Advanced Synthetic Routes for High-Purity Simvastatin: Commercial Scalability and Impurity Control
The pharmaceutical landscape for cardiovascular therapeutics continues to demand higher standards of purity and process efficiency, particularly for blockbuster drugs like simvastatin. Patent CN1446088A introduces a transformative methodology for preparing semi-synthetic statins, specifically focusing on the production of highly purified simvastatin compositions. This technology addresses the critical industry challenge of controlling trace impurities such as simvastatin oxolactone, dehydrated simvastatin, and dimers, which are often difficult to eliminate using conventional esterification routes. By shifting the synthetic strategy to involve stable amide intermediates and strategic protection of the 1,3-diol system, this process enables the production of simvastatin with impurity levels consistently below 0.1% by weight. For R&D directors and procurement specialists, understanding this mechanistic shift is vital for securing a reliable simvastatin supplier capable of meeting stringent regulatory specifications while optimizing manufacturing costs.
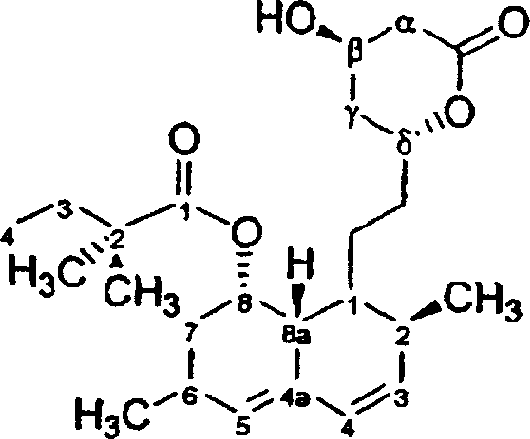
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for converting lovastatin to simvastatin often rely on direct C-methylation or ester hydrolysis followed by re-acylation, which present significant chemical engineering hurdles. These conventional pathways frequently suffer from poor selectivity, leading to the formation of stubborn byproducts like dehydrated simvastatin and simvastatin dimers that co-elute during purification. The inherent instability of the lactone ring under basic hydrolysis conditions often necessitates harsh reaction environments that degrade the hexahydronaphthalene core, resulting in lower overall yields and complex waste streams. Furthermore, the direct manipulation of the 2-methylbutyryl side chain without adequate protection of the neighboring hydroxyl groups can lead to scrambling and incomplete conversion, requiring expensive chromatographic separations that are not feasible for commercial scale-up of complex statin intermediates.
The Novel Approach
The innovative process disclosed in CN1446088A circumvents these pitfalls by introducing a robust amide intermediate strategy that stabilizes the molecule during the critical side-chain exchange phase. Instead of struggling with labile ester bonds, the method first opens the lactone ring using amines to form a chemically resilient amide, which serves as a sturdy scaffold for subsequent modifications. This approach allows for the selective protection of the 1,3-diol moiety using ketals or cyclic esters, effectively shielding the sensitive alcohol groups from unwanted side reactions during the removal of the original side chain. By decoupling the side-chain modification from the lactone integrity, the process achieves a level of control that drastically simplifies downstream purification, offering a clear pathway for cost reduction in API manufacturing through improved yield and reduced solvent consumption.
Mechanistic Insights into Amide-Mediated Statin Synthesis
The core of this technological advancement lies in the precise orchestration of functional group transformations, beginning with the nucleophilic attack of an amine on the lactone carbonyl to generate a hydroxy-amide species. This step is crucial as it converts the cyclic ester into an acyclic amide, preventing premature ring closure and providing a handle for solubility adjustments. Following this, the 1,3-diol system is masked using protecting groups such as acetones or cyclic borates, which are selected for their stability under the reducing conditions required for side-chain removal. The removal of the 2-methylbutyryl group is then executed using reducing agents like lithium aluminum hydride or organometallic reagents, a step that would be catastrophic on an unprotected lactone but proceeds cleanly on the protected amide intermediate. This sequence ensures that the hexahydronaphthalene ring system remains intact while the C-8 position is prepared for the installation of the pharmacologically essential 2,2-dimethylbutyrate side chain.
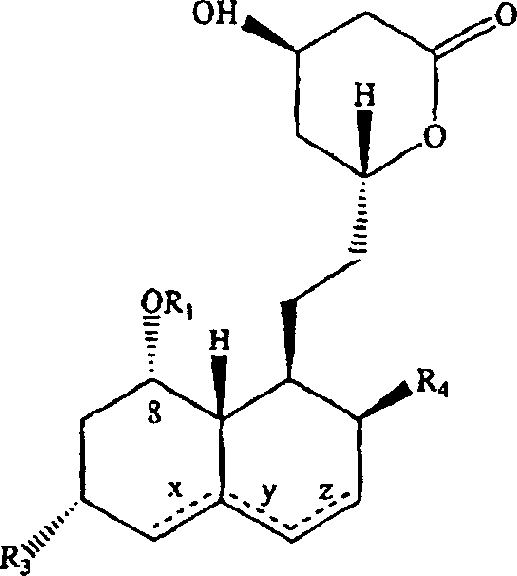
Following the installation of the new side chain via acylation with 2,2-dimethylbutyryl chloride or anhydrides, the molecule undergoes a carefully controlled deprotection and cyclization sequence. The removal of the protecting group restores the free hydroxyls necessary for biological activity, while the conversion of the amide back to a carboxylic acid salt sets the stage for the final lactonization. This final ring-closing step is performed under mild acidic conditions that favor the formation of the thermodynamically stable six-membered lactone ring found in simvastatin. The mechanistic elegance of this route lies in its ability to isolate and purify intermediates at multiple stages, effectively filtering out impurities before they can propagate to the final API. This multi-stage purification capability is what allows the process to achieve the claimed purity levels of less than 0.1% for critical impurities, a specification that is increasingly demanded by global regulatory bodies for cardiovascular medications.
