Advanced Bio-Chemical Synthesis of Prednisolone Acetate for Industrial Scale Manufacturing
Advanced Bio-Chemical Synthesis of Prednisolone Acetate for Industrial Scale Manufacturing
The pharmaceutical industry constantly seeks robust pathways for glucocorticoid production that balance high purity with economic feasibility. A significant breakthrough in this domain is detailed in patent CN112375114A, which outlines a sophisticated preparation method for prednisolone acetate and its precursor, prednisolone. This innovative approach utilizes anecortave acetate as a primary raw material, subjecting it to a sequential series of biological fermentation, esterification, bromination, and debromination reactions. The technical significance of this patent lies in its ability to achieve an impressive overall yield of up to 81.75 percent, with the final prednisolone product demonstrating an HPLC area normalization content of up to 99.5 percent. For R&D directors and procurement specialists, this represents a pivotal shift towards more efficient pharmaceutical intermediates manufacturing, offering a short synthetic route that is inherently suitable for industrial production and delivers substantial cost reductions compared to legacy methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional preparation methods for prednisolone acetate have long been plagued by inefficiencies that hinder scalable cost reduction in glucocorticoid manufacturing. Historically, synthesis has relied on biological conversion or complex chemical routes starting from expensive substrates like 17 alpha-hydroxy-1, 4, 9-triene-pregna-3, 20-diketone. These conventional pathways often suffer from low substrate feeding concentrations and extended production periods, which drastically inflate operational expenditures. Furthermore, the post-treatment processes in these older methods are notoriously difficult, leading to challenging separation protocols that compromise final purity. The reliance on multi-step protection and deprotection strategies, such as 3-position and 20-position ketosemicarbazone protection followed by 11-position ketoreduction, introduces unnecessary complexity. Consequently, the overall yield remains low, and the high cost of goods sold makes these traditional routes less attractive for reliable prednisolone acetate supplier networks aiming to optimize their supply chains.
The Novel Approach
In stark contrast, the novel approach described in the patent leverages a hybrid bio-chemical strategy that streamlines the entire synthesis. By initiating the process with anecortave acetate, a readily available and cost-effective starting material, the method bypasses many of the cumbersome steps associated with traditional steroid functionalization. The integration of biological fermentation to introduce critical hydroxyl groups, followed by precise chemical modifications, creates a highly efficient workflow. This route not only simplifies the operational steps but also ensures that the reaction conditions remain mild and manageable. The result is a process that effectively improves both yield and purity, making it ideally suited for the commercial scale-up of complex steroid syntheses. This modernization of the synthetic pathway addresses the core pain points of the industry by delivering a product that is economic, environment-friendly, and possesses superior physical properties such as a white color and good suction filtration characteristics.
Mechanistic Insights into Bromination and Debromination Strategy
The core chemical transformation in this synthesis involves a precise bromination followed by a debromination sequence, which is critical for establishing the correct stereochemistry and functionality of the steroid backbone. The bromination reaction is conducted using reagents such as dibromohydantoin or N-bromosuccinimide in a mixed solvent system of organic solvents and water, typically at temperatures ranging from -20°C to 15°C. This controlled environment ensures high regioselectivity. Following this, the debromination step utilizes a reducing system comprising zinc and chromium trichloride. A key mechanistic advantage here is the use of a specific post-treatment protocol where sodium salts, such as sodium thiosulfate or sodium sulfite, are introduced. This addition is not merely a quenching step; it actively destroys residual solvents like thioglycolic acid, which are often responsible for impurity entrapment and unpleasant odors. By neutralizing these reactive species, the process prevents the product from being wrapped or dissolved within the reaction matrix, thereby securing a high-purity isolate.
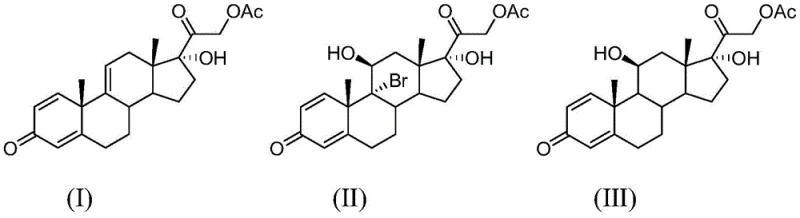
Furthermore, the esterification and fermentation steps are equally critical in defining the impurity profile of the final API intermediate. The esterification of the precursor compound (IV) to form compound (I) employs catalysts like 4-dimethylaminopyridine (DMAP) and acetylating agents such as acetic anhydride. This step must be tightly controlled to prevent over-acetylation or degradation of the sensitive steroid nucleus. Similarly, the biological fermentation of anecortave acetate using strains like Nocardia or Arthrobacter simplex requires precise control of aeration and pH to maximize the conversion to the 11-beta-hydroxy intermediate. The synergy between these biological and chemical steps ensures that side reactions are minimized. The rigorous control over reaction parameters, such as maintaining the debromination temperature between 10°C and 25°C, further guarantees that the structural integrity of the molecule is preserved, resulting in a final product that meets stringent high-purity pharmaceutical intermediates specifications.
