Advanced Synthesis of Valnemulin Hydrochloride for Industrial Veterinary Applications
Advanced Synthesis of Valnemulin Hydrochloride for Industrial Veterinary Applications
The pharmaceutical landscape for veterinary antibiotics is constantly evolving, driven by the urgent need for high-efficacy, low-residue treatments such as Valnemulin. Patent CN101597248B introduces a groundbreaking preparation method for Valnemulin and its hydrochloride salt that addresses critical limitations in existing manufacturing technologies. This innovation focuses on a streamlined synthetic route that utilizes D-hydroxy valine Dane salt and a specific pleuromutilin derivative under remarkably mild conditions. By shifting away from harsh reaction environments and complex purification protocols, this technology offers a robust pathway for producing high-purity intermediates essential for modern animal health formulations. The method ensures that the final product achieves a purity level exceeding 97% via HPLC analysis without the need for column chromatography, representing a significant leap forward in process efficiency and product quality control for reliable veterinary drug intermediate suppliers.
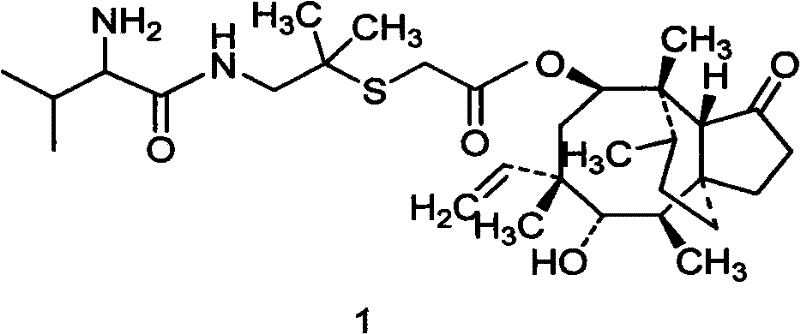
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Valnemulin has been plagued by inefficiencies that hinder large-scale production and economic viability. As illustrated in earlier patents such as US4675330, traditional routes often relied on the use of protected amino acid derivatives and required noble metal catalysts like Palladium on charcoal for hydrogenation steps. These conventional processes typically necessitated room temperature reactions extending over several hours, followed by tedious deprotection steps in methanol systems. The resulting crude product frequently exhibited poor purity profiles, mandating the use of column chromatography for purification. This reliance on chromatographic separation is a major bottleneck in industrial chemistry, as it drastically increases processing time, solvent consumption, and overall operational costs, making it unsuitable for the high-volume demands of the global livestock industry.
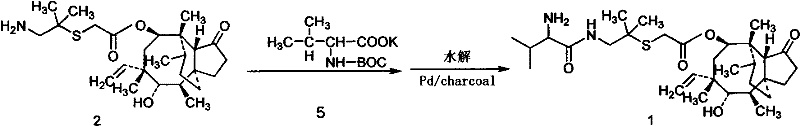
The Novel Approach
In stark contrast, the methodology disclosed in CN101597248B revolutionizes the production workflow by eliminating the need for expensive catalysts and complex protection-deprotection sequences. The new approach employs a direct coupling strategy between an activated D-hydroxy valine derivative and the pleuromutilin core in a tetrahydrofuran (THF) system. By utilizing vinyl chloroformate and organic bases like triethylamine for activation, the reaction proceeds smoothly at temperatures between 0°C and 20°C. This mild thermal profile preserves the integrity of the sensitive macrocyclic structure, preventing the formation of degradation byproducts common in harsher conditions. Furthermore, the workup procedure is drastically simplified to solvent removal, pH adjustment with hydrochloric acid, and freeze-drying, thereby bypassing the need for chromatography entirely and delivering a product ready for pharmaceutical use.
Mechanistic Insights into Mild Temperature Amide Coupling
The core of this technological advancement lies in the precise activation of the amino acid component under controlled低温 conditions. The mechanism involves the suspension of D-hydroxy valine Dane salt in THF, where it is treated with triethylamine and additives such as N,N-dimethylformamide or N-methylmorpholine to ensure complete solubility and reactivity. The subsequent addition of vinyl chloroformate generates a highly reactive mixed anhydride or activated carbamate intermediate in situ. This activation step is critical; it must be performed at 0-20°C to prevent racemization or decomposition of the chiral center. Once activated, the pleuromutilin derivative, dissolved in an equal mass of THF, is added dropwise while maintaining the temperature strictly between 0°C and 10°C. This rigorous temperature control during the addition phase is paramount for minimizing exothermic spikes that could compromise the stereochemical integrity of the final Valnemulin molecule.
