Optimizing Balofloxacin Production: A Novel Synthetic Route for 3-Methylaminopiperidine Intermediates
Introduction to Advanced 3-Methylaminopiperidine Manufacturing
The pharmaceutical industry constantly seeks robust and scalable synthetic routes for key active pharmaceutical ingredient (API) intermediates, particularly for fluoroquinolone antibiotics like Balofloxacin. Patent CN1305849C, granted in March 2007, introduces a groundbreaking methodology for the preparation of 3-methylaminopiperidine and its salts, addressing critical bottlenecks in existing manufacturing processes. This technology shifts the synthetic paradigm from complex, low-yield multi-step sequences to a streamlined, three-step protocol starting from commercially abundant 3-aminopyridine. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize supply chains for quinolone antibacterials. By eliminating the reliance on scarce noble metal catalysts and reducing the overall step count, this method not only enhances process efficiency but also aligns with modern green chemistry principles by minimizing waste generation. The following analysis details how this innovation translates into tangible commercial advantages for global pharmaceutical manufacturers seeking reliable partners for complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the technology disclosed in CN1305849C, the industrial synthesis of 3-methylaminopiperidine was plagued by inefficiencies that severely impacted cost structures and supply reliability. The traditional route, often cited in earlier literature, utilized gamma-butyrolactone as the starting material, necessitating a cumbersome eight-step reaction sequence to reach the target piperidine scaffold. This extended synthetic pathway inherently accumulates impurities at each stage, leading to a dismal overall yield of merely 11.5%, which is economically unsustainable for large-scale production. Furthermore, a critical bottleneck in the conventional method was the dependence on platinum oxide (PtO2), a precious metal catalyst that not only drives up raw material costs but also introduces complexities in metal residue removal, a stringent requirement for pharmaceutical grade intermediates. These factors combined to create a high-barrier entry for manufacturers, limiting the availability of high-quality 3-methylaminopiperidine and inflating the cost of goods for downstream antibiotics like Balofloxacin.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent leverages 3-aminopyridine as a strategic starting point, capitalizing on its ready availability and lower cost profile compared to specialized lactones. The new synthetic route condenses the entire process into just three primary chemical transformations: a condensation reaction with an orthoformate reagent, a selective reduction to form the methylamino group, and a final dissolving metal reduction to saturate the pyridine ring. This drastic reduction in step count from eight to three fundamentally alters the economic model of production by minimizing unit operations, solvent consumption, and labor hours. Moreover, the complete elimination of platinum-based catalysts removes a significant cost driver and simplifies the purification workflow, as there is no need for expensive heavy metal scavenging processes. This streamlined methodology offers a robust platform for commercial scale-up, ensuring that manufacturers can meet increasing global demand for fluoroquinolone intermediates with greater agility and cost certainty.
Mechanistic Insights into the Three-Step Reductive Sequence
The core of this technological breakthrough lies in the precise control of nitrogen functionality and ring saturation through a carefully orchestrated sequence of reductions. The first stage involves the reaction of 3-aminopyridine with an orthoformate reagent, such as triethyl orthoformate, under reflux conditions to generate an imino ether intermediate (Formula II). This protection-activation strategy is crucial as it masks the amine functionality while setting up the molecule for subsequent selective reduction. The molar ratio of 3-aminopyridine to orthoformate is optimized between 1:1.5 and 1:10, with a preferred range of 1:4 to 1:7, ensuring high conversion rates while minimizing excess reagent waste. This step effectively transforms the electron-rich aniline-like nitrogen into a more stable imine derivative, which serves as the precursor for the introduction of the methyl group in the subsequent step.
