Optimizing Methyl Ionone Production: A Technical Breakdown of Recyclable PEG Catalysis
The global demand for high-quality fragrance ingredients continues to drive innovation in synthetic organic chemistry, particularly for complex terpenoids like methyl ionone. Patent CN106748696B introduces a transformative preparation method that addresses longstanding inefficiencies in the aldol condensation of citral and butanone. Traditional synthesis routes often suffer from poor regioselectivity and excessive waste generation, but this novel approach utilizes a Polyethylene Glycol (PEG) solvent system coupled with metal hydroxide catalysts to fundamentally alter the reaction microenvironment. By leveraging the unique solvation properties of PEG, the process not only achieves superior yields of the critical pseudo-isomethyl ionone intermediate but also enables the recycling of the catalytic phase. This technical advancement represents a significant leap forward for manufacturers seeking to optimize both the economic and environmental footprint of their fragrance production lines.
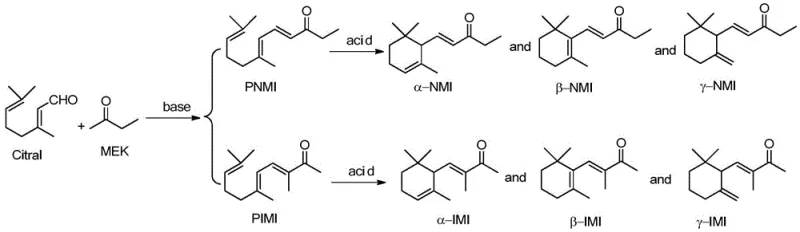
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of methyl ionone has relied heavily on homogeneous base catalysis in volatile organic solvents such as methanol or ethanol. These conventional pathways are plagued by inherent thermodynamic and kinetic limitations that result in suboptimal isomer distributions. Specifically, the nucleophilic attack of butanone on citral tends to favor the less valuable pseudo-n-methyl ionone isomer due to lower steric hindrance at the C1 position of the ketone. Furthermore, traditional processes generate substantial quantities of saline wastewater during the neutralization and workup phases, creating a heavy burden on waste treatment facilities. The inability to recover the base catalyst efficiently means that every batch requires fresh reagents, driving up operational expenditures and complicating supply chain logistics for large-scale producers.
The Novel Approach
The methodology disclosed in the patent circumvents these issues by replacing traditional alcoholic solvents with high-molecular-weight PEG, creating a distinct biphasic reaction environment. This system utilizes metal hydroxides like potassium hydroxide or cesium hydroxide dissolved directly in the PEG matrix, which acts as both solvent and phase transfer mediator. The innovation lies in the post-reaction separation strategy: because PEG is immiscible with non-polar solvents like n-hexane, the product can be extracted cleanly while the catalyst-rich PEG phase remains intact for reuse. This eliminates the need for energy-intensive solvent recovery distillation and neutralization steps associated with prior art, offering a streamlined pathway that maintains high selectivity for the desired pseudo-isomethyl ionone isomer throughout repeated cycles.
Mechanistic Insights into PEG-Mediated Regioselective Aldol Condensation
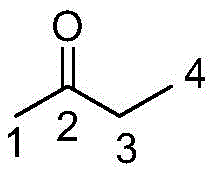
The core chemical breakthrough of this process is the manipulation of regioselectivity during the initial aldol condensation step. In standard conditions, the protons at the C1 position of butanone are more acidic and less sterically hindered than those at the C3 position, leading to a predominance of the linear pseudo-n-methyl ionone. However, the introduction of PEG fundamentally reshapes the solvation shell around the reactants. The ether oxygen atoms within the PEG chain coordinate with the metal cations of the base, effectively increasing the localized basicity and stabilizing the enolate formed at the more hindered C3 position. Simultaneously, the terminal hydroxyl groups of the PEG polymer form hydrogen bonds with the carbonyl oxygen of the butanone. This specific interaction increases the steric bulk around the C1 position, kinetically disfavoring attack there and directing the nucleophilic addition towards the C3 site to form the branched pseudo-isomethyl ionone precursor.
Beyond mere selectivity enhancement, this mechanism provides robust control over impurity profiles, which is critical for downstream purification. The stabilization of the transition state by the PEG matrix ensures that side reactions, such as polymerization of the sensitive citral aldehyde or over-condensation, are minimized. The resulting reaction mixture is cleaner, containing a higher concentration of the target pseudo-isomethyl ionone isomer (maintained at 68-79% according to patent data). This high initial purity simplifies the subsequent acid-catalyzed cyclization step, where the pseudo-isomethyl ionone is converted into the final alpha-isomethyl ionone. The consistency of the intermediate quality ensures that the final cyclization yield remains high, typically exceeding 90%, with the valuable alpha-isomer constituting the majority of the product mixture.
