Advanced Manufacturing of 1H-1,2,3-Triazole: A Safer Route for Pharmaceutical Intermediates
The landscape of fine chemical synthesis is constantly evolving, driven by the dual imperatives of operational safety and economic efficiency. A pivotal advancement in this domain is documented in Chinese Patent CN1242996C, which outlines a robust method for synthesizing 1H-1,2,3-triazole, a critical intermediate for the novel penicillin synergist Tazobactam Sodium. This patent addresses long-standing inefficiencies in traditional manufacturing protocols by introducing a novel pretreatment strategy and a streamlined workup procedure. By leveraging ion exchange resin technology to stabilize glyoxal feedstocks and replacing hazardous high-temperature rearrangement steps with a simultaneous distillation-rearrangement technique, this methodology offers a compelling value proposition for industrial scale-up. For procurement leaders and technical directors seeking a reliable pharmaceutical intermediates supplier, understanding these mechanistic improvements is essential for securing a stable supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1H-1,2,3-triazole has relied on routes described in literature such as the work by Harada et al., which, while chemically valid, present significant operational bottlenecks for large-scale production. The conventional pathway typically involves the formation of glyoxal monoxime hydrazone followed by a cumbersome isolation process that necessitates azeotropic water removal using n-butyl acetate. Furthermore, the critical rearrangement step requires heating the reaction mixture in toluene at approximately 150°C for nearly 20 hours. This extended exposure to high thermal energy not only poses severe safety risks regarding thermal decomposition and potential runaway reactions but also leads to product degradation and discoloration. Additionally, the reliance on toluene, a solvent with known toxicity and strict environmental regulations, complicates waste management and increases the burden on occupational health and safety protocols, making the traditional route less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the methodology disclosed in CN1242996C introduces a paradigm shift by fundamentally altering the reaction environment and workup sequence to enhance both safety and yield. The most significant innovation lies in the elimination of the toluene heating step; instead, the process utilizes ethyl acetate for extraction, followed by distillation under reduced pressure. This clever engineering allows for the simultaneous removal of the solvent and the completion of the rearrangement reaction, effectively collapsing two distinct unit operations into one. By avoiding the use of high-boiling n-butyl acetate and toxic toluene, the new route drastically reduces energy consumption and minimizes the risk of product deterioration caused by prolonged heating. This streamlined approach not only simplifies the equipment requirements but also aligns perfectly with the goals of cost reduction in pharmaceutical intermediates manufacturing by shortening cycle times and improving overall process throughput.
Mechanistic Insights into Ion Exchange Stabilization and Rearrangement
To fully appreciate the technical superiority of this synthesis, one must examine the specific chemical mechanisms that govern the stability of the starting materials and the efficiency of the cyclization. A primary challenge in using glyoxal is its tendency to undergo acid-catalyzed polymerization, which generates difficult-to-remove yellow polymeric byproducts and consumes the valuable starting material. The patented process mitigates this by employing an anion exchange resin to pretreat the glyoxal aqueous solution. This step effectively scavenges acidic impurities that act as polymerization catalysts, thereby stabilizing the glyoxal and ensuring that the subsequent condensation with hydroxylamine hydrochloride and hydrazine hydrate proceeds with high selectivity towards the desired monoxime hydrazone, dioxime, and dihydrazone mixture.
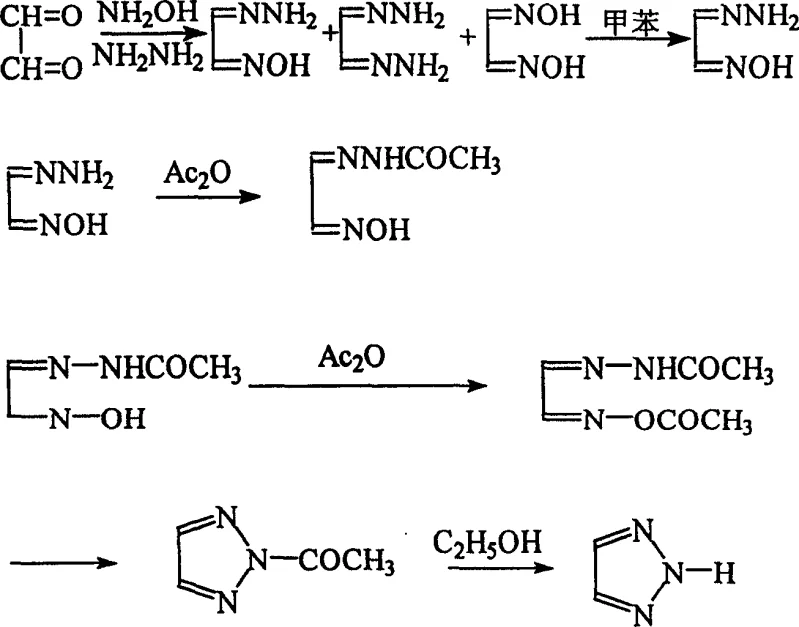
Following the formation of the intermediate mixture, the process leverages a unique physicochemical phenomenon during the solvent removal phase. Rather than isolating the crude mixture through crystallization or complex extractions, the reaction solution is subjected to vacuum distillation. As the ethyl acetate is removed, the increasing concentration and thermal energy provided during distillation drive the rearrangement of the dioxime and dihydrazone species into the single, stable glyoxal monoxime hydrazone. This in-situ transformation ensures that the downstream acetylation and cyclization steps begin with a highly enriched intermediate, which is crucial for achieving the reported final product purity of 98% as measured by HPLC. This mechanistic control over impurity profiles is vital for R&D directors focused on the quality of high-purity pharmaceutical intermediates.
