Advanced Enzymatic Resolution of D-Tyrosine for Industrial Pharmaceutical Applications
The pharmaceutical industry's relentless pursuit of chiral purity has brought significant attention to the efficient synthesis of non-natural amino acids, specifically D-tyrosine. Patent CN1900298A introduces a groundbreaking enzymatic method for preparing D-tyrosine from DL-tyrosine, addressing the critical need for scalable and cost-effective chiral intermediates. Unlike traditional extraction methods which are limited by natural abundance, this novel biocatalytic route leverages the high stereoselectivity of D-acylating hydrolase to achieve exceptional optical purity. The process begins with the acylation of racemic DL-tyrosine, followed by a highly specific enzymatic hydrolysis that selectively targets the D-isomer precursor. This technological advancement is particularly relevant for the synthesis of complex peptide drugs, such as the tocolytic agent atosiban, where the D-configuration is essential for biological activity. By shifting from laborious chemical resolution to a streamlined enzymatic process, manufacturers can now access high-purity D-tyrosine with yields exceeding 88%, marking a substantial leap forward in industrial amino acid production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of D-tyrosine has been fraught with significant technical and economic challenges that hinder large-scale adoption. Conventional chemical resolution methods often rely on the formation of diastereomeric salts using expensive chiral resolving agents, a process that is inherently inefficient due to the theoretical maximum yield of 50% for the desired isomer. Furthermore, these chemical processes frequently require harsh reaction conditions, including extreme pH levels and elevated temperatures, which can lead to racemization and the degradation of the sensitive amino acid structure. The downstream processing in traditional methods is also cumbersome, involving multiple recrystallization steps to achieve acceptable enantiomeric excess, which drastically increases solvent consumption and waste generation. Additionally, the recovery of the resolving agent is often incomplete, adding to the overall raw material costs and environmental footprint. For procurement managers, these inefficiencies translate into volatile supply chains and unpredictable pricing, making it difficult to secure a consistent supply of high-quality D-tyrosine for critical drug manufacturing pipelines.
The Novel Approach
In stark contrast, the enzymatic method disclosed in the patent offers a transformative solution by utilizing immobilized D-acylating hydrolase to drive the resolution with unparalleled specificity. This biocatalytic approach operates under mild physiological conditions, typically between 40°C and 60°C, preserving the structural integrity of the amino acid while minimizing energy consumption. The use of an immobilized enzyme column allows for a continuous flow process, where the substrate solution passes through the catalyst bed, enabling high throughput and easy separation of the product from the biocatalyst. The enzyme exhibits remarkable stability, maintaining high activity over extended periods, which eliminates the need for frequent catalyst replenishment and reduces operational downtime. Moreover, the process utilizes common industrial reagents such as acetic anhydride and acetic acid for the initial acylation step, ensuring that raw material sourcing remains straightforward and cost-effective. This novel pathway not only boosts the total yield to nearly 90% but also simplifies the purification workflow, making it an ideal candidate for robust commercial scale-up of complex amino acid derivatives.
Mechanistic Insights into D-Acylating Hydrolase Catalyzed Resolution
The core of this innovative synthesis lies in the precise mechanistic action of the D-acylating hydrolase, an enzyme engineered for high stereoselectivity towards N-acetyl-D-tyrosine. The process initiates with the chemical acylation of DL-tyrosine, converting the free amine group into an N-acetyl derivative, which serves as the specific substrate for the enzyme. Once the N-acetyl-DL-tyrosine solution is adjusted to a neutral pH of 7 to 8, it is introduced to the immobilized enzyme column. The D-acylating hydrolase selectively recognizes and hydrolyzes the amide bond of the D-enantiomer precursor, releasing free D-tyrosine while leaving the L-enantiomer precursor untouched in the solution. This kinetic resolution is driven by the enzyme's active site geometry, which perfectly accommodates the D-configuration but sterically hinders the L-configuration. The result is a reaction mixture where the desired D-tyrosine is freely soluble, while the unreacted N-acetyl-L-tyrosine can be separated or recycled. This mechanism ensures that the optical purity of the final product consistently exceeds 99.5% ee, meeting the stringent requirements for pharmaceutical intermediates without the need for extensive chiral chromatography.
