Scalable Synthesis of Valnemulin Hydrochloride for Veterinary Pharmaceutical Manufacturing
Scalable Synthesis of Valnemulin Hydrochloride for Veterinary Pharmaceutical Manufacturing
The global demand for effective veterinary antibiotics continues to drive innovation in the synthesis of pleuromutilin derivatives, specifically Valnemulin Hydrochloride. A pivotal advancement in this field is detailed in patent CN101735123A, which discloses a robust method for synthesizing Valnemulin Hydrochloride starting from Pleuromutilin. This technology addresses critical bottlenecks in traditional manufacturing by introducing a mixed anhydride route that significantly enhances product purity and reaction efficiency. Unlike earlier methods that struggled with impurity profiles, this approach utilizes precise control over reaction temperatures, drying conditions, and pH levels to achieve a final product purity exceeding 97%. For pharmaceutical manufacturers and supply chain leaders, this represents a substantial opportunity to optimize the production of high-quality veterinary drugs while adhering to stringent regulatory standards for animal health medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Valnemulin has relied on processes such as those disclosed in US4675330, which involve reacting Pleuromutilin derivatives with valine in the presence of coupling agents followed by deprotection using Palladium on Carbon (Pd/C). A major drawback of these conventional techniques is the inability to consistently achieve high purity in the crude product, which typically remains below 93%. To meet pharmaceutical standards, these methods necessitate complex and costly purification steps, specifically column chromatography. This reliance on chromatography creates a significant barrier to industrial scalability, as it is time-consuming, solvent-intensive, and difficult to operate continuously in large-scale reactors. Furthermore, the use of precious metal catalysts like Pd/C introduces risks of heavy metal contamination, requiring additional downstream processing to ensure the final API meets safety specifications for veterinary use.
The Novel Approach
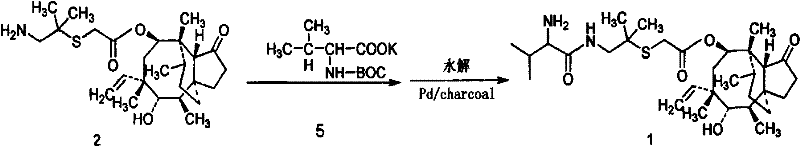
The methodology presented in CN101735123A offers a transformative alternative by bypassing the need for chromatographic purification entirely. The core innovation lies in the preparation of a specific intermediate, 14-O-[(1-amino-2-methyl-propyl-2-yl)acetyl] mutelin (ATAM), via a sulfonation pathway, followed by a mixed anhydride coupling reaction. By utilizing high boiling point solvents and optimizing the sequence of reagent addition—specifically mixing D-Valine Dane salt, N-methylmorpholine, and allyl chloroformate under controlled低温 conditions—the process drives the reaction towards higher selectivity. This strategic shift allows for the isolation of the product through crystallization and liquid-liquid extraction rather than column separation. Consequently, the process not only simplifies the operational workflow but also drastically reduces solvent consumption and waste generation, aligning with modern green chemistry principles in pharmaceutical manufacturing.
Mechanistic Insights into Mixed Anhydride Coupling and Purification
The chemical elegance of this synthesis is rooted in the precise formation of the mixed anhydride intermediate, which serves as the activated species for the acylation of the pleuromutilin core. The reaction is initiated by generating the mixed anhydride from D-Valine Dane salt and allyl chloroformate in the presence of N-methylmorpholine at temperatures between -10°C and -15°C. This low-temperature regime is critical for suppressing racemization and minimizing the formation of N-acyl urea byproducts, which are common impurities in peptide-like couplings. Once the mixed anhydride is formed, the ATAM intermediate is introduced, and the temperature is carefully ramped to 20-25°C to complete the acylation. The subsequent hydrolysis step is equally sophisticated; by adjusting the pH to 3.0-3.5 using dilute hydrochloric acid at 40-45°C, the protecting groups are selectively removed without degrading the sensitive pleuromutilin skeleton. This level of kinetic control ensures that the reaction proceeds with high fidelity, preserving the stereochemical integrity of the molecule.
