Advanced PEG-Catalyzed Synthesis of Methyl Ionone Intermediates for Commercial Scale Production
Introduction to Advanced Methyl Ionone Manufacturing
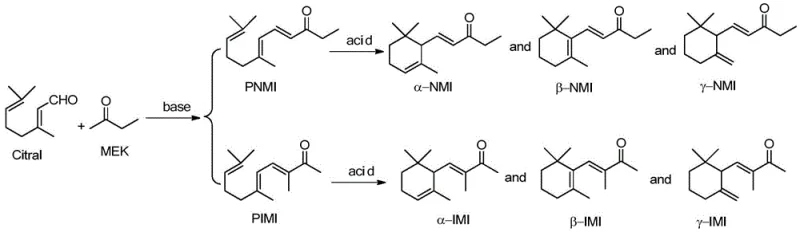
The synthesis of methyl ionone, a high-value fragrance ingredient known for its complex floral and woody notes, has long presented significant challenges in regioselectivity and environmental sustainability. As detailed in patent CN106748696B, traditional manufacturing routes often struggle with the inherent chemical bias of butanone (MEK) during Aldol condensation, leading to excessive formation of unwanted isomers and substantial chemical waste. This new technical disclosure introduces a transformative approach utilizing Polyethylene Glycol (PEG) as a dual-function solvent and phase-transfer mediator, coupled with metal hydroxides, to fundamentally alter the reaction microenvironment. By leveraging this unique PEG-M(OH)x catalytic system, manufacturers can achieve a dramatic shift in selectivity towards the commercially critical pseudoisomethyl ionone isomer, while simultaneously enabling a closed-loop catalyst recycling process. This innovation addresses the critical pain points of modern fine chemical production, offering a pathway to high-purity intermediates with drastically reduced ecological footprints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of methyl ionone relies heavily on standard base-catalyzed Aldol condensations in solvents like methanol or ethanol, often utilizing potassium hydroxide. While these methods are chemically feasible, they suffer from severe thermodynamic and kinetic limitations that hinder commercial efficiency. In conventional polar protic solvents, the acidity of the protons at the 1-position of butanone is significantly higher than those at the 3-position due to lower steric hindrance and better stabilization of the resulting enolate. Consequently, the reaction overwhelmingly favors the formation of pseudon-methyl ionone (PNMI), often exceeding 90% selectivity, which is not the desired precursor for the most valuable alpha-isomethyl ionone aroma profile. Furthermore, these traditional processes generate massive quantities of saline wastewater during the neutralization and workup phases, creating a heavy burden on waste treatment facilities and inflating operational costs through expensive effluent management protocols.
The Novel Approach
The methodology described in the patent represents a paradigm shift by replacing volatile organic solvents with a tunable PEG-based system that actively manipulates molecular interactions. Instead of passively dissolving reactants, the PEG solvent acts as a supramolecular host that coordinates with metal cations and engages in hydrogen bonding with the carbonyl oxygen of butanone. This specific interaction effectively masks the reactivity of the 1-position protons through increased steric bulk while simultaneously enhancing the nucleophilicity of the 3-position carbanions. The result is a remarkable inversion of natural selectivity, maintaining the pseudoisomethyl ionone (PIMI) ratio between 68% and 79%, which is essential for downstream cyclization efficiency. Additionally, the physical properties of PEG allow for a biphasic workup using n-hexane, where the product partitions into the organic layer while the catalyst remains in the PEG phase, enabling direct reuse and eliminating the need for energy-intensive solvent recovery distillation columns.
Mechanistic Insights into PEG-Mediated Regioselective Aldol Condensation
To fully appreciate the technical sophistication of this process, one must examine the molecular dynamics occurring within the PEG solvent cage. The core challenge in methyl ionone synthesis lies in the structural asymmetry of butanone, which possesses two distinct sets of alpha-protons capable of nucleophilic attack on the aldehyde carbonyl of citral. Under standard conditions, the kinetic product derived from the less hindered methyl group (position 1) dominates. However, the introduction of PEG creates a unique solvation shell around the metal hydroxide catalyst. The ether oxygens in the PEG chain chelate the metal cations, effectively increasing the "nakedness" and basicity of the hydroxide anions, which facilitates the deprotonation of the more sterically demanding methylene group at position 3.
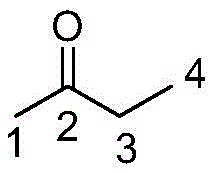
Furthermore, the terminal hydroxyl groups of the PEG polymer form intermolecular hydrogen bonds with the ketone oxygen of the butanone substrate. This interaction imposes a conformational constraint that effectively increases the steric hindrance around the 1-position methyl group, disfavoring enolate formation at that site. Simultaneously, this solvation effect stabilizes the transition state for the attack at the 3-position, leading to the preferential formation of the pseudoiso-isomer. This precise control over the impurity profile is critical for R&D directors, as it simplifies the downstream purification train. By minimizing the generation of the wrong isomer at the source, the process reduces the load on rectification columns and ensures that the subsequent acid-catalyzed cyclization step proceeds with higher fidelity towards the target alpha-isomethyl ionone, ultimately yielding a fragrance ingredient with superior olfactory characteristics and consistency.
