Optimizing Rivaroxaban Production: A Technical Analysis of Novel Amide Coupling Strategies
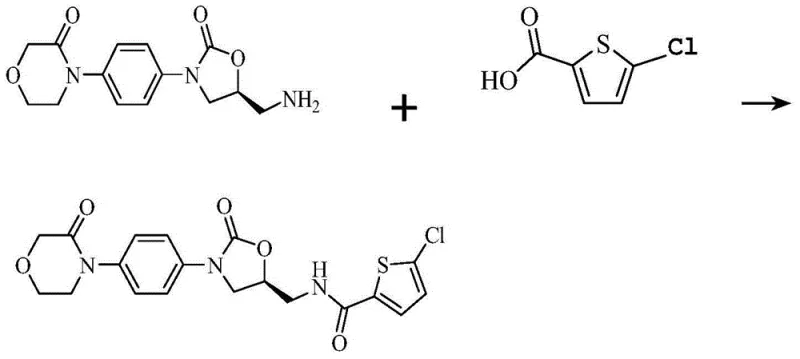
The pharmaceutical landscape for anticoagulants has been revolutionized by direct Factor Xa inhibitors, with Rivaroxaban standing out as a critical therapeutic agent for preventing deep vein thrombosis and pulmonary embolism. As the demand for this high-value Active Pharmaceutical Ingredient (API) continues to surge globally, the efficiency of its synthetic route becomes a paramount concern for manufacturers. Patent CN103724336A introduces a significant technological breakthrough in the final coupling step of Rivaroxaban synthesis, addressing long-standing issues related to cost, yield, and operational safety. This patent discloses a novel method wherein 4-[4-[(5S)-5-(aminomethyl)-2-oxo-3-oxazolidinyl]phenyl]-3-morpholinone reacts with 5-chlorothiophene-2-carboxylic acid using specific condensing agents like N,N'-carbonyldiimidazole (CDI) or N,N'-dicyclohexylcarbodiimide (DCC). By shifting away from traditional, hazardous reagents, this innovation offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates, ensuring a more stable supply chain for downstream drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rivaroxaban has relied on methodologies that present substantial hurdles for industrial-scale manufacturing. One prevalent prior art method utilizes 1-ethyl-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDCI) as the condensing agent. While effective in laboratory settings, EDCI is notoriously expensive, which inflates the overall production cost of the API. Furthermore, reactions mediated by EDCI often suffer from lower molar yields and generate urea byproducts that can be challenging to separate, complicating the purification process and potentially impacting the impurity profile of the final drug substance. Another conventional approach involves converting 5-chlorothiophene-2-carboxylic acid into its corresponding acid chloride using thionyl chloride (SOCl2).
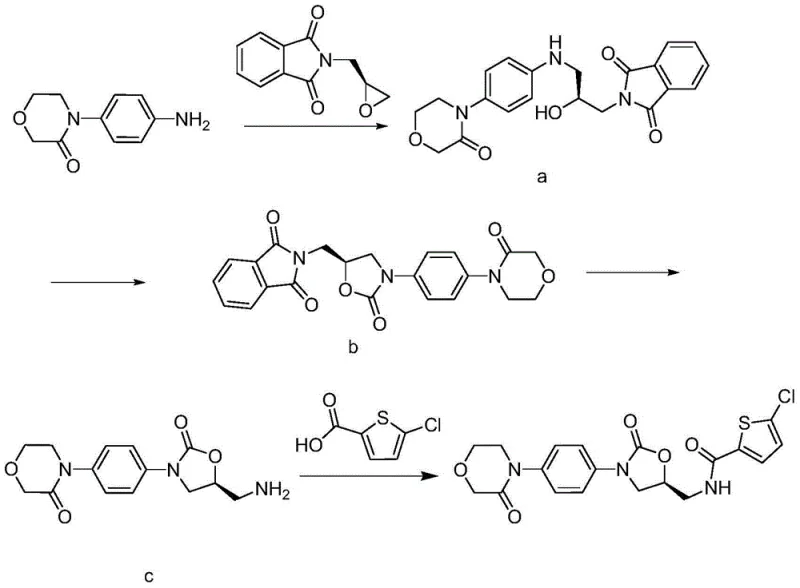
This chlorination step is highly problematic from an engineering perspective; thionyl chloride is extremely corrosive and reacts violently to release large volumes of hydrogen chloride gas. This necessitates the use of specialized, corrosion-resistant reactor equipment and sophisticated scrubbing systems to handle toxic emissions, thereby increasing capital expenditure and operational complexity. The handling of such hazardous materials also poses significant safety risks to personnel and complicates regulatory compliance regarding environmental discharge.
