Optimizing Prednisolone Acetate Production: A Zinc-Mediated Route for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for critical corticosteroids, and the preparation of prednisolone acetate stands as a prime example of process innovation. Patent CN103396468A introduces a transformative synthesis method that addresses long-standing inefficiencies in steroid manufacturing. This technical disclosure outlines a streamlined four-step sequence converting a specific triene precursor into high-purity prednisolone acetate through a series of controlled bromination and reduction reactions. Unlike legacy biotransformation methods that suffer from low conversion rates and difficult downstream processing, this chemical approach offers a deterministic, high-yield alternative. By leveraging selective reagents such as N-bromosuccinimide (NBS) and zinc powder, the process effectively navigates the complex stereochemistry of the steroid nucleus. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable prednisolone acetate supplier capable of delivering consistent quality without the baggage of heavy metal contaminants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of prednisolone acetate has been plagued by significant technical and economic bottlenecks. Traditional biotransformation routes, while conceptually green, often exhibit transformation efficiencies capped below 80%, leading to substantial material loss and extended production cycles. Furthermore, the extraction and purification of the target molecule from fermentation broths are notoriously difficult, introducing variability that complicates quality control. On the chemical synthesis front, earlier patents disclosed routes relying on chromium-based reduction agents and iodine intermediates. The use of chromium poses severe environmental and safety liabilities due to toxic heavy metal residues that require expensive and rigorous removal protocols to meet pharmacopeial standards. Additionally, iodine-based intermediates are inherently unstable, prone to decomposition, and necessitate storage under nitrogen in dark conditions, thereby inflating raw material costs and logistical complexity. These factors collectively hinder the commercial scale-up of complex steroid intermediates using older methodologies.
The Novel Approach
The methodology disclosed in the patent data presents a decisive break from these constraints by utilizing a zinc-mediated debromination strategy. This novel approach initiates with a highly selective bromination of the 1,4,9-triene precursor, followed by a gentle reduction using zinc powder that preserves sensitive carbonyl functionalities while restoring the necessary double bond geometry. The elimination of chromium not only resolves regulatory compliance issues regarding heavy metals but also drastically simplifies the workup procedure. The process is designed to be operationally simple, potentially executable in a single reactor or a simplified two-vessel setup, which minimizes capital expenditure on acid-alkali resistant equipment. By replacing unstable iodine intermediates with robust bromo-derivatives and utilizing cost-effective reagents like NBS and zinc, the route achieves a total recovery rate exceeding 90%. This efficiency directly translates to cost reduction in corticosteroid manufacturing, making it an attractive option for high-volume production.
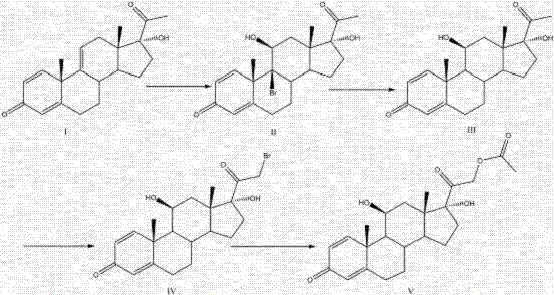
Mechanistic Insights into Zinc-Mediated Debromination and Substitution
The core chemical innovation lies in the precise manipulation of the steroid backbone through electrophilic addition and subsequent elimination. In the first stage, N-bromosuccinimide (NBS) acts as a source of electrophilic bromine, selectively attacking the electron-rich 9,11-double bond of the 1,4,9-triene system to form the 9-bromo intermediate (Compound II). This step is critical as it sets the stage for the introduction of the 11-beta-hydroxyl group functionality indirectly. The subsequent reduction step employs activated zinc powder in an ether solvent, which serves as a mild reducing agent. Mechanistically, the zinc facilitates a reductive debromination that eliminates the bromine atom at the 9-position while simultaneously isomerizing the double bond system to the thermodynamically stable 1,4-diene configuration found in Compound III. Crucially, the reaction conditions are tuned to ensure that the ketone groups at positions 3 and 20 remain untouched, preserving the oxidation state required for the final API.
Following the restoration of the diene system, the synthesis proceeds with an acidic bromination at the 21-position, catalyzed by aluminum chloride, to generate the 21-bromo derivative (Compound IV). This activated halide is then subjected to a nucleophilic substitution reaction in the presence of sodium hydroxide and triethylamine. The acetate ion, generated in situ or added as a salt, displaces the bromide to form the final 21-acetate ester (Compound V). The choice of solvents, particularly acetone and ether, plays a pivotal role in maintaining the solubility of intermediates while allowing for the precipitation of inorganic salts. This mechanistic pathway ensures that impurities are largely water-soluble byproducts, allowing the final product to be isolated via simple water precipitation. Such a clean reaction profile is essential for achieving high-purity prednisolone acetate without the need for resource-intensive chromatography.
