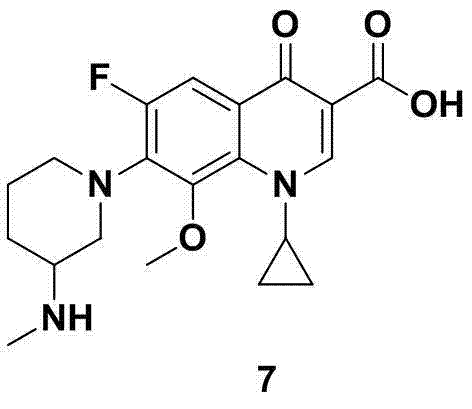
Optimizing Balofloxacin Intermediate Production via Advanced Catalytic Hydrogenation
Optimizing Balofloxacin Intermediate Production via Advanced Catalytic Hydrogenation
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical antibiotic intermediates, particularly for fluoroquinolones like Balofloxacin. Patent CN103113290A introduces a refined methodology for synthesizing 3-methylaminopiperidine, a pivotal building block in the manufacture of this potent antibacterial agent. This innovation addresses longstanding challenges associated with catalyst costs and reaction severity, proposing a streamlined three-step sequence that begins with 3-methylaminopyridine. By shifting from traditional platinum-based catalysts to a more economical palladium system and utilizing acetic acid as a recyclable solvent, this process demonstrates significant potential for industrial adoption. The strategic implementation of a protective group prior to ring saturation ensures high chemical fidelity, ultimately delivering the target amine with exceptional purity suitable for stringent pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct catalytic hydrogenation of 3-methylaminopyridine to form 3-methylaminopiperidine has been plagued by economic and technical inefficiencies. Prior art methodologies often relied on platinum dioxide (PtO2) as the primary catalyst, a precious metal reagent that imposes a substantial financial burden on large-scale manufacturing operations. Furthermore, literature precedents indicate that achieving acceptable conversion rates frequently required extreme reaction conditions, such as pressures reaching 50 atm and temperatures around 70°C in ethanol, which yielded only moderate results of approximately 78%. Alternative approaches using palladium carbon in acetic acid under high pressure showed even lower yields, sometimes dropping to 46%, rendering them commercially unviable. These conventional pathways also suffered from a lack of reported catalyst recycling protocols, leading to increased waste generation and higher overall production costs due to the single-use nature of expensive catalytic materials.
The Novel Approach
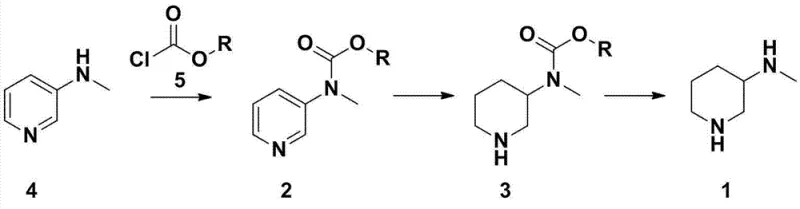
In stark contrast to these inefficient legacy processes, the novel approach detailed in the patent employs a protective group strategy that fundamentally alters the reaction landscape. Instead of subjecting the free amine directly to hydrogenation, the process first converts 3-methylaminopyridine into a carbamate derivative, specifically ethyl 3-pyridinemethylcarbamate. This modification allows for the use of 5% palladium on carbon (Pd/C), a significantly cheaper and more accessible catalyst than platinum dioxide. The reaction proceeds in glacial acetic acid, a solvent chosen for its superior ability to dissolve intermediates and enhance catalytic efficiency compared to alcohols. By optimizing the temperature to a range of 90-95°C and maintaining a hydrogen pressure of 2 MPa, the method achieves a combined two-step yield exceeding 73%, with some embodiments reporting up to 85.9%. This shift not only lowers raw material costs but also simplifies the downstream processing through improved impurity profiles.
Mechanistic Insights into Protective Group Assisted Hydrogenation
The core mechanistic advantage of this synthesis lies in the temporary masking of the secondary amine functionality. In the initial step, 3-methylaminopyridine reacts with ethyl chloroformate or di-tert-butyl dicarbonate under alkaline conditions to form a urethane or carbamate linkage. This electron-withdrawing group reduces the nucleophilicity of the nitrogen atom, thereby preventing potential catalyst poisoning or unwanted side reactions that often occur when free amines interact with transition metal surfaces during hydrogenation. The resulting protected pyridine derivative is then subjected to catalytic reduction. The presence of the carbamate group appears to facilitate a smoother addition of hydrogen across the aromatic ring, likely by modulating the electronic density of the pyridine system, making it more susceptible to reduction under the specified mild conditions. This controlled environment ensures that the stereochemistry and structural integrity of the piperidine ring are maintained without degradation.