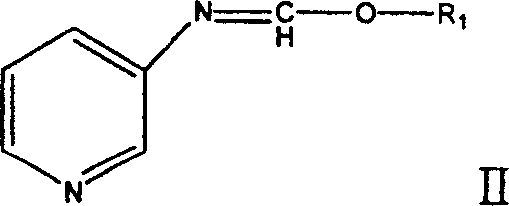
Following the formation of the imino ether, the second stage employs a mild hydride reduction to install the methylamino group, yielding 3-methylaminopyridine (Formula III). This transformation is achieved using borohydride reducing agents, such as sodium borohydride or potassium borohydride, in an alcoholic solvent like ethanol. The choice of potassium borohydride is often preferred for its solubility and reactivity profile in ethanol, allowing the reaction to proceed smoothly from 0°C to reflux temperatures. This step is highly selective, reducing the C=N bond of the imino ether without affecting the aromatic pyridine ring, a feat that requires precise control of reaction conditions to avoid over-reduction or side reactions. The resulting 3-methylaminopyridine is isolated as a colorless liquid with high purity, serving as the critical substrate for the final ring saturation step.
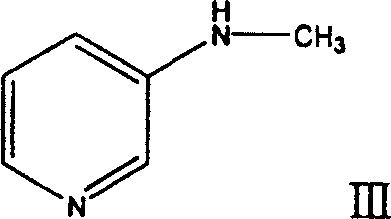
The final and most chemically demanding step is the reduction of the aromatic pyridine ring to the saturated piperidine ring, converting 3-methylaminopyridine into the target 3-methylaminopiperidine. This is accomplished via a dissolving metal reduction using metallic sodium in a higher boiling alcohol solvent, with n-butanol being the preferred medium due to its ability to dissolve both the substrate and the sodium alkoxide by-products. The reaction typically operates with a volume ratio of raw material to alcohol between 1:10 and 1:40, ensuring efficient heat transfer and mixing during the exothermic addition of sodium wire. This Birch-type reduction effectively breaks the aromaticity of the pyridine ring, adding hydrogen across the double bonds to form the piperidine scaffold while preserving the secondary amine functionality. The process concludes with a straightforward acidification step, where dry hydrogen chloride or aqueous hydrochloric acid is introduced to form stable salts like the dihydrochloride, facilitating isolation and purification to pharmaceutical grades exceeding 99% purity.
How to Synthesize 3-Methylaminopiperidine Efficiently
Implementing this synthetic route requires careful attention to reagent quality and thermal management, particularly during the exothermic sodium reduction phase. The process begins with the condensation of 3-aminopyridine and triethyl orthoformate, followed by distillation to isolate the imino ether intermediate. Subsequent reduction with potassium borohydride in ethanol yields the key 3-methylaminopyridine precursor, which is then subjected to the dissolving metal reduction in n-butanol. Detailed operational parameters, including specific temperature ramps, addition rates for metallic sodium, and workup procedures for salt formation, are critical for maximizing yield and safety. For a comprehensive breakdown of the standardized operating procedures and safety protocols required for GMP-compliant manufacturing, please refer to the technical guide below.
- Condense 3-aminopyridine with triethyl orthoformate under reflux to form the imino ether intermediate.
- Reduce the imino ether intermediate using sodium borohydride or potassium borohydride in an alcoholic solvent to yield 3-methylaminopyridine.
- Perform a dissolving metal reduction on 3-methylaminopyridine using metallic sodium in n-butanol to saturate the pyridine ring, followed by salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthetic route described in CN1305849C offers profound strategic benefits that extend beyond simple unit cost savings. The primary advantage lies in the radical simplification of the supply chain; by reducing the synthesis from eight steps to three, the number of potential failure points, quality control checkpoints, and intermediate storage requirements is drastically cut. This compression of the manufacturing timeline directly translates to reduced lead times and enhanced responsiveness to market fluctuations in the demand for Balofloxacin and related antibiotics. Furthermore, the shift away from gamma-butyrolactone and platinum catalysts towards commodity chemicals like 3-aminopyridine and sodium metal mitigates supply risk, as these raw materials are produced on a massive global scale with stable pricing dynamics, insulating the manufacturer from the volatility often associated with specialty fine chemicals and precious metals.
