Advanced Transimidation Technology for Commercial Scale Production of High-Purity Substituted Phthalic Anhydrides
The chemical industry is constantly evolving, driven by the need for more efficient and sustainable synthetic pathways for critical intermediates. Patent CN1329360C introduces a groundbreaking methodology for the preparation of substituted phthalic anhydrides, specifically targeting the production of high-value compounds like 4-chlorophthalic anhydride. This innovation addresses long-standing challenges in the synthesis of polyetherimide precursors by utilizing a novel transimidation reaction between substituted N-alkylphthalimides and substituted tetrahydrophthalic anhydrides. Unlike traditional methods that often struggle with isomer separation and low yields, this approach offers a streamlined route that enhances purity and operational feasibility. For R&D directors and procurement specialists, understanding the nuances of this patent is crucial for optimizing supply chains and reducing manufacturing costs in the production of advanced engineering plastics and pharmaceutical intermediates. 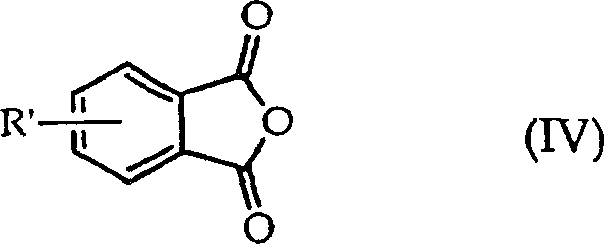
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-chlorophthalic anhydride has relied heavily on Diels-Alder condensation reactions involving maleic anhydride and chloroprene, followed by complex aromatization steps. These conventional routes are fraught with significant technical hurdles, primarily the formation of intricate mixtures of isomers that are notoriously difficult and expensive to separate. The necessity for subsequent recovery of hydrogen bromide in certain variations adds another layer of complexity and safety concern to the process. Furthermore, thermal aromatization attempts often result in low yields and the formation of undesirable tars, which complicate purification and reduce overall process efficiency. For large-scale manufacturers, these inefficiencies translate directly into higher operational expenditures and inconsistent product quality, making the search for alternative synthetic strategies a top priority for maintaining competitiveness in the global market.
The Novel Approach
The innovative method disclosed in the patent circumvents these issues by employing an imide exchange reaction, fundamentally altering the synthetic landscape for these valuable anhydrides. By reacting a substituted N-alkylphthalimide with a substituted tetrahydrophthalic anhydride, the process achieves the desired transformation with greater specificity and control. This route eliminates the need for harsh halogenation conditions and minimizes the generation of isomeric by-products that plague older methodologies. The ability to conduct this reaction in both batch and continuous modes provides exceptional flexibility for manufacturing operations, allowing producers to adapt quickly to fluctuating market demands. Moreover, the inherent design of this chemistry facilitates the recycling of by-products, further enhancing the economic and environmental profile of the synthesis, making it an attractive option for sustainable chemical manufacturing.
Mechanistic Insights into Transimidation-Based Anhydride Synthesis
At the heart of this technology lies the transimidation mechanism, a sophisticated chemical exchange that swaps the imide functionality between two distinct molecular frameworks. The reaction initiates when the nitrogen atom of the N-alkylphthalimide attacks the carbonyl carbon of the tetrahydrophthalic anhydride, leading to a transient intermediate that eventually collapses to release the target substituted phthalic anhydride. This mechanistic pathway is highly favorable because it leverages the thermodynamic stability of the aromatic phthalic anhydride product to drive the equilibrium forward. Understanding this mechanism is vital for process chemists aiming to optimize reaction conditions, such as temperature and solvent choice, to maximize conversion rates. The precision of this exchange ensures that the resulting product possesses the exact substitution pattern required for downstream applications, such as the synthesis of high-performance polyetherimides, without the contamination of structural isomers. 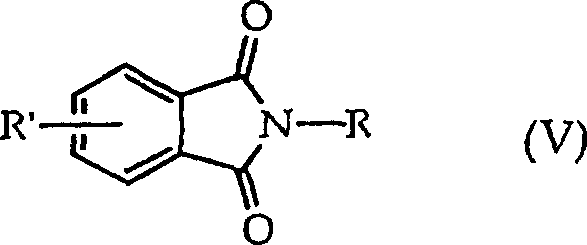
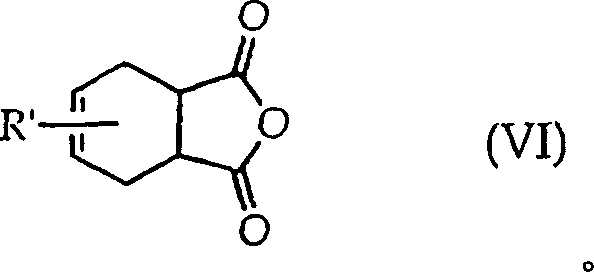
Impurity control is another critical aspect where this mechanism excels, particularly through the management of the N-alkyltetrahydrophthalimide by-product. Instead of discarding this side product, the process allows for its conversion back into the starting N-alkylphthalimide via a subsequent aromatization step. This closed-loop capability significantly reduces waste and improves the overall atom economy of the synthesis. By passing the by-product over a transition metal catalyst like vanadium pentoxide at elevated temperatures, manufacturers can recover valuable starting materials, thereby lowering the net consumption of raw chemicals. This level of impurity management not only enhances the purity profile of the final anhydride but also aligns with stringent environmental regulations, providing a dual benefit of cost savings and regulatory compliance for forward-thinking chemical enterprises.
