Industrial Scale Production of High-Purity D-Tyrosine via Novel Chemical Resolution Technology
Industrial Scale Production of High-Purity D-Tyrosine via Novel Chemical Resolution Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for the production of non-natural amino acids, particularly D-tyrosine, which serves as a critical chiral building block for a myriad of bioactive compounds. A pivotal advancement in this domain is documented in patent CN1235871C, which outlines a sophisticated yet practical process for preparing D-tyrosine through chemical resolution. Unlike traditional enzymatic approaches that often suffer from substrate specificity limitations and high operational costs, this novel method leverages the differential solubility of diastereomeric salts in pure alcoholic solvents. By employing chiral tartrates as resolving agents, the process achieves high optical purity while maintaining a workflow that is inherently compatible with large-scale manufacturing environments. This technical breakthrough addresses the growing demand for D-tyrosine as a precursor for potent tocolytic agents, antitumor polypeptides, and antimicrobial drugs, offering a reliable pathway for secure supply chains in the competitive API intermediate market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of D-tyrosine from its racemic mixture has been fraught with significant technical and economic hurdles that hindered widespread commercial adoption. Early methodologies, such as those utilizing brucine for resolution, were capable of yielding optically pure products but were plagued by the prohibitive cost and inherent toxicity of the resolving agent, rendering them unsuitable for modern green chemistry standards. Furthermore, enzymatic resolution strategies, while specific, introduce complex operational variables including the need for precise pH control, temperature sensitivity, and the frequent deactivation of biocatalysts, which complicates process continuity. The requirement for multiple derivation and de-derivation steps in enzymatic routes not only extends the production timeline but also accumulates impurities that are difficult to remove, ultimately impacting the quality profile required for high-grade pharmaceutical intermediates. These legacy constraints have created a bottleneck in the supply of D-tyrosine, limiting its application in the synthesis of advanced therapeutic agents despite its recognized pharmacological potential.
The Novel Approach
The methodology described in patent CN1235871C represents a paradigm shift by simplifying the resolution architecture through the strategic use of tartrate salts in anhydrous alcohol systems. This approach eliminates the reliance on expensive biological catalysts or toxic alkaloids, substituting them with cost-effective, chemically stable tartrates that are readily available in bulk quantities. The core innovation lies in the esterification of DL-tyrosine followed by reaction with chiral tartrates to form diastereomeric tyrosine ester tartrate salts, which possess distinct physical properties amenable to separation. By exploiting the solubility differences of these salts in solvents such as methanol, ethanol, or propanol, the process allows for the selective crystallization of the desired isomer with high efficiency. This chemical resolution route not only streamlines the synthetic sequence by combining hydrolysis and resolving agent removal into a unified step but also ensures that the final product meets stringent officinal requirements for specific rotation, thereby establishing a new benchmark for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Tartrate-Mediated Chiral Resolution
The fundamental mechanism driving the success of this synthesis lies in the thermodynamic principles governing diastereomeric salt formation and crystallization. When DL-tyrosine ester reacts with a chiral tartrate resolving agent, such as L-dihydrobutanedioic acid or D-dihydrobutanedioic acid, it generates a pair of diastereomeric salts: L-tyrosine ester tartrate and D-tyrosine ester tartrate. Although these salts share similar chemical connectivity, their three-dimensional spatial arrangements differ significantly, leading to divergent lattice energies and solvation dynamics within the alcoholic medium. In a pure alcohol solvent, one diastereomer exhibits markedly lower solubility than the other, precipitating out of the solution upon cooling or concentration, while the counterpart remains dissolved. This physical separation is the cornerstone of the purification process, allowing for the isolation of the D-isomer salt with exceptional stereochemical fidelity without the need for complex chromatographic techniques.
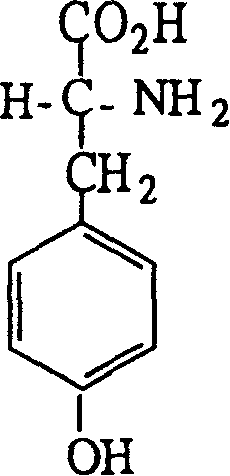
Following the isolation of the diastereomeric salt, the process employs a basic hydrolysis step using sodium hydroxide solution to cleave the ester bond and liberate the free amino acid. Crucially, this hydrolysis step is designed to simultaneously release the resolving agent, which can then be recovered and recycled, enhancing the overall atom economy of the process. The control of impurities is intrinsic to this mechanism; by rigorously controlling the molar ratios of tartrate to tyrosine ester and optimizing the solvent volume, the formation of co-crystals or inclusion complexes is minimized. The resulting D-tyrosine demonstrates a specific rotation of [α]D = +10.6 (c=4, 1N HCl), confirming the high enantiomeric excess achieved through this physicochemical separation strategy. This mechanistic robustness ensures that the final product is suitable for downstream applications in the synthesis of complex chiral drugs, where even trace amounts of the L-enantiomer could compromise therapeutic efficacy or safety profiles.