Following the coupling and deprotection, the purification strategy leverages differential solubility and pH-dependent partitioning to remove impurities. The process involves a series of extractions where the pH is modulated between acidic (pH 3.0-3.5) and alkaline (pH 10.0-10.5) conditions. In the alkaline phase, unwanted acidic byproducts and residual starting materials are retained in the aqueous layer or washed away, while the desired Valnemulin base partitions into the organic phase (typically butyl acetate). Finally, the product is converted to its hydrochloride salt and isolated via freeze-drying. This multi-stage extraction protocol effectively scrubs the product of colored impurities and side-products, resulting in a white to off-white powder with a purity profile that consistently surpasses 97%, and in optimized embodiments, reaches up to 99.5%. Such rigorous control over the impurity spectrum is essential for ensuring the safety and efficacy of the final veterinary medication.
How to Synthesize Valnemulin Hydrochloride Efficiently
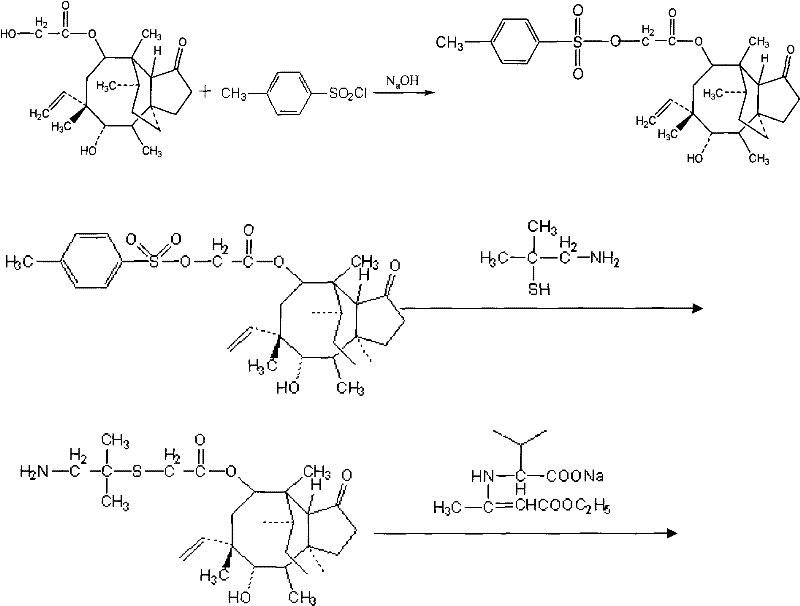
Implementing this synthesis route requires strict adherence to the specified reaction parameters to replicate the high yields and purity reported in the patent data. The process begins with the sulfonation of Pleuromutilin using Tosyl chloride, followed by the nucleophilic substitution with dimethyl mercaptamine to generate the key ATAM intermediate. The final stage involves the mixed anhydride coupling described previously. Operators must pay close attention to the temperature gradients during the anhydride formation and the precise pH adjustments during the workup phase, as these are the critical control points that determine the success of the batch. For a comprehensive breakdown of the reagent quantities, solvent volumes, and specific timing for each unit operation, please refer to the standardized synthesis guide provided below.
- Sulfonation of Pleuromutilin: React Pleuromutilin with Tosyl chloride in organic solvent (acetone/ethanol) at -5 to 5°C under alkaline conditions to form sulfonated pleurin.
- Preparation of ATAM: React sulfonated pleurin with dimethyl mercaptamine using a phase transfer catalyst at 50-55°C to obtain 14-O-[(1-amino-2-methyl-propyl-2-yl)acetyl] mutelin.
