Scalable Synthesis of Sacubitril Key Intermediate Using Cost-Effective Nickel Catalysis
Introduction to Advanced LCZ696 Intermediate Synthesis
The pharmaceutical landscape for cardiovascular treatments has been significantly reshaped by the introduction of LCZ696 (Entresto), a dual-acting angiotensin receptor-neprilysin inhibitor. Central to the supply chain of this blockbuster drug is the efficient production of its key chiral building block, (R)-tert-butyl (1-([1,1'-biphenyl]-4-yl)-3-hydroxypropane-2-yl) carbamate. Patent CN109053495B discloses a groundbreaking synthetic methodology that addresses critical bottlenecks in the manufacturing of this high-value pharmaceutical intermediate. By fundamentally re-engineering the synthetic pathway, this technology replaces prohibitively expensive and toxic reagents with cost-effective, industrially viable alternatives. The innovation lies in a strategic three-step sequence that constructs the complex biphenyl scaffold while maintaining strict stereochemical integrity, offering a robust solution for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
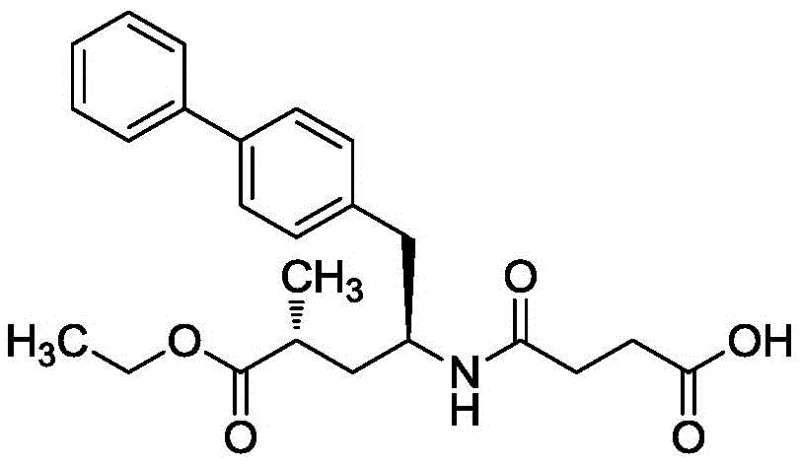
Historically, the synthesis of the Sacubitril fragment has relied on routes that are chemically elegant but economically and environmentally burdensome. As illustrated in prior art literature, such as the route reported in J. Med. Chem. (1995), the construction of the biphenyl moiety often necessitates the use of trifluoromethanesulfonic anhydride (Tf2O). This reagent is not only exceptionally expensive but also highly toxic and corrosive, posing severe safety risks during handling and requiring specialized containment infrastructure. Furthermore, these traditional pathways typically depend on palladium-catalyzed cross-coupling reactions using catalysts like tetrakis(triphenylphosphine)palladium. The reliance on noble metals introduces significant cost volatility and necessitates rigorous downstream purification to remove trace metal residues to meet stringent regulatory limits for API intermediates.
