Advanced Metal-Free Synthesis of Benzoxazole Compounds for Commercial Pharmaceutical Applications
Advanced Metal-Free Synthesis of Benzoxazole Compounds for Commercial Pharmaceutical Applications
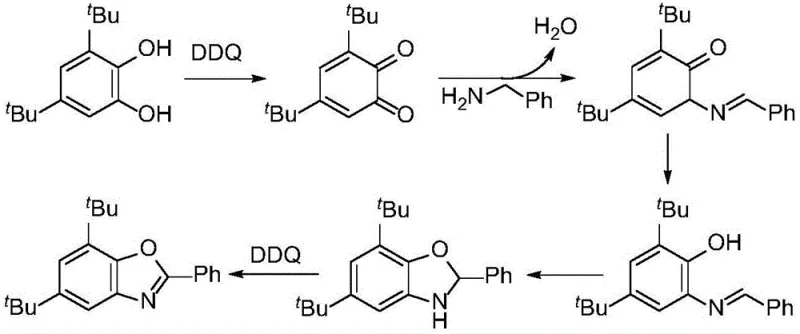
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for safer, greener, and more economically viable synthetic routes. A pivotal advancement in this domain is detailed in patent CN113264895A, which discloses a robust method for preparing benzoxazole compounds directly from catechol compounds and amine compounds. This technology represents a paradigm shift away from traditional, hazard-prone methodologies that rely on o-aminophenol substrates derived from risky nitration processes. By leveraging a metal-free oxidative cyclization strategy mediated by 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ), this innovation offers a streamlined pathway to high-value scaffolds essential for modern drug development. For R&D directors and procurement specialists seeking a reliable benzoxazole intermediate supplier, understanding the mechanistic elegance and operational simplicity of this patent is crucial for securing a competitive edge in the pharmaceutical supply chain.
The core breakthrough lies in the substitution of traditional precursors with readily available catechol derivatives, which are abundant in nature and significantly safer to handle on an industrial scale. The reaction system is remarkably simple, requiring only the mixing of the catechol substrate, the amine component, and the DDQ oxidant in a common organic solvent under an inert atmosphere. This approach not only simplifies the reaction setup but also drastically reduces the complexity of downstream processing. The resulting benzoxazole compounds, characterized by the general structural formula (I) where R and R1 represent a wide array of substituents, are obtained with high conversion rates and purity. This technological leap addresses critical pain points in cost reduction in pharmaceutical intermediates manufacturing by eliminating the need for expensive transition metal catalysts and complex ligand systems that often plague conventional synthesis routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzoxazole cores has heavily relied on o-aminophenol as the primary starting material. However, the industrial preparation of o-aminophenol typically involves the nitration of phenol or aniline, a process fraught with severe safety hazards including strong exothermic reactions and the potential for runaway temperature control leading to explosions. Furthermore, these traditional routes necessitate the use of highly corrosive mixed acids, posing significant environmental and equipment corrosion challenges. Beyond the safety concerns, many modern synthetic strategies for constructing the benzoxazole ring involve intramolecular cyclization of ortho-halogen or ortho-hydroxyphenyl amine compounds catalyzed by transition metals. While effective in the laboratory, these metal-catalyzed methods introduce the persistent risk of heavy metal residues in the final active pharmaceutical ingredients (APIs), necessitating costly and time-consuming purification steps to meet stringent regulatory limits. The reliance on precious metal catalysts also introduces volatility in raw material costs and supply chain stability, making these conventional methods less attractive for large-scale commercial production.
The Novel Approach
In stark contrast, the methodology outlined in CN113264895A circumvents these historical bottlenecks by utilizing catechol compounds, which are widely present in natural sources such as tea leaves and various plant derivatives, thereby bypassing the dangerous nitration steps entirely. This novel approach employs DDQ as a mild yet effective oxidant to drive the cyclization reaction under metal-free conditions, typically at temperatures ranging from 25°C to 70°C. The absence of transition metals not only eliminates the risk of metal contamination in the final product but also removes the economic burden associated with purchasing and recovering expensive catalysts and ligands. The reaction demonstrates exceptional functional group tolerance, accommodating a broad spectrum of substituents including halogens, esters, trifluoromethyl groups, and various alkyl or alkenyl chains without the need for protective group strategies. This versatility ensures that the process can be adapted for the commercial scale-up of complex pharmaceutical intermediates with minimal re-optimization, providing a robust platform for diverse chemical libraries.
Mechanistic Insights into DDQ-Mediated Oxidative Cyclization
The success of this synthetic route hinges on the unique oxidative properties of DDQ, which facilitates the dehydrogenative coupling of the catechol and amine substrates. Mechanistically, the reaction likely proceeds through an initial condensation between the amine and one of the hydroxyl-adjacent carbons of the catechol, followed by an oxidative aromatization step driven by the quinone. This sequence effectively constructs the oxazole ring while restoring the aromaticity of the benzene core. The use of DDQ is particularly advantageous because it acts as a hydride acceptor, driving the equilibrium towards the formation of the stable benzoxazole product without generating acidic byproducts that could degrade sensitive functional groups. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters such as stoichiometry and solvent choice to maximize yield and minimize impurity formation.