How to Synthesize Simvastatin Efficiently
The synthesis of high-purity simvastatin via this patented route involves a series of well-defined unit operations that transform readily available lovastatin into the desired dimethylated product. The process begins with the amidation of the lactone ring, followed by protection, side-chain exchange, and final cyclization, each step optimized for maximum recovery and minimal byproduct formation. While the general chemical transformations are outlined above, the specific operational parameters such as temperature controls, solvent choices, and stoichiometric ratios are critical for reproducing the high purity results described in the patent literature. Detailed standard operating procedures for each reaction stage, including workup and isolation techniques, are essential for transferring this laboratory-scale success to industrial production environments.
- React lovastatin with an amine to open the lactone ring and form a stable amide intermediate.
- Protect the 1,3-diol moiety using protecting groups such as ketals, acetals, or cyclic esters.
- Remove the original 2-methylbutyryl side chain via reduction or organometallic reaction.
- Attach the new 2,2-dimethylbutyrate side chain through acylation at the 8-position hydroxyl.
- Deprotect the diol, convert the amide to an acid salt, and perform lactone ring closure to finalize simvastatin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this amide-mediated synthesis route offers substantial strategic advantages beyond mere chemical elegance. The ability to utilize robust intermediates that can be crystallized and purified before the final steps significantly de-risks the manufacturing process, ensuring consistent batch-to-batch quality and reducing the likelihood of costly production failures. This reliability translates directly into enhanced supply chain continuity, as the process is less susceptible to the variability often seen in direct fermentation or less controlled semi-synthetic routes. Moreover, the flexibility to use starting materials with higher impurity profiles without compromising the final product quality provides a buffer against raw material price fluctuations, contributing to long-term cost stability.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps, which are often required in traditional routes to remove closely related impurities, leads to significant operational savings. By relying on crystallization and extraction for purification, the process reduces solvent usage and waste disposal costs, aligning with green chemistry principles while lowering the overall cost of goods sold. The high yield of the amide formation and subsequent steps further amplifies these savings by maximizing the throughput of the existing reactor infrastructure.
- Enhanced Supply Chain Reliability: The robustness of the amide intermediates allows for extended storage and transportation if necessary, providing greater flexibility in production scheduling and inventory management. This stability ensures that supply disruptions at one stage of the synthesis do not necessarily halt the entire production line, as intermediates can be stockpiled safely. Additionally, the use of common reagents and solvents reduces dependency on specialized or scarce chemicals, mitigating supply risk and ensuring a steady flow of materials for continuous manufacturing.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plants to multi-ton commercial reactors without significant re-engineering. The reduction in hazardous waste generation, particularly through the avoidance of heavy metal catalysts or excessive chlorinated solvents in certain steps, simplifies environmental compliance and permitting. This eco-friendly profile not only reduces regulatory burden but also enhances the corporate sustainability metrics of the manufacturing entity, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity simvastatin synthesis technology. These answers are derived from the specific embodiments and claims within the patent data, providing clarity on the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production needs or for sourcing partners who utilize similar methodologies.
Q: What is the primary advantage of the amide intermediate route in simvastatin synthesis?
A: The amide intermediate route offers superior stability compared to direct ester manipulation, allowing for rigorous purification steps before the final lactonization, which significantly reduces critical impurities like simvastatin oxolactone and dimers.
Q: How does this process control specific impurities below 0.1%?
A: By employing specific protecting groups for the 1,3-diol moiety and utilizing controlled reduction conditions for side-chain removal, the process minimizes dehydration and dimerization side reactions, ensuring impurity levels remain below 0.1% by weight.
Q: Can this synthesis method utilize lower grade lovastatin starting materials?
A: Yes, the robustness of the amide formation and subsequent purification steps allows the process to tolerate starting materials with higher impurity profiles, potentially reducing raw material costs without compromising final API quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Simvastatin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and state-of-the-art facilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate steps of the amide-mediated simvastatin synthesis are executed with precision and consistency. We maintain stringent purity specifications across all our batches, supported by rigorous QC labs equipped with advanced analytical instrumentation to detect impurities at the ppm level. Our commitment to quality assurance means that every kilogram of simvastatin we produce meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized processes can reduce your overall procurement costs while enhancing product quality. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless integration of our high-purity intermediates into your final drug formulations.