How to Synthesize D-Tyrosine Efficiently
The practical execution of this synthesis requires precise control over reaction conditions, particularly during the esterification and crystallization phases, to maximize yield and optical purity. The process begins with the conversion of DL-tyrosine into its ester hydrochloride using thionyl chloride in anhydrous alcohol at low temperatures, followed by neutralization to obtain the free ester. Subsequent resolution involves heating the ester with tartrate in alcohol, followed by controlled cooling to induce selective crystallization of the diastereomeric salt. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized protocol below.
- Esterify DL-tyrosine using thionyl chloride in anhydrous alcohol to form DL-tyrosine ester hydrochloride, followed by neutralization.
- React the DL-tyrosine ester with a chiral tartrate resolving agent in pure alcohol to form diastereomeric tyrosine ester tartrate salts.
- Separate the diastereomers based on solubility differences, then hydrolyze the specific salt with alkali to remove the ester group and resolving agent, yielding D-tyrosine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from enzymatic to this chemical resolution process offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of specialized enzymes removes a significant variable from the supply chain, reducing dependency on niche biotech suppliers and mitigating the risk of batch-to-batch variability that often plagues biological processes. Furthermore, the use of commodity chemicals such as tartrates and common alcohols ensures that raw material sourcing is stable and resilient against market fluctuations, providing a predictable cost structure for long-term production planning. This stability is crucial for maintaining continuous manufacturing operations, especially when scaling up to meet the surging demand for D-tyrosine derivatives in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the substitution of high-cost resolving agents with inexpensive tartrates. By avoiding the use of precious metal catalysts or proprietary enzymes, the direct material costs are drastically simplified, leading to substantial cost savings in the overall production budget. Additionally, the ability to recover and recycle the resolving agent after hydrolysis further amplifies these savings, creating a closed-loop system that minimizes waste disposal costs and maximizes resource utilization. This efficiency translates directly into a more competitive pricing structure for the final high-purity D-tyrosine, allowing downstream manufacturers to optimize their own cost of goods sold without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on stable, non-perishable chemical reagents significantly enhances supply chain reliability. Unlike enzymes which require cold chain storage and have limited shelf lives, tartrates and organic solvents can be stored under ambient conditions for extended periods, reducing inventory management complexity. This robustness ensures that production schedules are not disrupted by reagent degradation or delivery delays, facilitating just-in-time manufacturing strategies. Moreover, the simplicity of the reaction setup means that production can be easily transferred between different manufacturing sites or scaled up rapidly in response to market demand, ensuring a consistent and uninterrupted flow of critical intermediates to clients.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard unit operations such as esterification, crystallization, and filtration that are well-understood in industrial chemical engineering. This familiarity reduces the capital expenditure required for equipment modification and accelerates the timeline from pilot plant to commercial scale-up of complex pharmaceutical intermediates. Environmentally, the process aligns with modern sustainability goals by minimizing the use of toxic substances like brucine and reducing the generation of hazardous waste streams. The use of recyclable solvents and the recovery of resolving agents contribute to a lower environmental footprint, helping manufacturers meet increasingly stringent regulatory compliance standards while maintaining operational efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of D-tyrosine via this resolution method. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality attributes of the process for potential partners and technical stakeholders.
Q: What are the advantages of chemical resolution over enzymatic methods for D-tyrosine?
A: Chemical resolution using tartrates avoids the high costs and stability issues associated with specific enzymes. It utilizes inexpensive, widely available resolving agents and standard solvent systems, making it more robust for large-scale industrial production.
Q: How is optical purity achieved in this process?
A: Optical purity is achieved through the formation of diastereomeric salts between the racemic tyrosine ester and chiral tartrate. These salts exhibit distinct solubility profiles in pure alcoholic solvents, allowing for the selective crystallization and isolation of the desired D-isomer salt.
Q: Can the resolving agent be recovered in this synthesis route?
A: Yes, the process is designed for efficiency. The hydrolysis step simultaneously removes the ester group and the resolving agent, facilitating the recovery and potential recycling of the tartrate, which further enhances the economic viability of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Tyrosine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We are committed to delivering D-tyrosine that meets stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify enantiomeric excess and impurity profiles. Our facility is equipped to handle the specific requirements of amino acid resolution, guaranteeing a supply of material that is consistent, reliable, and fully compliant with international pharmaceutical standards.
We invite you to collaborate with us to leverage this advanced resolution technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized process can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-purity D-tyrosine and other complex chiral building blocks in the global market.