How to Synthesize 4-Chlorophthalic Anhydride Efficiently
Executing this synthesis requires careful attention to reaction parameters to ensure optimal yield and purity. The process begins with the preparation of the key starting materials, specifically the N-alkyl-4-chlorophthalimide and 4-chlorotetrahydrophthalic anhydride, which are combined in a reactor under controlled conditions. The reaction mixture is then heated to facilitate the imide exchange, typically in the presence of a base or specific solvent system to promote kinetics. Following the reaction, the crude product undergoes separation, often involving extraction and distillation steps to isolate the pure 4-chlorophthalic anhydride from unreacted starting materials and by-products. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining substituted N-alkylphthalimide and substituted tetrahydrophthalic anhydride in a suitable solvent system under inert atmosphere.
- Heat the reaction mixture to facilitate the imide exchange reaction, ensuring precise temperature control to maximize conversion rates while minimizing side reactions.
- Isolate the target substituted phthalic anhydride through distillation or crystallization, and optionally recycle the by-product N-alkyltetrahydrophthalimide via aromatization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this transimidation technology represents a strategic opportunity to enhance operational resilience and cost efficiency. By shifting away from complex multi-step syntheses that rely on hazardous reagents and generate difficult waste streams, companies can streamline their production workflows. This simplification reduces the burden on waste treatment facilities and lowers the risk of production delays caused by purification bottlenecks. Furthermore, the ability to recycle by-products internally creates a more self-sufficient manufacturing loop, reducing dependency on external raw material suppliers and mitigating the impact of market volatility on input costs. These factors collectively contribute to a more robust and predictable supply chain, essential for meeting the rigorous delivery schedules of downstream customers in the pharmaceutical and polymer industries.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous halogenation steps, coupled with the ability to recycle key intermediates, leads to substantial cost savings in the overall manufacturing process. By avoiding the formation of hard-to-separate isomers, the need for extensive and energy-intensive purification processes is drastically reduced, lowering utility consumption and labor costs associated with quality control. Additionally, the higher selectivity of the transimidation reaction means less raw material is wasted on off-spec products, directly improving the yield per batch and maximizing the return on investment for every kilogram of feedstock purchased. These cumulative efficiencies translate into a significantly lower cost of goods sold, providing a competitive pricing advantage in the marketplace.
- Enhanced Supply Chain Reliability: The simplified nature of this synthetic route enhances supply chain reliability by reducing the number of critical processing steps that could potentially fail or cause delays. With fewer unit operations and less reliance on specialized reagents that may have long lead times, manufacturers can maintain more consistent production schedules. The flexibility to operate in continuous mode further supports just-in-time manufacturing strategies, allowing for rapid response to sudden spikes in demand from key accounts. This agility ensures that customers receive their orders on time, fostering stronger long-term partnerships and reducing the risk of stockouts that can disrupt downstream production lines for critical applications like medical devices or aerospace components.
- Scalability and Environmental Compliance: From a scalability perspective, the process is designed to transition smoothly from laboratory benchtop to industrial-scale reactors without significant re-engineering. The reaction conditions are moderate and manageable, reducing the engineering challenges associated with heat transfer and mixing in large vessels. Environmentally, the reduction in hazardous waste generation and the ability to recycle solvents and catalysts align perfectly with modern green chemistry principles. This compliance not only minimizes regulatory risks but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with environmentally conscious multinational corporations. The combination of scalability and eco-friendliness makes this technology a future-proof solution for long-term growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from a detailed analysis of the patent specifications and are intended to provide clarity on the practical aspects of adopting this synthesis route. Understanding these details helps stakeholders make informed decisions about integrating this method into their existing production portfolios. The answers reflect the specific advantages and operational parameters outlined in the intellectual property documentation.
Q: What is the primary advantage of the transimidation method over traditional Diels-Alder routes?
A: The transimidation method avoids the formation of difficult-to-separate isomer mixtures common in Diels-Alder condensations, significantly simplifying downstream purification and reducing overall processing time.
Q: Can the by-products of this reaction be recycled to improve atom economy?
A: Yes, the process allows for the aromatization of the N-alkyltetrahydrophthalimide by-product back into the starting N-alkylphthalimide, creating a closed-loop system that enhances material efficiency.
Q: Is this synthesis route scalable for industrial production of polyetherimide precursors?
A: The patent data indicates that the reaction can be conducted in both batch and continuous modes, demonstrating strong potential for commercial scale-up to meet the demands of engineering plastic manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chlorophthalic Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic routes like the transimidation process described in CN1329360C for producing high-quality 4-chlorophthalic anhydride. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to full-scale manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical and high-performance polymer applications. We are committed to leveraging our technical expertise to optimize this specific chemistry for your unique needs, delivering consistent quality and reliability.
We invite you to collaborate with us to explore how this innovative synthesis can drive value for your organization. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that validate the commercial viability of this approach. Contact us today to initiate a discussion on optimizing your supply chain for substituted phthalic anhydrides and securing a competitive edge in your market sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
