Advanced Roxadustat Manufacturing: Overcoming Conventional Synthesis Limitations for Commercial Scale
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical therapeutic agents, and the synthesis of Roxadustat, a hypoxia-inducible factor prolyl hydroxylase inhibitor, represents a significant area of focus for treating anemia associated with chronic kidney disease. Patent CN114933563B discloses a novel synthesis method for Roxadustat and its key intermediates, addressing the longstanding challenges of cost, complexity, and impurity control found in earlier methodologies. This technical insight report analyzes the strategic advantages of this new pathway, which utilizes oxidative functionalization to construct the isoquinoline core without relying on expensive precious metal catalysts. By leveraging specific oxidants such as persulfates and optimizing solvent systems, the disclosed method achieves high yields under mild conditions, offering a compelling value proposition for procurement and supply chain stakeholders seeking a reliable pharmaceutical intermediates supplier. The structural integrity of the final API is paramount, and the patent emphasizes rigorous control over process-related impurities to ensure compliance with international regulatory standards.
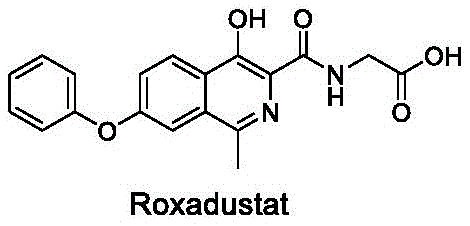
The limitations of conventional methods for synthesizing Roxadustat are well-documented in the prior art, often involving multi-step sequences that are economically and operationally inefficient. For instance, earlier routes frequently relied on palladium-catalyzed hydrogenation for methylation steps, which not only introduces high raw material costs due to the price of palladium but also necessitates complex purification processes to remove trace metal residues from the final product. Additionally, some traditional pathways require high-temperature reactions or the use of hazardous reagents like hydrogen peroxide in unoptimized conditions, posing safety risks and energy consumption challenges during commercial scale-up. The presence of difficult-to-remove high-boiling solvents and the generation of significant by-products further complicate the downstream processing, often requiring column chromatography which is impractical for multi-kilogram or ton-scale production. These factors collectively contribute to extended lead times and increased manufacturing costs, creating bottlenecks for supply chain heads aiming for consistent availability of high-purity pharmaceutical intermediates.
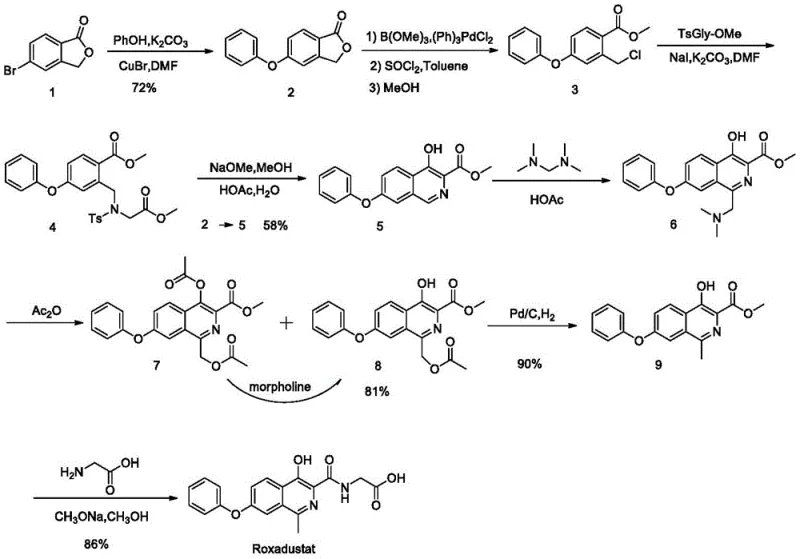
In contrast, the novel approach detailed in the patent utilizes a streamlined oxidative strategy that fundamentally simplifies the construction of the key intermediates. The core innovation involves the reaction of a starting material SM with a compound SM-A in the presence of an oxidant, such as sodium persulfate, to directly introduce the necessary functional groups without the need for precious metals. This method operates under mild temperature ranges, typically between 30°C and 100°C, which significantly reduces energy consumption and enhances operational safety compared to high-temperature alternatives. The use of common amides like N,N-dimethylacetamide (DMA) or N,N-dimethylformamide (DMF) as solvents or reagents further simplifies the supply chain, as these are readily available commodity chemicals. By eliminating the need for column chromatography and enabling simple work-up procedures like filtration and washing, the new route drastically reduces solvent waste and processing time. This shift from complex catalytic cycles to straightforward oxidative transformations represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, aligning perfectly with the goals of modern green chemistry and efficient process engineering.