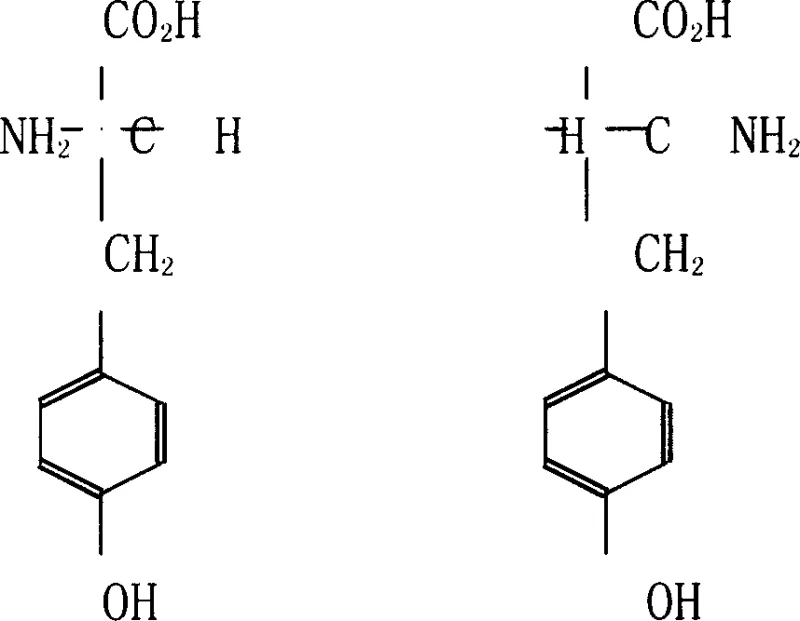
Controlling impurities in this enzymatic process is achieved through a combination of selective catalysis and strategic downstream processing. The high specificity of the D-acylating hydrolase inherently minimizes the formation of unwanted byproducts, as the enzyme does not promote side reactions such as racemization or peptide bond formation under the controlled reaction conditions. Following the enzymatic step, the reaction liquid undergoes a decolorization process using adsorptive agents like activated carbon or diatomaceous earth at elevated temperatures of 80°C to 90°C. This step effectively removes trace organic impurities, colored bodies, and any residual enzyme proteins that might have leached from the column. Subsequent concentration and cooling crystallization further purify the D-tyrosine, as the solubility differences between the product and any remaining impurities allow for the growth of high-purity crystals. The mother liquor, rich in the L-isomer precursor, is not discarded but can be processed to recover L-tyrosine, thereby enhancing the overall atom economy of the synthesis and ensuring a clean impurity profile suitable for regulatory submission.
How to Synthesize D-Tyrosine Efficiently
The synthesis of D-tyrosine via this enzymatic route represents a benchmark for modern biocatalytic manufacturing, combining high efficiency with operational simplicity. The process is designed to be easily adaptable to existing fermentation and chemical processing infrastructure, requiring only standard reactor vessels and filtration equipment. By following the optimized parameters outlined in the patent, such as maintaining the substrate concentration between 0.5 and 1.0 mol/L and controlling the reaction temperature precisely, manufacturers can reproducibly achieve high yields. The detailed标准化 synthesis steps below provide a comprehensive guide for R&D teams looking to implement this technology, covering everything from the initial acylation to the final drying of the crystalline product. Adhering to these protocols ensures that the full potential of the immobilized enzyme is realized, maximizing catalyst lifespan and product quality.
- Perform acylation of DL-tyrosine with acetic anhydride in acetic acid to generate N-acetyl-DL-tyrosine.
- Dissolve the N-acetyl-DL-tyrosine in deionized water, adjust pH to 7-8, and pass through an immobilized D-acylating hydrolase column.