From an impurity control perspective, the metal-free nature of this reaction significantly simplifies the impurity profile. In traditional metal-catalyzed reactions, side products often arise from metal-ligand interactions or incomplete catalyst turnover, leading to complex mixtures that are difficult to separate. In this DDQ-mediated system, the primary byproducts are the reduced forms of the quinone, which are generally easier to remove during the aqueous workup and extraction phases described in the patent examples. The high tolerance for functional groups means that reactive moieties such as vinyl, ethynyl, or halogen substituents remain intact throughout the cyclization process, preserving the chemical integrity required for subsequent derivatization steps in drug synthesis. This level of control over the reaction pathway ensures that the resulting high-purity benzoxazole derivatives meet the rigorous quality standards demanded by global regulatory bodies, thereby reducing the risk of batch rejection and ensuring consistent supply continuity.
How to Synthesize Benzoxazole Efficiently
Implementing this synthesis protocol in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure optimal results. The general procedure involves mixing the catechol compound, the amine compound, and DDQ in a molar ratio of approximately 1:1.2:2.0, although slight variations within the range of 1:(1.1-1.5):(2-2.3) are permissible depending on the specific substrate reactivity. The reaction is conducted in solvents such as ethyl acetate or 1,2-dichloroethane under an inert nitrogen or argon atmosphere to prevent unwanted side reactions with atmospheric oxygen or moisture. Heating the mixture to a moderate temperature of 25-50°C for a duration of 5-10 hours typically drives the reaction to completion. Following the reaction, the workup is straightforward: the mixture is cooled, washed with saturated sodium chloride solution to remove water-soluble impurities, and extracted with ethyl acetate. The crude product is then concentrated and purified via standard column chromatography to yield the analytically pure benzoxazole derivative.
- Mix catechol compound, amine compound, and DDQ oxidant in a reaction vessel under inert gas atmosphere.
- Add solvent such as ethyl acetate or 1,2-dichloroethane and heat to 25-70°C for 3-12 hours with stirring.
- Cool to room temperature, wash with saturated NaCl, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of transition metal catalysts represents a direct reduction in raw material costs, as there is no longer a need to procure expensive palladium, copper, or iron salts along with their specialized ligands. Furthermore, the removal of metal catalysts obviates the need for sophisticated metal scavenging resins or additional purification steps designed to lower residual metal levels, which significantly streamlines the manufacturing workflow. This simplification of the process flow leads to shorter production cycles and reduced labor costs, contributing to substantial overall cost savings in the manufacturing of benzoxazole-based intermediates. The use of benign solvents like ethyl acetate further aligns with green chemistry principles, potentially lowering waste disposal costs and easing regulatory compliance burdens associated with volatile organic compound (VOC) emissions.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound. By removing the dependency on precious metal catalysts, manufacturers can avoid the price volatility associated with commodities like palladium or rhodium. Additionally, the simplified workup procedure, which relies on basic extraction and chromatography rather than complex metal removal technologies, reduces the consumption of auxiliary materials and energy. The high yields reported in the patent examples, often exceeding 80-90%, indicate a highly efficient atom economy that minimizes waste generation. This efficiency directly correlates to a lower cost of goods sold (COGS), allowing companies to offer more competitive pricing to their clients while maintaining healthy profit margins. The avoidance of hazardous nitration steps also reduces the capital expenditure required for specialized explosion-proof infrastructure, further enhancing the financial viability of the process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of catechol compounds, which are naturally abundant and commercially available in large quantities from diverse global sources. Unlike specialized organometallic reagents that may have limited suppliers and long lead times, catechols and simple amines are commodity chemicals with stable supply lines. This abundance ensures that production schedules are less likely to be disrupted by raw material shortages. Moreover, the mild reaction conditions (25-70°C) reduce the stress on reactor equipment, leading to lower maintenance requirements and higher equipment availability. The robustness of the reaction against varying substrate structures means that a single production line can be easily adapted to manufacture a wide variety of benzoxazole derivatives, providing the flexibility needed to respond quickly to changing market demands without significant retooling investments.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory synthesis to multi-ton commercial production is facilitated by the absence of exothermic hazards associated with traditional nitration or strong acid treatments. The moderate temperatures and ambient pressure conditions make the reaction inherently safer to operate at scale, reducing the risk of thermal runaways. From an environmental standpoint, the metal-free nature of the process ensures that the final effluent streams are free from heavy metal contamination, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The use of DDQ, while an oxidant, generates byproducts that are manageable within standard waste streams, avoiding the creation of hazardous heavy metal sludge. This environmental compatibility not only protects the ecosystem but also safeguards the company's reputation and license to operate in regions with rigorous environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel benzoxazole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the practical aspects of adopting this method. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the safety advantages of using catechol over o-aminophenol?
A: Traditional o-aminophenol synthesis involves dangerous nitration reactions with strong corrosive mixed acids and explosion risks. Using naturally abundant catechol compounds eliminates these hazardous preparation steps, significantly improving industrial safety.
Q: Does this method leave heavy metal residues in the final product?
A: No. Unlike conventional transition metal-catalyzed methods, this novel process operates under metal-free conditions using DDQ as the oxidant, ensuring the final benzoxazole products are free from toxic metal contamination.
Q: What is the substrate scope for this synthesis method?
A: The method exhibits high functional group tolerance, successfully synthesizing derivatives with aryl, trifluoromethyl, ester, halogen, alkenyl, alkynyl, and alkyl substituents, making it versatile for diverse drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis route described in CN113264895A for the production of high-quality benzoxazole intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of benzoxazole derivative meets the exacting standards required for pharmaceutical applications. We are committed to leveraging this advanced technology to deliver superior value to our partners, combining technical excellence with operational efficiency to support your drug development timelines.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol for your specific target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your supply chain. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing and bringing your next-generation therapeutics to market faster.