Mechanistic Insights into Persulfate-Mediated Oxidative Functionalization
The mechanistic pathway of this synthesis relies on the efficient generation of radical species or electrophilic intermediates facilitated by the persulfate oxidant system. In the critical step converting compound SM to intermediate M1, the oxidant activates the amide reagent (SM-A), allowing for the selective functionalization of the isoquinoline ring system. This reaction is highly sensitive to the molar ratios of the reagents, with the patent specifying that an excess of SM-A relative to SM is beneficial for driving the reaction to completion while maintaining cost efficiency. The choice of oxidant is also crucial, with alkali metal persulfates like sodium persulfate or potassium persulfate offering a balance of reactivity and stability. The reaction mechanism avoids the formation of stable metal-ligand complexes that are typical in palladium chemistry, thereby preventing the entrapment of metal impurities within the crystal lattice of the product. This chemical elegance ensures that the resulting intermediate M1 possesses a high degree of purity, which is essential for the subsequent steps in the synthesis cascade. Furthermore, the reaction conditions are tuned to minimize side reactions, such as over-oxidation or hydrolysis, which could lead to the formation of hard-to-remove impurities that would compromise the quality of the final API.
Impurity control is a central theme of this patent, particularly regarding the management of Compound I and Compound II, which are identified as critical process impurities. The patent details how Compound I can be generated during the hydrolysis of intermediate M1-A to M2-A and how it can subsequently transform into Compound II during the reduction step. To mitigate this, the process includes specific control strategies, such as optimizing the acid concentration and reaction temperature during hydrolysis to minimize the formation of Compound I. Additionally, the reduction step using zinc powder is carefully monitored to prevent the carryover of precursors that could lead to Compound II. The use of preparative HPLC is described as a method for isolating and characterizing these impurities, ensuring that the manufacturing process can be adjusted to keep their levels well below the ICH identification threshold. This rigorous approach to impurity profiling demonstrates a deep understanding of the reaction chemistry and provides a robust framework for quality assurance, ensuring that the high-purity pharmaceutical intermediates produced meet the stringent requirements of global regulatory bodies.
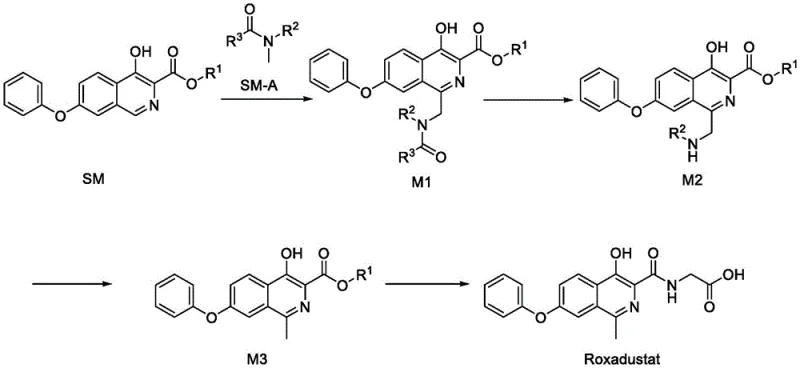
How to Synthesize Roxadustat Efficiently
The synthesis of Roxadustat via this patented method involves a sequence of well-defined chemical transformations that prioritize yield and purity at every stage. The process begins with the oxidative coupling of the isoquinoline starting material with an amide derivative, followed by acidic hydrolysis to reveal the amine functionality. Subsequent reduction of the intermediate using zinc powder in an acidic medium installs the methyl group at the 1-position of the isoquinoline ring, a critical structural feature for biological activity. The final step involves the amidation with glycine or its salt to form the final API. Each step is designed to be telescoped or simplified to reduce isolation losses and solvent usage. The detailed standardized synthesis steps see the guide below.
