide equivalent that is activated in situ by the base reagent. Upon addition of a desiliconization agent such as potassium hydroxide or cesium carbonate, the silyl group is cleaved to generate a reactive acetylide species that immediately undergoes nucleophilic attack on the terminal nitrogen of the organic azide. This intramolecular cyclization proceeds through a concerted pathway that inherently favors the formation of the 1,5-regioisomer due to the electronic and steric influence of the transient intermediate, thereby bypassing the thermodynamic mixtures observed in uncatalyzed thermal reactions. The absence of a metal coordination sphere eliminates the possibility of metal-ligand complexation side reactions, resulting in a cleaner reaction profile with fewer byproducts and a simplified impurity spectrum that facilitates downstream purification efforts.
Furthermore, the tolerance of this system to various functional groups on both the alkyne and azide components demonstrates remarkable versatility, allowing for the incorporation of diverse aryl and alkyl substituents without compromising yield or selectivity. The reaction mechanism is robust enough to proceed efficiently even in the presence of moisture or air, which is a rare and valuable trait for anionic cyclization processes that typically require glovebox conditions. This resilience is attributed to the rapid kinetics of the desilylation-cyclization sequence, which outcompetes potential degradation pathways of the reactive intermediates. For R&D teams, understanding this mechanism provides confidence in scaling the process, as the lack of sensitive catalytic cycles means that minor fluctuations in feedstock quality or atmospheric conditions will not lead to catastrophic batch failures, ensuring consistent production of high-purity OLED material or pharmaceutical precursors.
How to Synthesize 1,5-Disubstituted 1,2,3-Triazoles Efficiently
Implementing this synthesis route in a pilot or production plant involves a straightforward charging sequence where silyl-alkynes, azides, and a polar organic solvent are combined in a standard stirred tank reactor. The process is initiated by the controlled addition of a solid or liquid base reagent, after which the mixture is maintained at a mild temperature, typically between 20°C and 50°C, to drive the reaction to completion within a few hours. Monitoring is easily achieved via thin-layer chromatography or HPLC, and upon consumption of the starting materials, the reaction is quenched with aqueous acid to neutralize the base and facilitate phase separation. The detailed standardized synthetic steps, including specific molar ratios and workup parameters, are outlined in the guide below to ensure reproducibility and compliance with good manufacturing practices.
- Charge a reactor with silyl-alkyne compounds, azide compounds, organic solvent, and a desiliconization base reagent.
- Maintain the reaction temperature between 10°C and 100°C, preferably 20°C to 50°C, and stir until raw materials are consumed.
- Acidify the mixture with inorganic acid, extract with ethyl acetate, wash, dry, and purify via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this metal-free synthesis route offers profound economic and logistical benefits that directly impact the bottom line of chemical manufacturing operations. By completely eliminating the need for precious metal catalysts like Ruthenium or Copper, the process removes a major variable cost driver and mitigates the risk associated with the volatile pricing and geopolitical supply constraints of rare earth elements. The simplified workup procedure, which relies on standard aqueous extraction and crystallization rather than specialized metal scavenging resins or chromatography, significantly reduces the consumption of auxiliary materials and shortens the overall cycle time per batch. This efficiency translates into substantial cost savings in both raw material procurement and waste disposal, as the effluent streams are free from heavy metal contaminants that require expensive treatment protocols before discharge.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts fundamentally lowers the direct material costs associated with each production run, as expensive ligands and metal salts are replaced by commodity chemicals like potassium hydroxide. Additionally, the removal of metal scavenging steps and the associated filtration media reduces the operational expenditure related to consumables and labor, leading to a more lean and cost-effective manufacturing process. The high atom economy and selectivity of the reaction minimize the formation of isomeric byproducts, thereby maximizing the yield of the desired 1,5-isomer and reducing the loss of valuable starting materials during purification. These factors collectively contribute to a significantly reduced cost of goods sold (COGS), enhancing the competitiveness of the final pharmaceutical intermediate in the global market.
- Enhanced Supply Chain Reliability: The reliance on readily available silyl-alkynes and organic azides, which are commercially sourced from stable supply chains, ensures a consistent availability of raw materials without the bottlenecks often seen with specialized catalysts. The robustness of the reaction conditions, which do not require inert gas protection or anhydrous solvents, allows for production in standard multipurpose reactors, increasing the flexibility of manufacturing sites to allocate capacity as needed. This operational simplicity reduces the dependency on specialized equipment and highly trained personnel for handling sensitive reagents, thereby minimizing the risk of production delays due to equipment failure or staffing issues. Consequently, partners can rely on a more resilient supply chain capable of meeting demanding delivery schedules for complex polymer additives or drug candidates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the exothermic nature of the reaction being manageable under mild thermal conditions, reducing the engineering controls needed for heat dissipation compared to highly exothermic metal-catalyzed reactions. The absence of heavy metals in the waste stream simplifies environmental compliance and permitting, as the effluent can be treated using standard wastewater protocols without the need for specialized heavy metal precipitation units. This alignment with green chemistry principles not only reduces the environmental footprint of the manufacturing facility but also future-proofs the supply chain against increasingly stringent global regulations on chemical emissions and waste disposal. The ability to produce high volumes with minimal environmental impact makes this technology a sustainable choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silyl-alkyne based triazole synthesis, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and technical specifications disclosed in the patent literature, ensuring accuracy and relevance for industrial application. Understanding these details is crucial for assessing the feasibility of integrating this route into existing manufacturing workflows for fine chemical intermediates.
Q: What is the primary advantage of using silyl-alkynes over terminal alkynes in triazole synthesis?
A: Using silyl-alkynes allows for the regioselective synthesis of 1,5-disubstituted 1,2,3-triazoles without the need for expensive transition metal catalysts like Ruthenium, while avoiding the 1,4-isomer formation typical of Copper-catalyzed reactions.
Q: Does this process require strict anaerobic conditions?
A: No, one of the significant operational benefits of this patented method is that it does not require anaerobic protection or inert gas shielding, simplifying the equipment requirements and reducing operational complexity.
Q: How is the product purified after the reaction is complete?
A: The workup involves standard aqueous acidification followed by organic extraction, washing with brine, and drying. The final product is isolated through recrystallization or column chromatography, ensuring high purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic technologies like the metal-free silyl-alkyne route to enhance the efficiency and sustainability of our clients' supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with international quality standards. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 1,2,3-triazole intermediate meets the exacting requirements of the pharmaceutical and agrochemical industries. Our commitment to technical excellence allows us to deliver high-purity intermediates that accelerate your drug development timelines while minimizing regulatory risks.
We invite you to collaborate with us to leverage this advanced chemistry for your next project, where our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume and purity needs. By partnering with our technical procurement team, you can access specific COA data and comprehensive route feasibility assessments that demonstrate the tangible benefits of switching to this metal-free platform. Contact us today to discuss how we can support your R&D and commercial goals with reliable, cost-effective, and scalable chemical solutions.