Following the saturation of the heterocyclic ring, the final mechanistic step involves the cleavage of the protecting group to regenerate the free amine. This is achieved through acid hydrolysis using concentrated hydrochloric acid or sulfuric acid at elevated temperatures. The robustness of the carbamate bond allows it to withstand the hydrogenation conditions but succumb readily to acidic hydrolysis, providing a clean switch to release the final 3-methylaminopiperidine product. This sequential logic—protect, reduce, deprotect—minimizes the formation of polymeric byproducts and over-reduced species that typically contaminate direct hydrogenation attempts. The result is a crude product that requires minimal purification, often yielding a final hydrochloride salt with gas chromatography purity exceeding 97%, which is critical for meeting the rigorous quality standards of active pharmaceutical ingredient (API) manufacturing.
How to Synthesize 3-Methylaminopiperidine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing safety and reproducibility. The process begins with the careful addition of ethyl chloroformate to a cooled solution of 3-methylaminopyridine and a base like triethylamine to manage the exothermic nature of the substitution reaction. Once the protected intermediate is isolated, it is dissolved in acetic acid and charged with 5% Pd/C catalyst. The hydrogenation vessel must be thoroughly purged with nitrogen before introducing hydrogen to prevent explosive mixtures, operating at a controlled pressure of 2 MPa and temperature of 90-95°C for approximately 40 hours.
- React 3-methylaminopyridine with ethyl chloroformate under alkaline conditions to form the protected carbamate derivative.
- Perform catalytic hydrogenation of the protected pyridine derivative using 5% Pd/C in acetic acid solvent at elevated temperature and pressure.
- Hydrolyze the protected piperidine intermediate using concentrated hydrochloric acid to remove the protecting group and isolate the final amine salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented methodology represents a strategic opportunity to optimize the cost structure of fluoroquinolone production. The replacement of platinum dioxide with palladium on carbon eliminates the dependency on one of the most expensive precious metals in the periodic table, directly translating to substantial cost reduction in pharmaceutical intermediate manufacturing. Additionally, the ability to recycle the acetic acid solvent and potentially recover the palladium catalyst further enhances the economic viability of the process, reducing the volume of hazardous waste requiring disposal. These factors collectively contribute to a more sustainable and budget-friendly supply chain model that is less susceptible to volatile fluctuations in precious metal markets.
- Cost Reduction in Manufacturing: The substitution of high-cost platinum catalysts with economical palladium on carbon significantly lowers the bill of materials for every batch produced. Furthermore, the use of acetic acid, a commodity chemical with a well-established global supply chain, replaces more specialized or less efficient solvents, driving down operational expenditures. The high yield and purity achieved reduce the need for extensive downstream purification steps, such as repeated recrystallizations or chromatography, which are often resource-intensive and time-consuming in a commercial setting.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents like ethyl chloroformate and standard hydrogenation catalysts, manufacturers can mitigate the risk of supply disruptions associated with niche or proprietary reagents. The robustness of the reaction conditions allows for flexibility in sourcing raw materials, as the process tolerates standard industrial grades of solvents and bases. This resilience ensures consistent production schedules and reliable delivery timelines for downstream API manufacturers, safeguarding against bottlenecks that could delay the availability of critical antibiotics.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction parameters that are easily transferable from laboratory glassware to industrial stainless steel reactors. The moderate pressure requirements (2 MPa) are well within the safety limits of standard hydrogenation equipment, avoiding the need for specialized high-pressure vessels that would require costly certifications and maintenance. Moreover, the recyclability of the solvent and the potential for catalyst recovery align with modern green chemistry principles, helping companies meet increasingly stringent environmental regulations regarding waste discharge and resource consumption.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this technology for adoption. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing production lines.
Q: Why is Pd/C preferred over PtO2 for this hydrogenation?
A: Palladium on carbon (Pd/C) is significantly more cost-effective than platinum dioxide (PtO2) and offers comparable or superior catalytic activity under the optimized conditions described in the patent, facilitating easier catalyst recovery and recycling.
Q: What is the role of the carbamate protecting group?
A: The carbamate protecting group stabilizes the amine functionality during the harsh hydrogenation conditions, preventing side reactions and ensuring high regioselectivity and purity of the final 3-methylaminopiperidine product.
Q: Can the acetic acid solvent be recycled?
A: Yes, the patent explicitly highlights that acetic acid serves as an ideal solvent not only for its reaction efficiency but also because it is inexpensive and can be easily recovered and recycled, reducing waste and operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylaminopiperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful formulation of life-saving antibiotics like Balofloxacin. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-methylaminopiperidine meets the exacting standards required by global regulatory bodies. Our commitment to excellence extends beyond mere compliance; we actively collaborate with clients to optimize processes for maximum yield and minimal environmental impact.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes. By leveraging our expertise in catalytic hydrogenation and protective group chemistry, we can help you realize the full economic potential of this patented route. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can strengthen your supply chain and drive your project forward.
