Scalable Metal-Free Synthesis of 2-Substituted Benzoxazoles and Benzothiazoles for Pharmaceutical Applications
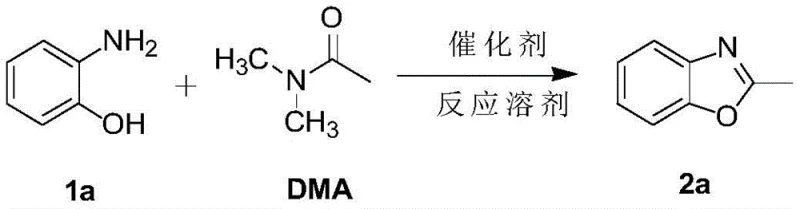
The pharmaceutical and fine chemical industries continuously seek robust, scalable methodologies for constructing privileged heterocyclic scaffolds, among which 2-substituted benzoxazoles and 2-substituted benzothiazoles hold paramount importance due to their prevalence in bioactive molecules. Patent CN109265409B introduces a transformative synthesis method that addresses long-standing inefficiencies in producing these critical structures. By leveraging imidazole hydrochloride as a potent organocatalyst, this technology enables the direct cyclization of o-aminophenols and o-aminothiophenols with amide derivatives under remarkably mild and metal-free conditions. This innovation represents a significant leap forward for any reliable pharmaceutical intermediate supplier aiming to streamline production workflows while maintaining stringent quality standards. The protocol eliminates the dependency on harsh reagents and complex transition metal systems, thereby offering a cleaner, more economically viable pathway for the commercial scale-up of complex heterocycles essential for modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzoxazole and benzothiazole core has relied heavily on condensation reactions involving o-aminophenols or o-aminothiophenols with carboxylic acids, aldehydes, or acyl chlorides, often necessitating aggressive dehydrating agents or Lewis acids. Traditional protocols frequently employ corrosive reagents such as phosphorus oxychloride (POCl3) or require stoichiometric amounts of expensive acyl transfer agents like 2-acyl-4,5-dichloropyridazin-3(2H)-ones, which significantly inflate raw material costs and complicate waste management. Furthermore, many established routes depend on transition metal catalysts that introduce the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet regulatory limits for high-purity pharmaceutical intermediates. These conventional approaches often suffer from limited substrate scope, poor atom economy, and the requirement for specialized high-pressure equipment, creating bottlenecks that hinder cost reduction in pharmaceutical manufacturing and delay time-to-market for critical therapeutic candidates.
The Novel Approach
In stark contrast, the methodology disclosed in CN109265409B utilizes a simple yet highly effective organocatalytic system driven by imidazole hydrochloride. This novel approach operates efficiently in N,N-dimethylacetamide (DMA) solvent at temperatures between 160°C and 170°C, achieving excellent conversion without the need for exotic ligands or toxic metals. The reaction demonstrates exceptional versatility, accommodating a wide array of substituents including halogens, nitro groups, and alkyl chains on both the aromatic ring and the amide component. As illustrated in the specific model reaction optimization, the process yields up to 87% of the desired 2-methylbenzoxazole under optimized conditions, vastly outperforming trace yields observed with non-catalytic or alternative acidic conditions.
This streamlined protocol not only simplifies the operational setup by avoiding autoclaves but also ensures reducing lead time for high-purity intermediates through straightforward workup procedures involving aqueous quenching and standard extraction. The elimination of corrosive concentrated hydrochloric acid and metal catalysts fundamentally alters the safety and environmental profile of the synthesis, making it an ideal candidate for green chemistry initiatives within large-scale production facilities.
Mechanistic Insights into Imidazole Hydrochloride-Catalyzed Cyclization
The efficacy of this synthetic route lies in the unique ability of imidazole hydrochloride to act as a Brønsted acid catalyst that activates the amide carbonyl towards nucleophilic attack by the ortho-amino group of the phenol or thiophenol substrate. The general reaction scheme encompasses the condensation of compound I (o-aminophenol/o-aminothiophenol) with compound III (amide derivative) to form the fused heterocyclic system II. The proton source facilitates the initial nucleophilic addition, followed by a dehydration step that drives the equilibrium towards the formation of the stable aromatic benzoxazole or benzothiazole ring. This mechanism avoids the formation of stable metal-complex intermediates that often plague transition-metal catalyzed cycles, thereby minimizing side reactions and impurity profiles associated with metal coordination.