How to Synthesize Prednisolone Acetate Efficiently
Implementing this synthesis requires a disciplined approach to process engineering, starting from the fermentation of anecortave acetate and proceeding through esterification to the final bromination-debromination sequence. The patent details specific mass ratios and solvent systems that are crucial for reproducibility, such as using a mixture of dimethyl sulfoxide and acetone for the debromination step to prevent system curing and ensure thorough reagent contact. Operators must pay close attention to the post-treatment phase, where the addition of sodium thiosulfate at a mass ratio of approximately 0.2:1 relative to the substrate is essential for odor control and impurity removal. The following guide outlines the standardized operational framework derived from the patent data, ensuring that the reducing lead time for high-purity API intermediates is achieved without compromising on quality or safety standards during the manufacturing process.
- Perform biological fermentation on anecortave acetate using Nocardia or Arthrobacter simplex to obtain the hydroxylated intermediate.
- Conduct esterification using acetic anhydride and a catalyst like DMAP to protect the 21-position hydroxyl group.
- Execute bromination followed by debromination using zinc and chromium trichloride, finishing with a sodium salt post-treatment for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers transformative benefits that extend beyond simple yield improvements. The shift to anecortave acetate as a starting material fundamentally alters the cost structure of production by utilizing a cheaper, more accessible substrate compared to traditional precursors. This change eliminates the need for expensive and complex protection-deprotection sequences, directly translating to significant operational savings. Moreover, the simplified post-treatment process reduces the burden on waste management systems, as the destruction of malodorous compounds like thioglycolic acid creates a more environmentally compliant manufacturing footprint. These factors combined create a resilient supply chain capable of meeting high-volume demands with greater reliability and lower risk of disruption due to raw material scarcity or regulatory hurdles associated with hazardous waste.
- Cost Reduction in Manufacturing: The elimination of complex multi-step protection strategies and the use of cost-effective biological fermentation significantly lowers the cost of goods sold. By avoiding expensive substrates and reducing the number of unit operations, the process minimizes energy consumption and labor costs. The high overall yield means less raw material is wasted per kilogram of final product, providing a direct margin improvement that enhances competitiveness in the global market for glucocorticoid APIs.
- Enhanced Supply Chain Reliability: Utilizing anecortave acetate, which is readily available, mitigates the risk of supply bottlenecks often associated with specialized steroid intermediates. The robustness of the fermentation step, which can achieve yields up to 98 percent, ensures a consistent flow of high-quality intermediate into the chemical synthesis phase. This stability allows for better production planning and inventory management, ensuring that downstream customers receive their orders on time without the delays typical of more fragile synthetic routes.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing common solvents and mild reaction conditions that are easily managed in large-scale reactors. The specific post-treatment with sodium salts not only improves product quality but also addresses environmental concerns by neutralizing noxious byproducts. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the costs associated with environmental remediation and worker safety protocols in the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent, providing clarity on how this method resolves historical challenges in prednisolone acetate production. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement teams assessing the long-term viability of suppliers utilizing this technology.
Q: What are the advantages of using anecortave acetate as a starting material?
A: Using anecortave acetate allows for a shorter synthetic route compared to traditional methods starting from 17 alpha-hydroxy-1, 4, 9-triene-pregna-3, 20-diketone. It leverages cost-effective biological fermentation to introduce the 11-beta hydroxyl group, significantly reducing substrate costs and simplifying the overall process complexity.
Q: How does the new post-treatment method improve product quality?
A: The novel post-treatment involves adding sodium salts such as sodium thiosulfate or sodium sulfite. This step effectively destroys residual thioglycolic acid and other solvents, eliminating sharp odors and preventing the product from being wrapped or dissolved by the reaction system. This results in a white, dry solid suitable for suction filtration with purity exceeding 99%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial applicability. It utilizes mild reaction conditions, such as bromination at -20°C to 15°C and debromination at 10°C to 25°C, which are easily manageable in standard reactors. The high overall yield of up to 81.75% and the use of common solvents make it economically viable for mass manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prednisolone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN112375114A to meet the evolving demands of the pharmaceutical sector. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of prednisolone acetate or related intermediate meets the highest global standards. We are committed to leveraging our technical expertise to optimize these bio-chemical processes, delivering products that offer both superior quality and economic value to our partners.
We invite you to collaborate with us to explore how this advanced synthesis can benefit your specific supply chain requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this route can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-performance pharmaceutical intermediates.