- React compound SM with compound SM-A in the presence of a persulfate oxidant to form intermediate M1.
- Hydrolyze intermediate M1 under acidic conditions to obtain intermediate M2.
- Reduce intermediate M2 using a metal powder and hydrogen donor to form intermediate M3, followed by amidation with glycine.
Commercial Advantages for Procurement and Supply Chain Teams
The commercial implications of adopting this synthesis route are profound, offering tangible benefits for procurement managers and supply chain heads focused on efficiency and reliability. By eliminating the dependency on precious metal catalysts like palladium, the manufacturing process becomes immune to the volatility of precious metal markets, ensuring more stable pricing and cost reduction in pharmaceutical intermediates manufacturing. The simplified post-treatment procedures, which often involve simple filtration and washing rather than complex chromatographic separations, significantly reduce the consumption of solvents and consumables. This reduction in material usage not only lowers direct costs but also simplifies waste management and environmental compliance, contributing to a more sustainable manufacturing footprint. The mild reaction conditions further enhance equipment longevity and reduce maintenance costs, as reactors are not subjected to extreme temperatures or corrosive environments typical of harsher synthetic methods. These factors combine to create a manufacturing process that is both economically attractive and operationally robust.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the associated metal scavenging steps results in substantial cost savings throughout the production lifecycle. The use of cheap and easily obtained raw materials, such as persulfates and common amides, further drives down the bill of materials. Additionally, the high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as by-products. The simplified purification process reduces the need for expensive chromatography resins and large volumes of high-purity solvents. These cumulative effects lead to a significantly lower cost of goods sold (COGS), allowing for more competitive pricing in the market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized catalysts enhances the resilience of the supply chain against disruptions. Since the key reagents are widely available from multiple suppliers, the risk of single-source bottlenecks is minimized. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even with standard grade reagents. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream customers. The ability to scale the process from laboratory to commercial production without significant re-engineering further supports supply chain stability, enabling rapid response to market demand fluctuations.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, with reaction conditions that are safe and manageable in large-scale reactors. The reduction in hazardous waste generation, due to the absence of heavy metals and the minimization of solvent use, simplifies environmental permitting and compliance. The mild temperatures and pressures reduce the energy load on the facility, contributing to lower carbon emissions and operational costs. The straightforward work-up procedures facilitate faster batch turnover, increasing the overall capacity of the manufacturing plant. This scalability ensures that the supply can grow in tandem with the market demand for Roxadustat, supporting long-term business growth and partnership stability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific technical advantages and impurity control strategies detailed in the patent documentation. Understanding these aspects is crucial for technical teams evaluating the feasibility of this route for their specific production needs. The answers provide clarity on how the process handles critical quality attributes and operational challenges.
Q: How does this new synthesis route improve impurity control compared to prior art?
A: The new method specifically controls the formation of Compound I and Compound II through optimized reaction conditions and purification steps, ensuring the final API meets strict ICH guidelines for impurity limits.
Q: What are the cost advantages of avoiding palladium catalysts in Roxadustat production?
A: Eliminating precious metal palladium catalysts removes the need for expensive metal scavenging steps and reduces raw material costs significantly, leading to substantial overall cost savings in manufacturing.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions, cheap and easily obtained raw materials, and simple post-treatment processes, making it highly suitable for commercial scale-up and continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Supplier
The technical potential of this synthesis route is best realized through partnership with an experienced CDMO capable of executing complex chemistry with precision. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab scale to industrial manufacturing is seamless. Our facility is equipped with stringent purity specifications and rigorous QC labs to monitor every critical parameter, from raw material intake to final API release. We understand the nuances of oxidative functionalization and impurity control, allowing us to consistently deliver high-purity pharmaceutical intermediates that meet global regulatory standards. Our commitment to quality ensures that the theoretical advantages of this patent are fully translated into commercial reality.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into the economic impact of switching to this palladium-free method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Together, we can secure a stable and cost-effective supply of Roxadustat intermediates, driving value for your organization and ensuring patient access to this critical medication.