Furthermore, the reaction exhibits remarkable functional group tolerance, a critical attribute for R&D teams designing diverse libraries of drug candidates. The catalytic system remains robust in the presence of electron-withdrawing groups such as nitro and halogen substituents, which typically deactivate aromatic rings in electrophilic substitutions, as well as electron-donating groups like methoxy and methyl. This broad compatibility ensures that the synthesis of high-purity benzoxazole derivatives can be achieved across a wide chemical space without requiring extensive re-optimization for each new substrate. The absence of radical pathways or harsh oxidative conditions further preserves sensitive functionalities, ensuring that the final impurity spectrum is clean and manageable, which is essential for meeting the rigorous specifications demanded by global regulatory bodies for active pharmaceutical ingredients.
How to Synthesize 2-Substituted Benzoxazoles Efficiently
The practical implementation of this technology involves a straightforward one-pot procedure that is easily adaptable to existing reactor infrastructure. The process begins by charging the reaction vessel with the o-aminophenol substrate, the appropriate amide derivative, and the imidazole hydrochloride catalyst in DMA solvent. The mixture is then heated to the optimal temperature range of 160°C to 170°C and maintained for 8 to 10 hours to ensure complete conversion. Following the reaction period, the mixture is cooled and quenched with water, allowing for the separation of the organic product via extraction with ethyl acetate. The detailed standardized synthesis steps, including specific molar ratios, washing protocols, and purification techniques via silica gel chromatography or recrystallization, are outlined below to ensure reproducibility and consistency across different production batches.
- Combine o-aminophenol substrate with imidazole hydrochloride catalyst (0.5 equivalents) and N,N-dimethylacetamide (DMA) solvent in a reaction vessel.
- Heat the reaction mixture to 160°C and stir for 8 to 10 hours to facilitate the cyclization process.
- Upon completion, quench with water, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthesis method offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility inherent in traditional heterocycle manufacturing. By replacing expensive transition metal catalysts and corrosive reagents with commodity-grade imidazole hydrochloride and DMA, the raw material cost structure is significantly optimized. The elimination of heavy metals removes the necessity for specialized scavenging resins and extensive analytical testing for residual metals, which translates into substantial operational expenditure savings and faster batch release times. Additionally, the use of readily available starting materials ensures a stable supply chain, mitigating the risks associated with sourcing specialized or proprietary reagents that often face geopolitical or logistical disruptions.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with an inexpensive organocatalyst drastically lowers the direct material costs associated with each production batch. Furthermore, the simplified workup procedure reduces solvent consumption and waste disposal fees, contributing to a leaner overall manufacturing budget. The avoidance of high-pressure autoclaves also lowers capital expenditure requirements for reactor equipment, allowing for production in standard glass-lined or stainless steel vessels.
- Enhanced Supply Chain Reliability: Utilizing common chemical feedstocks such as o-aminophenols and dimethylacetamide ensures that production is not held hostage by the availability of niche reagents. This commoditization of inputs enhances the resilience of the supply chain, enabling consistent delivery schedules even during market fluctuations. The robustness of the reaction conditions further minimizes the risk of batch failures, ensuring a steady flow of materials to downstream formulation teams.
- Scalability and Environmental Compliance: The metal-free nature of this process aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge and hazardous waste generation. The simplified effluent profile facilitates easier wastewater treatment and compliance with green chemistry mandates. Moreover, the reaction's tolerance to various functional groups allows for the consolidation of multiple synthetic steps, improving the overall process mass intensity and supporting sustainable manufacturing practices on a multi-ton scale.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these nuances is vital for process chemists evaluating the feasibility of adopting this route for their specific project needs.
Q: What are the advantages of using imidazole hydrochloride over traditional metal catalysts?
A: Imidazole hydrochloride eliminates the need for expensive transition metals and corrosive reagents like POCl3, simplifying downstream purification and reducing heavy metal contamination risks in pharmaceutical intermediates.
Q: What is the optimal temperature range for this cyclization reaction?
A: The patent data indicates that while reactions can occur between 60°C and 180°C, the optimal yield is achieved at 160°C to 170°C, where conversion is maximized without excessive degradation.
Q: Does this method tolerate electron-withdrawing groups on the substrate?
A: Yes, the method demonstrates excellent functional group tolerance, successfully synthesizing derivatives with nitro, halogen, and methoxy substituents with yields ranging from 60% to 88%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted Benzoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient heterocycle synthesis plays in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and robust. We are committed to delivering high-purity 2-substituted benzoxazoles that meet the most stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance guarantees that every batch delivered adheres to the highest industry standards, providing our partners with the confidence needed to advance their clinical programs.
We invite you to collaborate with us to leverage this advanced organocatalytic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can optimize your supply chain and reduce overall project costs.