How to Synthesize Pseudoisomethyl Ionone Efficiently
The implementation of this PEG-catalyzed route requires careful control of reaction parameters to maximize the benefits of the supramolecular solvent effects. The process begins with the preparation of the catalytic phase by dissolving a metal hydroxide, such as potassium hydroxide or cesium hydroxide, into PEG with a molecular weight between 200 and 600. Citral and butanone are then introduced, typically with the citral added dropwise to maintain a low concentration of aldehyde and prevent self-condensation side reactions. The reaction is conducted at mild temperatures, preferably between 10°C and 20°C, to further enhance kinetic selectivity. Following the condensation, the mixture undergoes a liquid-liquid extraction with n-hexane. This step is pivotal as it separates the lipophilic pseudo-methyl ionone product from the hydrophilic PEG-catalyst phase. The detailed standardized synthetic steps, including specific molar ratios, stirring rates, and extraction protocols, are outlined in the guide below.
- Conduct Aldol condensation between citral and butanone using PEG as solvent and metal hydroxide as catalyst at 10-20°C to maximize pseudoisomethyl ionone selectivity.
- Separate the product layer using n-hexane extraction, allowing the PEG-catalyst phase to be recovered and reused for subsequent batches.
- Perform acid-catalyzed cyclization of the pseudo-methyl ionone intermediate using phosphoric acid in n-hexane at 60-70°C to obtain high-purity methyl ionone isomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this PEG-mediated technology offers compelling economic and logistical advantages that extend far beyond simple yield improvements. The ability to recycle the catalytic system represents a fundamental change in the cost structure of methyl ionone manufacturing. In traditional batch processes, the base catalyst is consumed and neutralized in every run, generating stoichiometric amounts of salt waste that require disposal. In contrast, the PEG-M(OH)x system functions as a reusable liquid catalyst bed. Since the catalyst remains in the PEG phase after product extraction, it can be pumped directly back into the reactor for the next batch. This eliminates the recurring cost of purchasing fresh base catalyst and drastically reduces the volume of hazardous waste requiring treatment, leading to substantial operational expenditure savings over the lifecycle of the plant.
- Cost Reduction in Manufacturing: The elimination of solvent recovery distillation for the reaction medium is a major driver of cost efficiency. Traditional methods require energy-intensive stripping of solvents like methanol to isolate the product and recover the solvent. In this novel process, the product is simply extracted into n-hexane, which is easier to recover due to its lower boiling point and immiscibility with the reaction medium. Furthermore, the high selectivity for the desired isomer means that less raw material is wasted on producing off-spec byproducts that must be separated and discarded. This atom economy translates directly into a lower cost per kilogram of finished fragrance ingredient, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The robustness of the PEG catalytic system contributes significantly to supply continuity. Because the catalyst can be reused for multiple cycles without significant degradation in performance, the manufacturing process is less susceptible to fluctuations in the supply chain of auxiliary chemicals. There is no need to constantly source and qualify new batches of specialized catalysts or manage the logistics of large volumes of neutralizing acids. The simplified workup procedure also reduces the turnaround time between batches, allowing for higher throughput in existing reactor trains. This increased operational flexibility ensures that production schedules can be met consistently, even during periods of high demand or raw material volatility.
- Scalability and Environmental Compliance: From an environmental compliance perspective, this technology aligns perfectly with increasingly stringent global regulations regarding industrial effluent. The reduction in saline wastewater generation simplifies the permitting process for new manufacturing lines and reduces the risk of regulatory fines. The use of PEG, a non-volatile and relatively benign solvent, also improves workplace safety by reducing exposure to volatile organic compounds (VOCs). The process is inherently scalable; the phase separation mechanics work equally well in pilot plants and multi-ton commercial reactors, ensuring that the efficiency gains observed in the laboratory translate seamlessly to full-scale industrial production without the need for complex re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this PEG-catalyzed synthesis route. These insights are derived directly from the experimental data and mechanistic explanations provided in the patent literature, offering clarity on how this method compares to established industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this greener and more efficient manufacturing protocol for their specific production needs.
Q: How does the PEG solvent system improve selectivity for pseudoisomethyl ionone?
A: The PEG solvent remodels the microenvironment around the metal cations and forms hydrogen bonds with the ketone group of butanone. This interaction increases steric hindrance at the 1-position of butanone while enhancing the activity of the 3-position, thereby shifting the regioselectivity towards the desired pseudoisomethyl ionone isomer (68-79% ratio) compared to traditional solvents.
Q: Can the catalytic system be recycled in this methyl ionone production process?
A: Yes, a key advantage of this method is the immiscibility of the PEG-catalyst phase with the n-hexane extraction solvent. After the reaction, the product is extracted into the organic layer, leaving the alkali-containing PEG solution behind. This PEG phase can be directly reused for multiple cycles without significant loss in catalytic activity or selectivity.
Q: What are the typical yields for the cyclization step in this patented method?
A: The patent discloses that using a phosphoric acid-n-hexane catalytic system for the cyclization of pseudo-methyl ionone results in significantly improved yields, typically ranging from 92% to 95%, with the valuable alpha-isomethyl ionone isomer maintained at a high content of 60-80%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl Ionone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO specializing in complex fine chemical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped to handle the specific requirements of PEG-mediated reactions, including precise temperature control for selective Aldol condensations and efficient biphasic extraction systems. We are committed to delivering high-purity methyl ionone and its intermediates that meet stringent purity specifications, supported by our rigorous QC labs which utilize state-of-the-art analytical instrumentation to verify isomer ratios and impurity profiles.
We invite you to collaborate with us to leverage this innovative synthesis route for your fragrance portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how the recyclability of the PEG catalyst can impact your bottom line. Please contact our technical procurement team today to request specific COA data for our methyl ionone grades and to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a sustainable and cost-effective supply chain for your critical fragrance ingredients.