The Novel Approach
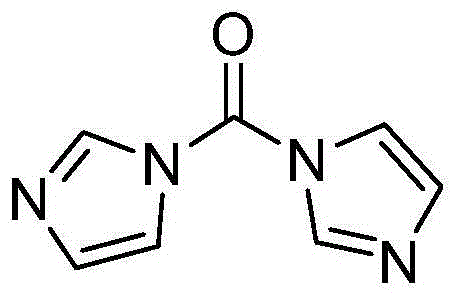
The methodology described in patent CN103724336A elegantly circumvents these deficiencies by employing CDI or DCC as the coupling agents directly with the carboxylic acid. This strategy eliminates the need for the corrosive thionyl chloride activation step entirely, allowing the reaction to proceed under much milder and safer conditions. The use of CDI, in particular, activates the carboxylic acid to form a reactive acyl imidazole intermediate, which then undergoes nucleophilic attack by the amine group of the morpholinone derivative. This pathway not only simplifies the workflow by removing a distinct activation stage but also significantly enhances the atom economy of the process. Moreover, the patent highlights the critical discovery that using specific salt forms of the amine precursor, particularly the L-mandelate salt, can drastically boost reaction yields to levels far exceeding those of conventional techniques, representing a major leap forward in cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into CDI-Mediated Amide Coupling
The core of this innovative synthesis lies in the efficient activation of the carboxylic acid moiety of 5-chlorothiophene-2-carboxylic acid. When N,N'-carbonyldiimidazole (CDI) is introduced to the reaction mixture, typically in a polar aprotic solvent like DMF, it reacts with the carboxylic acid to release carbon dioxide and imidazole, generating an N-acylimidazole intermediate. This intermediate is highly electrophilic and stable enough to be isolated if necessary, yet reactive enough to couple readily with amines at room temperature. The structure of CDI, characterized by two imidazole rings linked by a carbonyl group, makes it an ideal reagent for this transformation as the leaving group (imidazole) is non-nucleophilic and easily removed during workup.
Following the formation of the activated species, the amine component attacks the carbonyl carbon, displacing the imidazole group to form the desired amide bond found in Rivaroxaban. A crucial mechanistic insight provided by the patent is the influence of the amine salt form on this nucleophilic attack. While the free base or hydrochloride salt performs adequately, the L-mandelate salt of the morpholinone intermediate demonstrates superior reactivity. It is hypothesized that the mandelate counter-ion may facilitate a more favorable solubility profile or a specific transition state geometry that accelerates the coupling kinetics without requiring elevated temperatures. This results in a cleaner reaction profile with fewer side products, directly addressing the purity concerns of R&D directors who must ensure strict control over the impurity spectrum of the final API.
How to Synthesize Rivaroxaban Efficiently
The optimized protocol derived from this patent offers a straightforward, scalable procedure for producing high-purity Rivaroxaban. The process begins with the activation of 5-chlorothiophene-2-carboxylic acid in dimethylformamide (DMF) using a stoichiometric amount of CDI. After a brief stirring period to ensure complete activation, the amine component is added. The reaction is allowed to proceed at ambient temperature, eliminating the energy costs associated with heating or cooling. Upon completion, the product is precipitated by pouring the reaction mixture into water, a simple and effective isolation technique that avoids complex extraction procedures. For a detailed, step-by-step guide including precise stoichiometry and workup parameters, please refer to the standardized synthesis instructions below.
