Revolutionizing Roxadustat Production: A Green, High-Yield Synthetic Strategy for Global Supply Chains
Revolutionizing Roxadustat Production: A Green, High-Yield Synthetic Strategy for Global Supply Chains
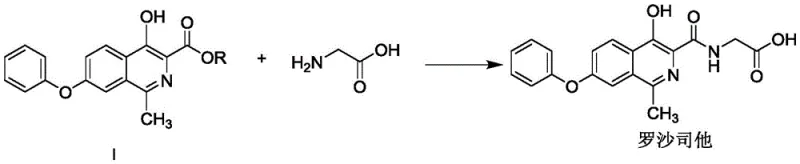
The pharmaceutical landscape for treating renal anemia has been significantly transformed by the introduction of Roxadustat, a hypoxia-inducible factor prolyl hydroxylase (HIF-PH) inhibitor. However, the complexity of its synthesis has long posed challenges for cost-effective and scalable manufacturing. Patent CN115197139A introduces a groundbreaking preparation method for Roxadustat and its key intermediate, 4-hydroxy-1-methyl-7-phenoxy-3-isoquinoline formate. This innovation addresses critical bottlenecks in the existing supply chain by offering a route that is not only shorter but also utilizes readily available, low-cost raw materials. The technical breakthrough lies in a novel one-pot strategy that combines deprotection and acetylation, drastically simplifying the process flow while maintaining high yields. For global procurement and R&D teams, this patent represents a pivotal shift towards more sustainable and economically viable production of this vital API intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Roxadustat has been plagued by inefficient multi-step processes that hinder large-scale industrial adoption. The original research route, disclosed in world patent WO2004108681, relies on 3,4-dicyano nitrobenzene as a starting material and requires eleven distinct reaction steps. This lengthy sequence involves hazardous reagents and expensive catalysts, such as palladium for methylation, which notoriously suffers from low yields of only 33%. Similarly, subsequent optimizations like those in WO2013013609 still retain excessive step counts and rely on costly initial raw materials that lack commercial availability. Other routes, such as those utilizing tyrosine or m-bromoacetophenone, face issues with harsh reaction conditions, poor selectivity leading to difficult-to-remove impurities, or the need for specialized high-pressure equipment for hydrogenation. These conventional methodologies collectively result in high production costs, significant environmental burdens due to waste generation, and fragile supply chains vulnerable to raw material shortages.
The Novel Approach
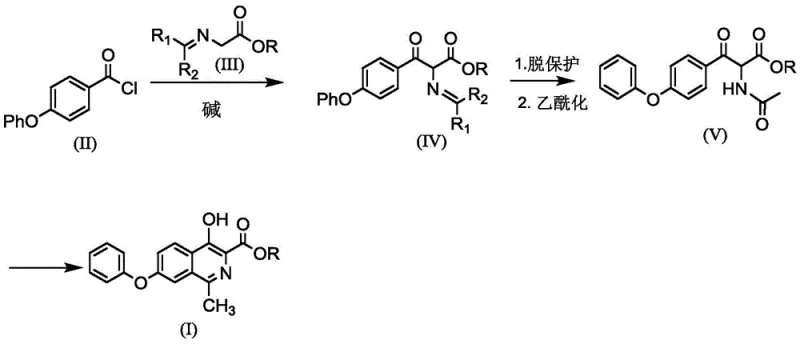
In stark contrast, the methodology outlined in CN115197139A offers a streamlined alternative that directly targets the economic and operational inefficiencies of prior art. The new route initiates with the condensation of 4-phenoxybenzoyl chloride and a protected glycine derivative, both of which are commercially accessible or easily prepared. A key innovation is the execution of deprotection and acetylation in a continuous one-pot manner, eliminating the need for intermediate isolation and purification. This telescoping of steps not only reduces solvent consumption and processing time but also minimizes product loss typically associated with multiple work-ups. Furthermore, the final cyclization step achieves yields exceeding 90% under mild conditions, a substantial improvement over the variable and often low yields of traditional methods. By focusing on atom economy and operational simplicity, this approach provides a robust foundation for reliable pharmaceutical intermediate supplier networks seeking to optimize their manufacturing portfolios.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core of this synthetic advancement lies in the sophisticated manipulation of reaction mechanisms to maximize efficiency and purity. The process begins with a base-catalyzed acylation where a metal alkoxide, such as potassium tert-butoxide, facilitates the nucleophilic attack of the glycine derivative on the acid chloride. This step is carefully controlled at low temperatures (-5 to 30°C) to prevent side reactions and ensure high regioselectivity. Following this, the protocol employs a strategic deprotection using strong acids like hydrochloric acid, which cleaves the protecting group to reveal the reactive amine. Crucially, instead of isolating this unstable intermediate, the process immediately proceeds to acetylation with acetic anhydride. This seamless transition prevents degradation and maintains the integrity of the molecular scaffold, ensuring that the subsequent cyclization precursor is formed with minimal impurity generation.