The Novel Approach
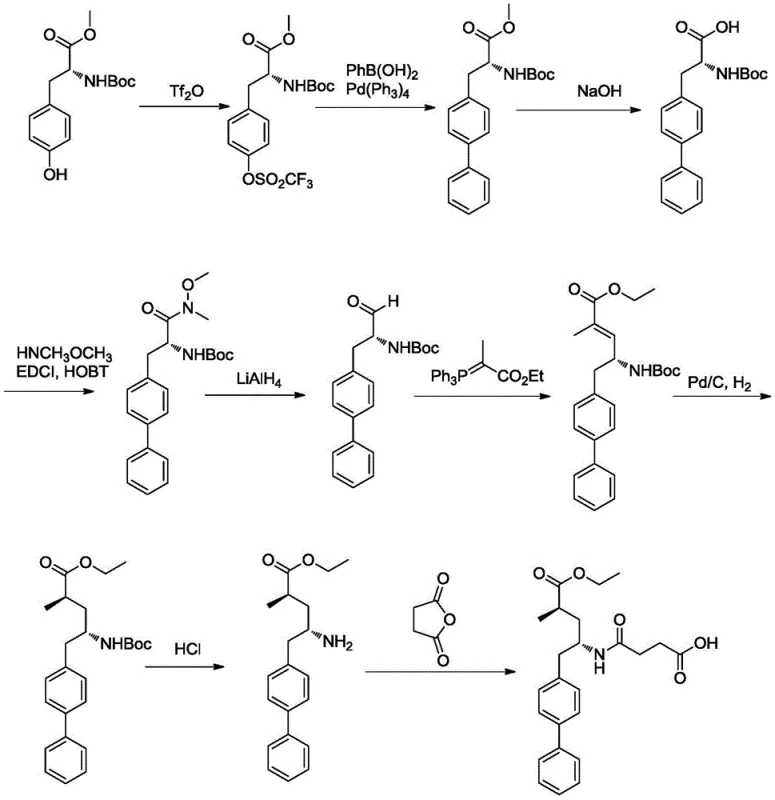
The methodology described in CN109053495B offers a paradigm shift by substituting the problematic triflic anhydride with readily available sulfonyl chlorides, such as p-toluenesulfonyl chloride. This substitution drastically reduces raw material costs and improves operational safety. More critically, the process replaces the palladium catalyst with a nickel-based catalytic system. Nickel is abundant and significantly cheaper than palladium, yet it demonstrates comparable efficacy in facilitating the cross-coupling reaction between the activated tyrosine derivative and the phenyl Grignard reagent. This novel approach streamlines the synthesis into a concise three-step sequence: activation, coupling, and reduction. The elimination of hazardous reagents like lithium aluminum hydride in favor of potassium borohydride further enhances the process profile, making it ideally suited for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Nickel-Catalyzed Cross-Coupling
The core of this technological advancement is the nickel-catalyzed cross-coupling reaction, which effectively constructs the biaryl bond essential for the drug's activity. In this mechanism, a nickel(0) species, generated in situ from a nickel(II) precursor like bis(triphenylphosphine)nickel dichloride, undergoes oxidative addition into the carbon-oxygen bond of the sulfonate intermediate. This step is facilitated by the electron-rich nature of the nickel center and the presence of phosphine ligands. Subsequently, the transmetallation with the phenyl Grignard reagent transfers the phenyl group to the nickel center. The final reductive elimination step releases the coupled biphenyl product and regenerates the active nickel(0) catalyst. This catalytic cycle is highly efficient and operates under mild conditions, typically between -10°C and 50°C, minimizing thermal degradation of the sensitive chiral centers.
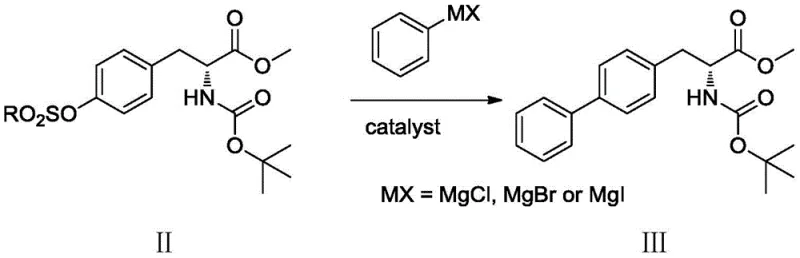
Impurity control is another critical aspect where this mechanism excels. Traditional palladium routes often suffer from homocoupling side reactions or incomplete conversion due to catalyst deactivation. The nickel system described here demonstrates high selectivity, ensuring that the chiral integrity of the amino acid backbone is preserved throughout the coupling process. The subsequent reduction step using potassium borohydride is chemoselective for the ester group, avoiding over-reduction or racemization issues often associated with stronger reducing agents. This precise control over the reaction pathway results in a final product with exceptional optical purity, a prerequisite for high-purity pharmaceutical intermediates intended for human consumption.