- Cost Reduction in Manufacturing: The economic impact of eliminating the platinum oxide catalyst cannot be overstated, as noble metals represent a significant capital tie-up and require costly recovery or disposal processes to meet regulatory limits on heavy metal residues. By replacing this expensive catalytic step with stoichiometric reductions using inexpensive borohydrides and sodium metal, the variable cost of goods sold is substantially lowered. Additionally, the increase in overall yield from the historical 11.5% to the significantly higher yields reported in the new method (with individual steps achieving 60-85%) means that less raw material is required to produce the same amount of final product, amplifying the cost savings across the entire production volume. This efficiency gain allows for more competitive pricing strategies in the tendering processes for generic antibiotic production.
- Enhanced Supply Chain Reliability: Relying on a shorter synthetic route inherently improves supply continuity, as there are fewer intermediate stages where batch failures or delays can occur, thereby increasing the overall reliability of the production schedule. The use of robust, non-sensitive reagents like metallic sodium and common alcohols ensures that the process is less susceptible to disruptions caused by the shortage of specialized catalysts or complex ligands that often plague modern organic synthesis. This resilience is critical for maintaining the continuous flow of materials required for just-in-time manufacturing models employed by major pharmaceutical companies, ensuring that production lines for critical antibiotics remain operational without interruption due to intermediate shortages.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the new method offers a cleaner profile by reducing the total volume of solvent and reagents consumed per kilogram of product, aligning with increasingly stringent global environmental regulations. The absence of heavy metal catalysts simplifies the waste treatment process, as effluent streams do not require complex remediation to remove platinum traces, reducing the operational burden on wastewater treatment facilities. This streamlined waste profile, combined with the use of standard reactor equipment capable of handling dissolving metal reductions, facilitates easier scale-up from pilot plant to multi-ton commercial production, enabling manufacturers to rapidly expand capacity to meet surging market demand without requiring significant capital investment in specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis for 3-methylaminopiperidine. These answers are derived directly from the experimental data and claims presented in patent CN1305849C, providing a factual basis for evaluating the technology's feasibility for your specific manufacturing needs. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement officers assessing supplier capabilities.
Q: Why is the new synthetic route for 3-methylaminopiperidine more cost-effective than conventional methods?
A: The conventional method starting from gamma-butyrolactone requires eight reaction steps and utilizes expensive platinum oxide (PtO2) catalysts, resulting in a low total yield of approximately 11.5%. The novel route described in patent CN1305849C reduces the process to just three steps, eliminates the need for noble metal catalysts, and uses readily available raw materials like 3-aminopyridine, drastically lowering production costs.
Q: What are the critical reaction conditions for the ring reduction step?
A: The critical ring reduction step involves treating 3-methylaminopyridine with metallic sodium in an alcoholic solvent. The patent specifies that n-butanol is the preferred solvent, with a volume ratio of raw material to alcohol ranging from 1:10 to 1:40. The reaction typically proceeds under reflux conditions to ensure complete saturation of the pyridine ring to form the piperidine structure.
Q: How does this synthesis method impact the purity of the final API intermediate?
A: By shortening the synthetic sequence from eight steps to three, the accumulation of impurities and by-products is significantly minimized. The use of specific reducing agents like potassium borohydride for the methylation step and controlled dissolving metal reduction allows for better selectivity. Experimental data in the patent indicates that the final product can achieve high purity levels, such as 99.7% by GC analysis, which is crucial for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylaminopiperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of final pharmaceutical products like Balofloxacin. Our technical team has extensively analyzed the route disclosed in CN1305849C and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient synthesis to life. We are committed to delivering 3-methylaminopiperidine with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for API synthesis, free from the heavy metal contaminants associated with older platinum-catalyzed methods.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized production of 3-methylaminopiperidine can enhance your operational efficiency and reduce your overall cost of goods.