- Coupling and Purification: Form a mixed anhydride using D-Valine Dane salt, N-methylmorpholine, and allyl chloroformate at -10 to -15°C, followed by hydrolysis and pH-controlled extraction to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound benefits for procurement managers and supply chain directors focused on cost efficiency and reliability. The most significant economic driver is the elimination of column chromatography, which is notoriously expensive due to the high cost of silica gel and the large volumes of solvents required for elution. By replacing this batch-intensive process with crystallization and extraction, manufacturers can achieve a drastic reduction in operating expenses (OPEX) and a significant increase in throughput capacity. Furthermore, the use of high boiling point solvents such as t-butyl methyl ether and butyl acetate reduces solvent loss through evaporation during the reaction and concentration steps. This not only lowers raw material costs but also simplifies solvent recovery systems, contributing to a more sustainable and cost-effective production cycle that enhances overall margin potential.
- Cost Reduction in Manufacturing: The structural simplification of the process flow directly translates to lower manufacturing costs. By removing the need for precious metal catalysts like Palladium and avoiding chromatographic purification, the process eliminates two of the most cost-prohibitive elements of traditional semi-synthetic antibiotic production. The ability to use readily available reagents such as Tosyl chloride and allyl chloroformate, combined with the high molar yield of up to 63% reported in the embodiments, ensures a favorable cost of goods sold (COGS). Additionally, the reduced solvent volatility minimizes losses, further compounding the financial savings over long-term production runs.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the robustness of the reaction conditions and the availability of raw materials. The process does not rely on specialized or scarce reagents; instead, it utilizes commodity chemicals that are widely accessible in the global market. The tolerance of the reaction to slight variations in conditions, provided the critical temperature and pH windows are respected, reduces the risk of batch failures. This reliability ensures a consistent output of high-purity Valnemulin Hydrochloride, allowing supply chain managers to maintain steady inventory levels and meet the fluctuating demands of the veterinary pharmaceutical market without interruption.
- Scalability and Environmental Compliance: The design of this synthesis is inherently scalable, moving seamlessly from laboratory glassware to multi-ton industrial reactors. The replacement of chromatography with filtration and extraction is a key enabler for scale-up, as these unit operations are standard in large-scale chemical plants. Moreover, the process aligns with increasingly strict environmental regulations by reducing the generation of hazardous solid waste (spent silica) and minimizing the emission of volatile organic compounds (VOCs) through the use of higher boiling solvents. This environmental compliance reduces the burden on waste treatment facilities and mitigates regulatory risks associated with pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Valnemulin Hydrochloride based on the patented methodology. These insights are derived directly from the experimental data and process descriptions found in the source documentation, providing clarity on the feasibility and advantages of this route for potential partners and manufacturers.
Q: What is the primary advantage of this synthesis method over conventional chromatography-based routes?
A: The primary advantage is the elimination of column chromatography purification. Conventional methods often yield crude products with purity below 93% requiring expensive chromatographic separation. This novel method achieves purity greater than 97% through crystallization and pH-controlled extraction, making it highly suitable for industrial scale-up.
Q: How does the process control reaction parameters to ensure high quality?
A: The process strictly controls reaction temperature, drying conditions, and pH levels. For instance, the mixed anhydride formation is conducted at -10 to -15°C to prevent side reactions, while hydrolysis is managed at pH 3.0-3.5 and subsequent extraction at pH 10.0-10.5 to ensure precise impurity removal.
Q: Can this method be scaled for commercial production of veterinary antibiotics?
A: Yes, the method is designed for scalability. It utilizes high boiling point solvents which reduce volatility and loss during production, and replaces complex purification steps with standard filtration and extraction, facilitating the transition from laboratory to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valnemulin Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation veterinary therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated mixed anhydride chemistry described in CN101735123A can be successfully translated to an industrial setting. We are committed to delivering Valnemulin Hydrochloride that meets stringent purity specifications, supported by our rigorous QC labs which utilize advanced analytical techniques to verify identity and assay. Our facility is equipped to handle the specific solvent systems and temperature controls required for this synthesis, guaranteeing a supply of material that is consistent, safe, and ready for formulation.
We invite pharmaceutical companies and veterinary drug manufacturers to collaborate with us to leverage this advanced synthesis technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our manufacturing capabilities can support your supply chain goals for Valnemulin Hydrochloride and related pleuromutilin derivatives.