- Elute the reaction mixture, decolorize with activated carbon, concentrate, and crystallize to isolate D-tyrosine with >99.5% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic process offers profound strategic advantages that extend beyond mere technical feasibility. The shift towards a biocatalytic resolution method fundamentally alters the cost structure of D-tyrosine production by replacing expensive chiral reagents with reusable biological catalysts and common bulk chemicals. This transition mitigates the risks associated with the volatility of specialty chemical markets, ensuring a more stable and predictable cost base for long-term contracts. Furthermore, the high yield and optical purity reduce the need for costly rework or secondary purification steps, directly impacting the bottom line by lowering the cost of goods sold. The ability to recover L-tyrosine from the mother liquor adds an additional revenue stream or cost offset, enhancing the overall economic viability of the production facility. These factors collectively position this technology as a superior choice for securing a reliable D-tyrosine supplier capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and the use of immobilized enzymes significantly lower the variable costs associated with each production batch. Since the enzyme column can be reused for extended periods without significant loss of activity, the amortized cost of the biocatalyst per kilogram of product is drastically reduced compared to single-use chemical catalysts. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to further operational savings. The high conversion efficiency means less raw material is wasted, optimizing the utilization of DL-tyrosine feedstock and minimizing waste disposal costs.
- Enhanced Supply Chain Reliability: The reliance on readily available industrial chemicals like acetic anhydride and acetic acid ensures that raw material supply is not a bottleneck, unlike processes dependent on scarce natural extracts. The robustness of the immobilized enzyme system allows for continuous or semi-continuous operation, which enhances production throughput and reduces lead times for order fulfillment. This reliability is crucial for maintaining uninterrupted supply chains for critical drug substances, preventing stockouts that could disrupt downstream pharmaceutical manufacturing. The scalability of the enzyme column technology means that production capacity can be increased linearly by adding more columns, providing flexibility to respond to surges in market demand.
- Scalability and Environmental Compliance: The process is inherently green, operating in aqueous media with minimal organic solvent usage, which simplifies compliance with increasingly stringent environmental regulations. The reduction in hazardous waste generation and the ability to recycle the mother liquor align with sustainability goals, making the facility more attractive to eco-conscious investors and partners. The mild operating conditions also reduce the safety risks associated with high-pressure or high-temperature reactions, lowering insurance premiums and improving workplace safety. This environmental and safety profile facilitates easier permitting for plant expansion, supporting the commercial scale-up of complex amino acid derivatives to meet global needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the enzymatic production of D-tyrosine, based on the detailed specifications provided in the patent literature. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this technology into their supply chain. The answers reflect the proven performance metrics of the immobilized enzyme system and the quality standards achievable through this method. These insights are intended to clarify the operational benefits and technical robustness of the process for potential partners and licensees.
Q: What is the optical purity achievable with this enzymatic process?
A: According to patent CN1900298A, the enzymatic resolution process achieves an optical purity of over 99.5% ee, which is critical for pharmaceutical applications requiring high stereochemical integrity.
Q: How does the immobilized enzyme contribute to cost efficiency?
A: The immobilized D-acylating hydrolase demonstrates excellent stability, retaining 90% activity after 20 days of continuous operation at 45°C, significantly reducing catalyst replacement frequency and operational costs.
Q: Can the mother liquor from this process be utilized?
A: Yes, the crystallization mother liquor containing the unreacted L-isomer can be further processed to recover L-tyrosine, maximizing atom economy and minimizing waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Tyrosine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation therapeutics. Our expertise in biocatalysis and process chemistry allows us to effectively translate laboratory innovations like the enzymatic resolution of D-tyrosine into robust industrial realities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, including the >99.5% ee required for sensitive pharmaceutical applications. By partnering with us, you gain access to a supply chain that is not only reliable but also optimized for cost and quality.
We invite you to collaborate with our technical team to explore how this advanced enzymatic route can enhance your product portfolio and reduce your overall manufacturing costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your sourcing strategy. Let us help you secure a competitive advantage in the market with our premium D-tyrosine solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