How to Synthesize Methyl Ionone Efficiently
The synthesis protocol outlined in the patent offers a reproducible framework for generating high-purity methyl ionone suitable for fine fragrance applications. The process begins with the preparation of the catalytic solution by dissolving a metal hydroxide, such as KOH or CsOH, into PEG-400 or PEG-600 under stirring. Once the catalyst is fully dispersed, butanone is added, followed by the controlled dropwise addition of citral to manage the exotherm and maintain selectivity. Following the condensation reaction, the mixture undergoes a liquid-liquid extraction using n-hexane, which separates the organic product layer from the aqueous-compatible PEG catalyst layer. The detailed standardized synthesis steps, including specific temperature ramps, molar ratios, and workup procedures, are provided in the guide below.
- Perform Aldol condensation of Citral and Butanone using PEG as solvent and Metal Hydroxide as catalyst at 0-60°C.
- Separate the product layer using n-hexane extraction, allowing the PEG-catalyst phase to be recovered and reused.
- Conduct acid-catalyzed cyclization of the pseudo-methyl ionone intermediate using phosphoric acid in n-hexane to yield methyl ionone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this PEG-based catalytic system offers tangible strategic benefits beyond simple yield improvements. The most significant advantage is the drastic reduction in variable costs associated with catalyst consumption and waste disposal. By enabling the repeated recycling of the PEG-metal hydroxide phase, the process decouples production volume from raw material usage for the catalyst, leading to substantial long-term savings. Additionally, the elimination of large volumes of saline wastewater reduces the load on environmental compliance systems, lowering the overhead costs related to effluent treatment and regulatory reporting. This efficiency translates directly into a more competitive cost structure for the final methyl ionone product.
- Cost Reduction in Manufacturing: The ability to recycle the catalytic system multiple times without significant degradation in performance removes the recurring cost of purchasing fresh base catalysts for every batch. Furthermore, the use of PEG eliminates the need for expensive solvent recovery units required for volatile alcohols, as the product is simply extracted with n-hexane. This simplification of the unit operations reduces energy consumption and capital expenditure on distillation columns, resulting in a leaner and more cost-effective manufacturing process that enhances overall margin potential.
- Enhanced Supply Chain Reliability: The robustness of the PEG-catalyst system contributes to greater supply chain stability by reducing the risk of batch failures due to catalyst variability. Since the catalyst phase is retained and reused, the process becomes less sensitive to fluctuations in the quality of fresh reagent inputs. Moreover, the high selectivity for the pseudo-isomethyl ionone intermediate ensures a consistent feed for the cyclization step, minimizing bottlenecks in production scheduling. This reliability allows for more accurate forecasting and inventory management, ensuring that delivery commitments to downstream fragrance houses can be met consistently.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns with green chemistry principles by minimizing waste generation at the source. The biphasic nature of the reaction prevents the formation of emulsions that are difficult to treat, facilitating cleaner separation and reducing the volume of hazardous waste requiring disposal. This environmental profile makes the technology highly scalable, as it avoids the permitting hurdles often associated with high-saline wastewater discharge. Consequently, manufacturers can expand capacity with greater ease, secure in the knowledge that the process meets stringent international environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this technology for commercial production. The answers are derived directly from the experimental data and mechanistic explanations provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does the PEG solvent system improve isomer selectivity in methyl ionone synthesis?
A: The PEG solvent remodels the microenvironment around the butanone molecule. Its ether chains wrap metal ions to enhance alkalinity, while terminal hydroxyl groups form hydrogen bonds with the ketone group. This steric and electronic modulation favors nucleophilic attack at the C3 position of butanone over the C1 position, significantly increasing the ratio of the valuable pseudo-isomethyl ionone isomer.
Q: Can the catalytic system be recycled in this preparation method?
A: Yes, a key advantage of this patented process is the recyclability of the PEG-Metal Hydroxide catalytic system. Since PEG is immiscible with the n-hexane extraction solvent, the catalyst remains in the bottom phase after product extraction. This phase can be recovered and reused for multiple cycles without significant loss in catalytic activity or yield, drastically reducing raw material costs.
Q: What are the typical yields and purity levels achievable with this method?
A: The process demonstrates high efficiency, with the condensation step achieving yields of 91-95% and maintaining a pseudo-isomethyl ionone proportion of 68-79%. The subsequent cyclization step yields 92-95% methyl ionone, with the target alpha-isomethyl ionone isomer maintained at 60-80%, resulting in a final product purity of up to 99% after rectification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl Ionone Supplier
As the fragrance and flavor industry demands increasingly sophisticated intermediates, partnering with a manufacturer that possesses deep technical expertise is crucial. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies like the PEG-mediated synthesis described above to deliver superior products. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of methyl ionone meets the exacting standards required for high-end perfume formulations.
We invite you to collaborate with us to optimize your supply chain for methyl ionone and related fragrance intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our efficient processes can reduce your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, high-quality supply of this critical ingredient.