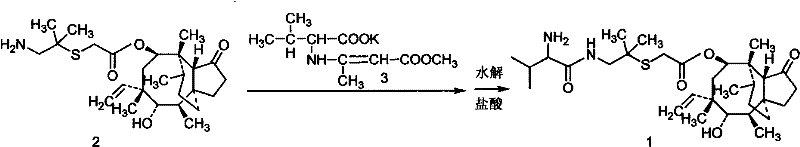
Following the coupling reaction, which typically proceeds for 2 to 5 hours, the process leverages the solubility differences of the product and impurities to achieve high purity. The removal of the organic solvent followed by the addition of water creates a biphasic environment where the product can be selectively protonated. Adjusting the pH to a range of 2.0 to 4.0 using hydrochloric acid facilitates the formation of the stable hydrochloride salt while keeping many organic impurities in solution or allowing them to be filtered off as insolubles. The final freeze-drying step removes residual water and volatile components, yielding a white powder with exceptional purity. This mechanism effectively controls the impurity profile by avoiding the high-energy states associated with reflux conditions or strong acidic/basic hydrolysis used in older methods, ensuring a cleaner reaction trajectory.
How to Synthesize Valnemulin Hydrochloride Efficiently
The synthesis of Valnemulin Hydrochloride via this patented route represents a paradigm shift towards greener and more efficient pharmaceutical manufacturing. The process is designed to be operationally simple, requiring standard reactor equipment capable of precise temperature control and vacuum distillation. Operators must focus on maintaining the strict thermal windows during the activation and coupling phases to ensure optimal yield and stereochemical retention. The elimination of chromatography not only reduces waste but also shortens the overall batch cycle time significantly. For detailed operational parameters, stoichiometry, and safety protocols, please refer to the standardized synthesis guide below which outlines the critical control points for successful implementation.
- Suspend D-hydroxy valine Dane salt in THF at 0-20°C and activate with triethylamine and vinyl chloroformate.
- Add the pleuromutilin derivative (Compound 2) dissolved in THF to the activated mixture while maintaining 0-10°C.
- Remove solvent, adjust pH to 2.0-4.0 with hydrochloric acid, filter insolubles, and freeze-dry the filtrate to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers profound strategic benefits that extend beyond mere technical superiority. The primary advantage lies in the substantial reduction of manufacturing costs driven by the simplification of the downstream processing. By removing the requirement for column chromatography and noble metal catalysts like palladium, the process eliminates two of the most cost-intensive variables in fine chemical production. This translates directly into a more competitive pricing structure for the final API intermediate, allowing buyers to optimize their raw material budgets without sacrificing quality. Additionally, the use of common, readily available solvents like THF and reagents like triethylamine ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce catalytic materials.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the complete removal of the chromatography purification step, which is traditionally a resource-heavy operation involving large volumes of silica gel and solvents. Furthermore, the avoidance of palladium catalysts removes the need for expensive metal recovery systems or the financial loss associated with metal scavenging resins. The simplified workup, consisting essentially of distillation, filtration, and lyophilization, reduces energy consumption and labor hours per batch. These cumulative efficiencies result in a drastically simplified cost structure, enabling significant savings in the overall cost of goods sold for cost reduction in antibiotic manufacturing.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on commodity chemicals rather than specialized catalysts enhances the robustness of the supply chain. Reagents such as vinyl chloroformate and D-hydroxy valine derivatives are widely produced and accessible from multiple global vendors, reducing the risk of single-source bottlenecks. The mild reaction conditions also imply lower safety risks and less stringent containment requirements compared to high-pressure hydrogenation processes, facilitating smoother transportation and storage of intermediates. This reliability ensures consistent delivery schedules and reduces the lead time for high-purity veterinary drug intermediates, allowing manufacturers to respond more agilely to market demands.
- Scalability and Environmental Compliance: The process is inherently scalable due to its linear workflow and absence of batch-limiting steps like column loading capacities. The mild temperature range of 0-20°C is easily achievable in large-scale reactors using standard cooling jackets, avoiding the engineering challenges of cryogenic or high-temperature systems. Environmentally, the reduction in solvent usage and the elimination of silica waste from chromatography contribute to a smaller environmental footprint. This aligns with increasingly stringent global regulations on pharmaceutical waste disposal, making the commercial scale-up of complex pleuromutilin derivatives more sustainable and compliant with modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Valnemulin synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality benchmarks associated with this method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production lines.
Q: How does this new method improve product purity compared to prior art?
A: Unlike previous methods requiring column chromatography which often resulted in poor bulk purity, this novel process achieves over 97% purity (HPLC) directly through controlled pH adjustment and freeze-drying, eliminating the need for complex purification steps.
Q: What are the critical reaction conditions for the coupling step?
A: The reaction must be conducted under mild temperature conditions between 0°C and 20°C, specifically maintaining 0-10°C during the dropwise addition of the pleuromutilin derivative to prevent degradation of the sensitive macrocyclic core.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for industrial suitability by using common solvents like THF, avoiding expensive noble metal catalysts like Palladium, and simplifying the workup to filtration and lyophilization, which significantly enhances scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valnemulin Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global veterinary pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering stringent purity specifications that meet or exceed international pharmacopoeia standards, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the mild temperature coupling process described in CN101597248B allows us to offer a superior product profile with minimized impurity levels.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
