Advanced Synthesis of LCZ696 Intermediates: Overcoming Stereochemical Challenges for Commercial Scale
Advanced Synthesis of LCZ696 Intermediates: Overcoming Stereochemical Challenges for Commercial Scale
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, with LCZ696 (Sacubitril/Valsartan) standing as a cornerstone treatment for heart failure. The efficient production of its key chiral building blocks remains a critical bottleneck for generic manufacturers and innovators alike. Patent CN107304179B, published in late 2020, introduces a transformative synthetic methodology for the preparation of Compound VII, a pivotal intermediate in the LCZ696 value chain. This technology addresses long-standing issues regarding stereochemical control and intermediate stability that have plagued previous generations of synthesis. By leveraging a clever oxazolidine protection strategy coupled with a thermodynamic equilibration step, this route offers a robust pathway to high-purity materials. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable supply of high-purity pharmaceutical intermediates while mitigating the risks associated with complex chiral synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art processes, such as those detailed in WO2008083967 and US20120122844, have historically relied on direct methylation strategies that suffer from significant technical drawbacks. As illustrated in the reaction scheme below, these conventional routes often encounter severe difficulties during the methylation step, typically the fifth step in the sequence. The primary challenge lies in the uncontrollable formation of dimethyl isomers, which drastically reduces the proportion of the desired stereoisomer. Furthermore, the methylated intermediates generated in these older pathways are chemically unstable, leading to decomposition and further compromising the overall yield and purity of the final product. This instability creates a cascade of purification challenges, necessitating expensive chromatographic separations that are ill-suited for cost-effective commercial manufacturing.
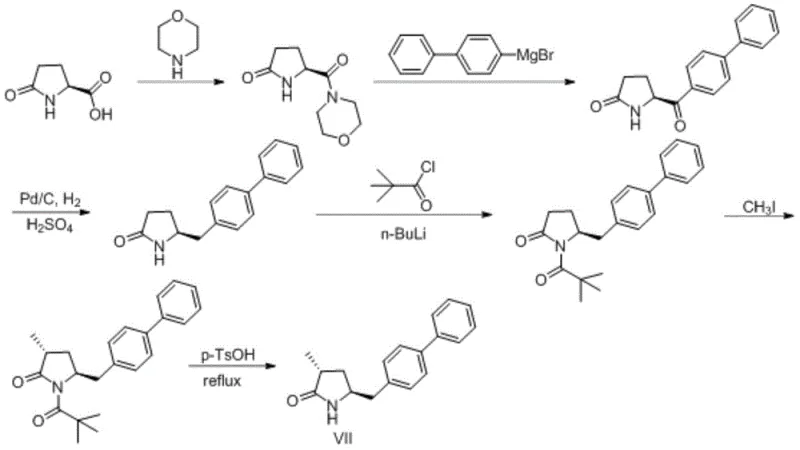
The Novel Approach
In stark contrast to the linear and problematic prior art, the methodology disclosed in CN107304179B employs a strategic detour through an oxazolidine intermediate to exert precise stereochemical control. The new route begins with the conversion of the starting pyrrolidone acid into an ester, followed by reduction to a hydroxyl species. Crucially, this hydroxyl group is then protected via condensation with benzaldehyde to form a rigid oxazolidine ring system. This structural modification not only stabilizes the molecule against degradation but also sets the stage for a highly selective methylation. By shifting the methylation to occur on this protected scaffold, the process avoids the formation of the troublesome dimethyl byproducts seen in conventional methods. The result is a synthetic pathway that is not only chemically more robust but also inherently designed to maximize the utilization rate of the chiral center, making it far superior for industrial application.
Mechanistic Insights into Oxazolidine-Mediated Stereocontrol
The core innovation of this patent lies in the mechanistic elegance of the stereochemical inversion and equilibration steps. After the initial methylation of the oxazolidine intermediate (Compound IV) using a strong base like LDA and a methyl halide, a mixture of stereoisomers (Compounds V and V') is obtained. Rather than attempting a difficult separation at this stage, the process utilizes a subsequent treatment with LDA followed by an aqueous quench. This step induces an epimerization, effectively turning the configuration of the intermediate to favor the thermodynamically more stable isomer (Compound V). This dynamic kinetic resolution approach ensures that even if the initial methylation is not perfectly selective, the subsequent equilibration corrects the stereochemistry, greatly improving the proportion of the required configuration. This mechanism effectively bypasses the purity limitations of direct alkylation, ensuring that the downstream Suzuki coupling proceeds with a substrate of exceptional optical purity.