The final and perhaps most critical transformation is the cyclization reaction, which constructs the isoquinoline core. Unlike traditional methods that rely on phosphorus oxychloride—a reagent known for generating significant phosphorus waste—this novel route utilizes oxalyl chloride in conjunction with a Lewis acid catalyst such as aluminum trichloride or ferric chloride. This mechanistic choice is twofold: firstly, oxalyl chloride acts as a potent dehydrating agent that activates the carbonyl group for intramolecular attack; secondly, the Lewis acid stabilizes the transition state, lowering the activation energy and allowing the reaction to proceed efficiently at room temperature. The result is a clean conversion to the target isoquinoline structure with yields consistently above 90%. This mechanism not only enhances the chemical yield but also drastically improves the impurity profile, making downstream purification significantly easier and more cost-effective for high-purity pharmaceutical intermediate manufacturing.
How to Synthesize 4-Hydroxy-1-methyl-7-phenoxy-3-isoquinolinecarboxylate Efficiently
The synthesis of this critical intermediate is achieved through a logical three-stage sequence designed for operational ease and high throughput. The process leverages commercially available starting materials to minimize supply chain risks and employs a one-pot technique to reduce unit operations. Detailed below is the strategic overview of the synthesis, highlighting the key transformations that define this efficient pathway. For laboratory and pilot plant teams looking to implement this technology, the following guide outlines the critical parameters and procedural logic derived directly from the patent specifications.
- React 4-phenoxybenzoyl chloride with a protected glycine derivative under basic conditions to form the keto-amide intermediate.
- Perform a one-pot deprotection using strong acid followed immediately by acetylation with acetic anhydride to generate the cyclization precursor.
- Execute the final cyclization using oxalyl chloride and a Lewis acid catalyst to obtain the isoquinoline core with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the manufacturing process, which directly translates to reduced operational expenditures. By eliminating the need for expensive transition metal catalysts like palladium and avoiding high-pressure hydrogenation steps, the capital expenditure required for plant equipment is significantly lowered. Additionally, the ability to recycle the benzophenone byproduct generated during the deprotection step back into the synthesis of the starting glycine derivative creates a closed-loop system that further drives down raw material costs. This circular economy approach within the synthesis ensures that the cost reduction in pharmaceutical intermediate manufacturing is structural and sustainable, rather than temporary.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the implementation of a one-pot deprotection-acetylation sequence remove several costly unit operations from the production line. This reduction in processing steps leads to substantial cost savings in terms of labor, energy consumption, and solvent usage. Furthermore, the high yield of the cyclization step minimizes the loss of valuable intermediates, ensuring that the overall material throughput is maximized. The use of oxalyl chloride instead of phosphorus reagents also reduces the financial burden associated with hazardous waste disposal and environmental compliance, contributing to a leaner and more profitable cost structure.
- Enhanced Supply Chain Reliability: The reliance on commercially available and easily preparable raw materials such as 4-phenoxybenzoic acid and glycine esters mitigates the risk of supply disruptions often associated with custom-synthesized starting materials. The robustness of the reaction conditions, which operate at mild temperatures and atmospheric pressure, ensures that production can be maintained consistently without the need for specialized or scarce equipment. This stability allows for better forecasting and inventory management, reducing lead time for high-purity pharmaceutical intermediates and ensuring a steady flow of material to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, featuring mild reaction conditions that are safe and easy to control on a multi-ton scale. The avoidance of toxic phosphorus waste and the reduction of solvent-intensive purification steps align with increasingly stringent global environmental regulations. This green chemistry profile not only simplifies the permitting process for new manufacturing facilities but also enhances the corporate sustainability metrics of the supply chain, making it an attractive option for environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived from the specific technical disclosures and beneficial effects outlined in the patent documentation, providing clarity on how this method compares to established industry standards.
Q: How does this new route improve cost efficiency compared to traditional Palladium-catalyzed methods?
A: This method eliminates the need for expensive Palladium catalysts and high-pressure hydrogenation equipment. Furthermore, the benzophenone byproduct generated during deprotection can be recycled to regenerate the starting glycine derivative, significantly reducing raw material costs.
Q: What are the environmental benefits of using oxalyl chloride over phosphorus oxychloride?
A: The use of oxalyl chloride avoids the generation of large quantities of phosphorus-containing waste, which is difficult to treat and environmentally hazardous. This results in a cleaner process with lower three-waste treatment costs and better compliance with green chemistry standards.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route features mild reaction conditions (0-30°C), avoids dangerous reagents, and utilizes a one-pot technique that simplifies operation. The high yields (>90% in cyclization) and use of commercially available raw materials make it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Intermediate Supplier
As the demand for effective treatments for renal anemia continues to grow, the need for a stable and cost-efficient supply of Roxadustat intermediates has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN115197139A to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the international pharmaceutical market. With our stringent purity specifications and rigorous QC labs, we guarantee that every batch of intermediate meets the highest standards of quality and consistency required for API synthesis.
We invite you to explore how our optimized manufacturing capabilities can enhance your supply chain resilience and cost efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our innovative approach can drive your project forward with speed and reliability.