How to Synthesize Prednisolone Acetate Efficiently
Implementing this synthesis route requires careful control of reaction parameters, particularly temperature and stoichiometry, to maximize yield and minimize side reactions. The process begins with the dissolution of the triene starting material in acetone, followed by the controlled addition of NBS at low temperatures (0-10°C) to prevent poly-bromination. After the initial bromination, the solvent is swapped or adjusted for the reduction phase, where zinc powder is added in batches to manage the exotherm and ensure complete consumption of the bromo-intermediate. The subsequent bromination and substitution steps require precise pH control and temperature management (50-55°C) to drive the esterification to completion. The detailed standardized operating procedures, including specific molar equivalents and workup protocols, are outlined in the technical guide below.
- Perform selective bromination of the starting triene compound using N-bromosuccinimide (NBS) in acetone at low temperature to form the 9-bromo intermediate.
- Execute reduction-debromination using activated zinc powder in an ether solvent to restore the double bond system without reducing carbonyl groups.
- Conduct acidic bromination followed by nucleophilic substitution with sodium acetate/triethylamine to install the 21-acetate group and finalize the steroid structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-mediated synthesis route offers compelling strategic advantages beyond mere technical feasibility. The primary benefit is the substantial simplification of the supply chain regarding raw material sourcing and waste management. By eliminating the need for chromium reagents and unstable iodine compounds, manufacturers can source cheaper, more stable, and widely available alternatives like zinc powder and NBS. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates the risk of supply disruptions associated with specialized or regulated reagents. Furthermore, the simplified purification process, which relies on water precipitation rather than complex extractions or column chromatography, significantly reduces solvent consumption and processing time. This efficiency gain allows for faster batch turnover, effectively reducing lead time for high-purity glucocorticoids and enhancing overall supply chain responsiveness.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous chromium reagents with economical zinc powder results in a direct decrease in raw material costs. Additionally, the ability to perform multiple steps in a single reactor (or a simplified two-reactor system) reduces the capital intensity of the production line. The high yield reported in the patent data implies less raw material waste per kilogram of finished product, further driving down the unit cost. The elimination of heavy metal removal steps also saves on the costs associated with specialized scavengers and extensive analytical testing for residual metals.
- Enhanced Supply Chain Reliability: The stability of the intermediates in this new route contrasts sharply with the light-sensitive and nitrogen-dependent iodine intermediates of the past. This robustness means that production does not need to be halted due to intermediate degradation, allowing for more flexible scheduling and inventory management. The use of common industrial solvents like acetone and ether ensures that solvent supply is never a bottleneck. Consequently, manufacturers can maintain a more consistent output schedule, ensuring reliable delivery to downstream pharmaceutical formulators.
- Scalability and Environmental Compliance: From an environmental perspective, the absence of hexavalent chromium simplifies wastewater treatment and reduces the regulatory burden on the manufacturing facility. The process generates water-soluble byproducts that are easily separated from the organic product, minimizing the volume of hazardous organic waste. This 'greener' profile facilitates easier permitting for capacity expansion and aligns with the increasing global demand for sustainable pharmaceutical manufacturing practices. The straightforward scale-up potential ensures that production can be ramped from pilot scale to multi-ton commercial volumes with minimal process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does this zinc-mediated route improve upon traditional chromium-based synthesis?
A: Traditional methods often utilize chromium reagents which leave toxic heavy metal residues requiring complex removal steps. This novel route replaces chromium with zinc powder, eliminating heavy metal contamination risks and simplifying the purification process significantly.
Q: What are the purity specifications achievable with this method?
A: The patent data indicates that the final product can achieve high purity levels, specifically citing HPLC purity of approximately 98.5% to 99.3% directly after simple water precipitation and filtration, minimizing the need for extensive chromatographic purification.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It features high yields (over 90%), utilizes common solvents like acetone and ether, and allows for a 'one-pot' or simplified two-reactor workflow that reduces equipment investment and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prednisolone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes for vital corticosteroids like prednisolone acetate. Our technical team has thoroughly analyzed the zinc-mediated pathway disclosed in CN103396468A and validated its potential for robust commercial production. As a dedicated CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of heavy metals and ensuring the stereochemical integrity of the final steroid product.
We invite pharmaceutical companies and chemical distributors to collaborate with us to leverage this advanced manufacturing technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain with high-quality, cost-effective prednisolone acetate produced via this superior synthetic method.