How to Synthesize (R)-tert-butyl (1-([1,1'-biphenyl]-4-yl)-3-hydroxypropane-2-yl) carbamate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the activation of BOC-D-tyrosine methyl ester, followed by the pivotal nickel-catalyzed coupling, and concludes with a mild reduction. Each step has been optimized for solvent choice, temperature, and stoichiometry to maximize yield and purity. For detailed operational parameters and specific molar ratios required to replicate these results, please refer to the standardized synthesis guide below.
- React BOC-D-tyrosine methyl ester with p-toluenesulfonyl chloride in the presence of an organic base to form the sulfonate intermediate.
- Perform a nickel-catalyzed cross-coupling reaction between the sulfonate intermediate and a phenyl Grignard reagent to construct the biphenyl skeleton.
- Reduce the resulting ester intermediate using potassium borohydride in a methanol and tetrahydrofuran solvent system to obtain the final chiral amino alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the decoupling of production costs from the volatile noble metal market. By shifting from palladium to nickel, manufacturers can stabilize their cost structures and mitigate the risk of supply disruptions associated with precious metal sourcing. Furthermore, the replacement of triflic anhydride with sulfonyl chlorides simplifies the procurement of raw materials, as these are commodity chemicals available from multiple global suppliers, enhancing supply chain resilience.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost inputs. Palladium catalysts are orders of magnitude more expensive than nickel salts, and their removal requires additional processing steps like scavenging, which adds to the operational expenditure. By eliminating the need for these expensive metals and toxic activators, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the use of potassium borohydride instead of lithium aluminum hydride reduces safety compliance costs and waste disposal fees, contributing to substantial long-term savings without compromising product quality.
- Enhanced Supply Chain Reliability: Reliance on single-source or limited-source suppliers for exotic reagents creates vulnerability. This new method utilizes widely available reagents such as p-toluenesulfonyl chloride and phenyl magnesium bromide, which are produced at scale by the global chemical industry. This abundance ensures consistent availability and shorter lead times for raw materials. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by minor variations in feedstock quality or environmental factors, ensuring a steady flow of high-purity Sacubitril intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling up chemical processes often amplifies safety and environmental challenges. This route is inherently safer due to the absence of pyrophoric reagents and highly corrosive acids. The milder reaction temperatures and the use of common solvents like tetrahydrofuran and methanol facilitate easier heat management in large reactors. From an environmental perspective, the reduction in heavy metal usage simplifies effluent treatment, helping manufacturers meet increasingly strict environmental regulations. This ease of scale-up supports the commercial scale-up of complex pharmaceutical intermediates from kilogram to multi-ton quantities with minimal engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational requirements.
Q: Why is the nickel-catalyzed route preferred over traditional palladium methods for this intermediate?
A: The nickel-catalyzed route significantly reduces raw material costs by replacing expensive palladium catalysts (such as tetrakis(triphenylphosphine)palladium) with inexpensive nickel salts. Additionally, it avoids the use of highly toxic and costly trifluoromethanesulfonic anhydride, utilizing safer sulfonyl chlorides instead, which simplifies waste treatment and improves overall process safety.
Q: What are the purity levels achievable with this synthetic method?
A: According to the patent data, the optimized process yields the final product with high purity, reaching up to 99.2% in specific embodiments. The use of mild reaction conditions and selective reagents like potassium borohydride helps minimize side reactions and impurity formation compared to harsher reducing agents.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It utilizes commercially available starting materials like BOC-D-tyrosine and avoids hazardous reagents like lithium aluminum hydride. The reaction conditions are moderate (temperatures ranging from -10°C to 50°C), making it easier to control heat transfer and safety parameters in large reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sacubitril Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern pharmaceutical supply chain. Our team of expert chemists has thoroughly evaluated the nickel-catalyzed pathway described in CN109053495B and confirmed its potential for industrial excellence. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest global standards.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains. By leveraging our expertise in process chemistry, we can help you achieve significant efficiencies and cost savings. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your next project milestone.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