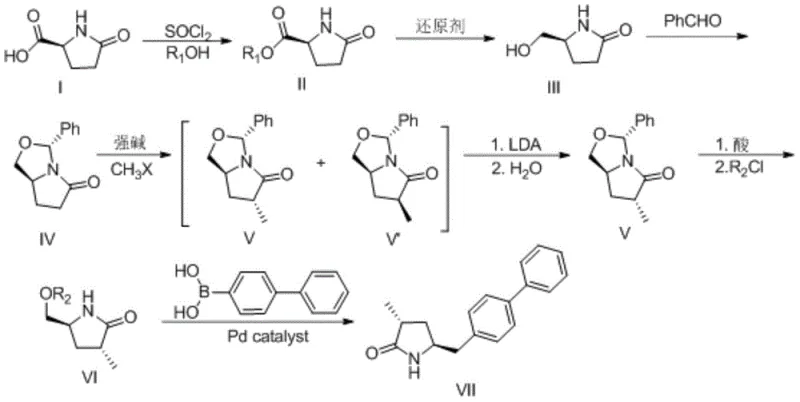
Furthermore, the impurity profile of this process is significantly cleaner due to the stability of the intermediates. In traditional routes, the instability of free amine or unstable alkylated species often leads to polymeric byproducts or degradation products that are difficult to remove. In this novel pathway, the nitrogen atom is tied up in the oxazolidine ring or the lactam, preventing unwanted side reactions during the harsh basic conditions of the methylation. The final steps involve a mild acidic deprotection followed by activation with a sulfonyl chloride (MsCl, TsCl, or TfCl) and a palladium-catalyzed cross-coupling with 4-biphenylboronic acid. The use of standard Suzuki coupling conditions ensures that the final biaryl bond formation is high-yielding and scalable, delivering the target Compound VII with the rigorous purity specifications required for API synthesis.
How to Synthesize LCZ696 Intermediate Efficiently
The synthesis of Compound VII via this patented route involves a sequence of seven distinct chemical transformations, each optimized for yield and operational simplicity. The process begins with the esterification of the starting material using thionyl chloride in an alcohol solvent, followed by a selective reduction using borohydrides to generate the key hydroxyl intermediate. The subsequent protection, methylation, and epimerization steps constitute the heart of the technology, requiring precise temperature control (typically between -80°C and 0°C) to manage the reactivity of the organolithium reagents. Following the stereochemical correction, the molecule is activated and subjected to a palladium-catalyzed coupling to install the biphenyl moiety. For a comprehensive breakdown of the specific reaction conditions, molar ratios, and workup procedures, please refer to the standardized synthesis guide below.
- Esterify the starting pyrrolidone acid with thionyl chloride and alcohol, followed by reduction to the corresponding hydroxyl compound.
- Protect the hydroxyl group with benzaldehyde to form an oxazolidine ring, then perform methylation using LDA and methyl halide.
- Epimerize the methylated mixture using LDA to enrich the desired stereoisomer, followed by deprotection, activation, and final Suzuki coupling with biphenylboronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical superiority. The primary advantage is the substantial reduction in manufacturing costs driven by the elimination of complex purification steps. Because the process inherently corrects stereochemical errors through epimerization, there is no need for costly chiral separations or the discarding of large fractions of unwanted isomers. This leads to a drastic simplification of the production workflow and a significant increase in the overall mass balance of the process. Additionally, the use of commercially available and inexpensive reagents such as benzaldehyde, methyl iodide, and biphenylboronic acid ensures that raw material costs remain low and supply is secure, avoiding the bottlenecks associated with exotic chiral catalysts.
- Cost Reduction in Manufacturing: The process achieves cost efficiency by maximizing the utilization of raw materials through its unique stereochemical correction mechanism. By converting unwanted isomers into the desired product rather than discarding them, the effective yield per kilogram of starting material is significantly enhanced. Furthermore, the stability of the intermediates reduces the loss of material due to decomposition during storage or processing, leading to substantial cost savings in the overall production budget without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood chemical transformations such as esterification, reduction, and Suzuki coupling ensures high process reliability. Unlike routes that depend on sensitive enzymatic steps or unstable intermediates, this chemical synthesis can be performed in standard stainless steel reactors with predictable outcomes. This predictability translates to reduced lead times for high-purity pharmaceutical intermediates, as the risk of batch failure is minimized, ensuring a continuous and dependable supply for downstream API manufacturing.
- Scalability and Environmental Compliance: The synthetic route is explicitly designed to be beneficial for industrial large-scale production, utilizing solvents and reagents that are manageable on a multi-ton scale. The avoidance of heavy metal contaminants (beyond the standard palladium catalyst which is easily scavenged) and the generation of fewer toxic byproducts align with modern green chemistry principles. This facilitates easier regulatory approval and waste management, allowing for the commercial scale-up of complex pharmaceutical intermediates with a reduced environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within patent CN107304179B, providing a clear picture of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing pipelines.
Q: How does this patent address the stereochemical purity issues found in prior art?
A: The patent utilizes an oxazolidine auxiliary strategy that allows for a thermodynamic equilibration step (epimerization) using LDA. This converts unwanted isomers into the desired configuration, significantly improving the proportion of the required stereoisomer compared to direct methylation methods.
Q: What are the key advantages of this route for large-scale manufacturing?
A: The process avoids the formation of unstable methylated intermediates common in older routes. By stabilizing the intermediate as an oxazolidine and using robust reagents like borohydrides and palladium catalysts, the method offers better yield consistency and is highly beneficial for industrial large-scale production.
Q: Can this synthesis method be adapted for different protecting groups?
A: Yes, the patent specifies flexibility in the activation step (Step 6), allowing for the use of MsCl, TsCl, or TfCl. Additionally, the initial esterification can utilize methanol, ethanol, or isopropanol, providing supply chain flexibility for raw material sourcing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable LCZ696 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated stereochemical controls described in this patent can be faithfully reproduced at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of LCZ696 intermediate meets the exacting standards required by top-tier pharmaceutical companies. Our commitment to technical excellence ensures that the transition from lab-scale discovery to commercial reality is seamless and efficient.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and security for your LCZ696 production needs.