- Dissolve 5-chlorothiophene-2-carboxylic acid in a suitable solvent such as DMF and add N,N'-carbonyldiimidazole (CDI) to activate the acid, stirring at room temperature for approximately 1 hour.
- Introduce the amine component, specifically 4-[4-[(5S)-5-(aminomethyl)-2-oxo-3-oxazolidinyl]phenyl]-3-morpholinone or its L-mandelate salt, to the activated acid solution.
- Continue stirring the reaction mixture at room temperature for 12 to 48 hours, then pour into water to precipitate the product, followed by filtration and drying to obtain pure Rivaroxaban.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis method outlined in CN103724336A translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage is the substantial reduction in raw material costs. By replacing the expensive EDCI with the more economical CDI or DCC, manufacturers can achieve significant savings on reagent spend per kilogram of API produced. Additionally, the elimination of thionyl chloride removes the need for costly corrosion-resistant infrastructure and reduces the overhead associated with hazardous waste disposal and emission control systems. This shift towards safer, less corrosive chemistry aligns perfectly with modern sustainability goals and reduces the regulatory burden on the manufacturing facility.
- Cost Reduction in Manufacturing: The switch to CDI/DCC coupling agents fundamentally alters the cost structure of Rivaroxaban production. Unlike EDCI, which is a premium-priced reagent, CDI is widely available and cost-effective. Furthermore, the ability to achieve high molar yields (up to 92% with the L-mandelate salt) means that less starting material is wasted, maximizing the output from every batch. This efficiency gain, combined with the removal of the acid chloride formation step, streamlines the entire production timeline, leading to lower operational expenditures and a more competitive price point for the final high-purity pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Relying on corrosive and hazardous reagents like thionyl chloride introduces volatility into the supply chain due to strict transportation regulations and potential supply disruptions. By utilizing stable, solid reagents like CDI and carboxylic acids, the manufacturing process becomes more resilient. These materials are easier to store, handle, and transport, ensuring a consistent flow of production inputs. This reliability is critical for maintaining continuous manufacturing schedules and meeting the rigorous delivery deadlines demanded by global pharmaceutical clients, effectively reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The gentle reaction conditions of this novel method—operating at room temperature without the evolution of toxic gases—make it exceptionally well-suited for large-scale production. The absence of hydrogen chloride gas generation simplifies the engineering requirements for reactor vessels and venting systems, allowing for easier scale-up from pilot plant to commercial tonnage. From an environmental perspective, the process generates less hazardous waste and avoids the release of corrosive fumes, facilitating compliance with increasingly stringent environmental protection laws and enhancing the corporate social responsibility profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on yield optimization, reagent selection, and process safety for technical stakeholders evaluating this technology for adoption.
Q: Why is N,N'-carbonyldiimidazole (CDI) preferred over EDCI for Rivaroxaban synthesis?
A: CDI is preferred because it is significantly more cost-effective than EDCI and avoids the formation of difficult-to-remove urea byproducts. Furthermore, the patent data indicates that CDI provides superior molar yields (up to 92% with specific salts) compared to the lower yields associated with EDCI methods.
Q: What is the advantage of using the L-mandelate salt of the morpholinone intermediate?
A: Utilizing the L-mandelate salt of the morpholinone intermediate dramatically improves the reaction outcome. Screening data reveals that this specific salt form achieves a molar yield of 92%, which is substantially higher than the free base or other salt forms like hydrochloride or tartrate, ensuring better process efficiency.
Q: Does this method avoid the use of corrosive reagents?
A: Yes, this method eliminates the need for thionyl chloride (SOCl2), which is traditionally used to convert carboxylic acids to acid chlorides. By avoiding SOCl2, the process prevents the generation of corrosive hydrogen chloride gas, thereby reducing equipment corrosion risks and simplifying waste gas treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivaroxaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensively analyzed the synthetic pathways described in patent CN103724336A and possesses the know-how to implement this efficient CDI-mediated coupling strategy at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Rivaroxaban intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthesis technology for your anticoagulant portfolio. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, taking the first step towards a more efficient and reliable supply chain for your critical pharmaceutical ingredients.