How to Synthesize 1H-1,2,3-Triazole Efficiently
The implementation of this synthesis route requires precise control over reaction parameters, particularly during the pretreatment and acetylation stages, to maximize the yield of the key intermediates. The process begins with the stabilization of the glyoxal feedstock, followed by a carefully controlled condensation reaction where temperature management is critical to prevent exothermic runaway. Subsequent acetylation using acetic anhydride converts the hydrazone into an N-acetyl derivative, which then undergoes cyclization in the presence of sodium acetate to form the triazole ring. The final step involves alcoholysis with ethanol to cleave the acetyl group and release the target molecule. For a detailed breakdown of the specific molar ratios, temperature gradients, and processing times required to replicate this high-yield pathway, please refer to the standardized synthesis guide below.
- Pretreat glyoxal aqueous solution with anion exchange resin to remove acids and prevent polymerization.
- React pretreated glyoxal with hydroxylamine hydrochloride and hydrazine hydrate, then extract with ethyl acetate.
- Distill ethyl acetate under reduced pressure to simultaneously remove solvent and induce rearrangement to monoxime hydrazone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic advantages for organizations looking to optimize their supply chain resilience and reduce total landed costs. The elimination of toxic solvents like toluene and the reduction in processing time directly translate to lower operational expenditures and reduced regulatory compliance burdens. By simplifying the purification train and removing the need for specialized high-temperature equipment capable of handling toluene reflux for extended periods, manufacturers can achieve significant capital expenditure savings. Furthermore, the improved stability of the raw materials ensures more consistent batch-to-batch quality, which is a critical factor for maintaining uninterrupted production schedules in the competitive pharmaceutical market.
- Cost Reduction in Manufacturing: The replacement of expensive and energy-intensive azeotropic distillation with a simplified vacuum distillation process significantly lowers utility costs. By eliminating the need for prolonged heating cycles and reducing the volume of solvents required for workup, the overall variable cost per kilogram of product is substantially decreased. Additionally, the higher selectivity achieved through glyoxal pretreatment reduces the loss of raw materials to polymeric waste, further enhancing the economic efficiency of the process without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: The use of widely available and less regulated solvents like ethyl acetate, as opposed to toluene, mitigates the risk of supply disruptions caused by environmental restrictions or transportation hazards. The robustness of the ion exchange pretreatment step ensures that variations in commercial glyoxal quality do not negatively impact the reaction outcome, providing a more stable and predictable supply of 1H-1,2,3-triazole. This reliability is essential for downstream manufacturers who depend on consistent feedstock availability to meet their own production commitments for beta-lactamase inhibitors.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, utilizing standard unit operations such as filtration, extraction, and distillation that are easily transferred from pilot plant to commercial scale. The reduction in hazardous waste generation, specifically the avoidance of toluene-contaminated streams, simplifies wastewater treatment and aligns with increasingly stringent global environmental standards. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability credentials of the supply chain, a factor that is becoming increasingly important for multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1H-1,2,3-triazole synthesized via this advanced method. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding vendor selection and process integration.
Q: How does the new synthesis method improve safety compared to conventional routes?
A: The novel process eliminates the use of toxic toluene for heating rearrangement and avoids prolonged heating at 150°C, significantly reducing operator health risks and thermal decomposition hazards.
Q: What is the purity level achievable with this optimized synthetic route?
A: High-pressure liquid phase analysis confirms that the final 1H-1,2,3-triazole product achieves a purity of 98%, meeting stringent specifications for pharmaceutical intermediates.
Q: Why is ion exchange resin pretreatment critical for glyoxal stability?
A: Glyoxal aqueous solutions naturally contain acids that catalyze unwanted polymerization. Removing these acids via ion exchange resin stabilizes the raw material, ensuring higher yields of the key monoxime hydrazone intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1H-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1242996C are fully realized in practical, high-volume manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1H-1,2,3-triazole meets the exacting standards required for the synthesis of critical antibiotics like Tazobactam Sodium.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to our supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate our commitment to quality and efficiency before making any long-term commitments.
