Advanced Methylprednisolone Manufacturing: Overcoming Traditional Iodine Limitations for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic pathways for critical corticosteroids like methylprednisolone, a potent anti-inflammatory agent widely used in treating autoimmune disorders and allergic reactions. Patent CN101418029B introduces a transformative methodology that fundamentally alters the economic and operational landscape of producing this high-value active pharmaceutical ingredient (API). By shifting away from traditional iodine-based halogenation towards a catalytic bromination strategy, this technology addresses long-standing issues regarding reagent stability, cost efficiency, and environmental impact. The core innovation lies in the strategic use of ammonium salt catalysts to drive the initial functionalization of the steroid backbone, followed by a highly efficient phase-transfer catalyzed substitution. This report analyzes the technical depth of this patent to demonstrate how it serves as a blueprint for reliable methylprednisolone supplier networks aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of methylprednisolone has been plagued by reliance on expensive and hazardous halogenating agents, particularly iodine-based reagents. Conventional routes, such as those referenced in prior art like GB 2318790, often involve multi-step sequences including enzymatic hydroxylation and complex chemical dehydrogenation that result in low overall yields. These traditional processes frequently require harsh reaction conditions and the use of toxic reagents that pose significant safety risks to operators and complicate waste management protocols. Furthermore, the intermediates generated in these older pathways are often chemically unstable, necessitating immediate downstream processing which restricts production scheduling flexibility. The accumulation of toxic by-products and the difficulty in recycling solvents further exacerbate the environmental burden, making cost reduction in API manufacturing increasingly difficult under modern regulatory frameworks.
The Novel Approach
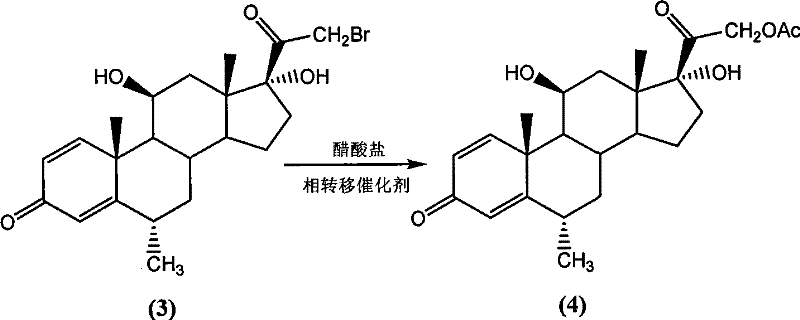
In stark contrast, the methodology disclosed in CN101418029B leverages a streamlined three-step sequence that prioritizes atom economy and operational simplicity. The process initiates with the bromination of 6-alpha-methyl-11-beta,17-alpha-dihydroxypregn-1,4-diene-3,20-diketone using affordable reagents like N-bromosuccinimide (NBS) rather than costly iodine. A pivotal advancement is the introduction of ammonium salts as catalysts, which dramatically enhances reaction kinetics and selectivity without requiring stoichiometric amounts of expensive additives. The subsequent substitution step utilizes a phase-transfer catalyst to efficiently convert the bromo-intermediate into the acetate derivative under mild thermal conditions. This approach not only simplifies the purification workflow but also ensures that the solvents employed can be effectively recovered and reused, thereby aligning with green chemistry principles while maintaining high throughput capabilities suitable for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ammonium Salt Catalyzed Bromination and PTC Substitution
The mechanistic elegance of this synthesis lies in the dual-catalyst system that governs the transformation of the steroid skeleton. In the first stage, the ammonium salt catalyst, such as ammonium acetate or ammonium formiate, plays a crucial role in activating the brominating reagent. Without this catalytic species, the reaction between the ketone substrate and N-bromosuccinimide proceeds sluggishly or fails to initiate, leading to poor conversion rates. The ammonium ion likely facilitates the generation of the active brominating species or stabilizes the transition state during the electrophilic attack at the C-21 position. This results in the formation of the 21-bromo intermediate with exceptional purity, often exceeding 95% HPLC content directly from the reaction mixture. The stability of this bromo-intermediate is a key technical advantage, as it allows for isolation and storage, decoupling the upstream and downstream manufacturing units and providing a buffer against supply chain disruptions.
Following the bromination, the second critical transformation involves the nucleophilic substitution of the bromine atom with an acetate group. This step is mediated by a quaternary ammonium phase-transfer catalyst (PTC), which enables the reaction between the organic-soluble bromo-steroid and the inorganic acetate salt in a homogeneous-like environment. The PTC shuttles the acetate anion into the organic phase, significantly accelerating the reaction rate at moderate temperatures ranging from 50°C to 120°C. This mechanism minimizes side reactions such as elimination or over-hydrolysis, which are common pitfalls in steroid chemistry. The resulting 21-acetate intermediate is then subjected to mild alkaline hydrolysis to yield the final target molecule. This controlled hydrolysis ensures that the sensitive 11-beta and 17-alpha hydroxyl groups remain intact while selectively cleaving the ester bond, delivering high-purity methylprednisolone with minimal impurity profiles.
How to Synthesize Methylprednisolone Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the catalytic systems described. The process begins with the dissolution of the starting pregnane derivative in a suitable solvent such as tetrahydrofuran or ether, followed by the addition of the brominating agent and the catalytic ammonium salt. Maintaining the reaction temperature between 20°C and 100°C allows for optimal conversion within a timeframe of 0.5 to 24 hours. Once the 21-bromo intermediate is isolated, it is reacted with an acetate salt like potassium acetate in the presence of a phase-transfer catalyst such as tetrabutylammonium bromide. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that ensure reproducibility and safety.
- React 6-alpha-methyl-11-beta,17-alpha-dihydroxypregn-1,4-diene-3,20-diketone with a brominating reagent like NBS using an ammonium salt catalyst to form the 21-bromo intermediate.
- Perform nucleophilic substitution on the 21-bromo intermediate using an acetate salt and a phase-transfer catalyst to generate the 21-acetate derivative.
- Hydrolyze the 21-acetate product under mild alkaline conditions at low temperature to obtain the final high-purity methylprednisolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this bromination-based pathway offers substantial strategic benefits beyond mere technical feasibility. The primary driver for value creation is the replacement of high-cost iodine reagents with economically superior bromine sources, which directly impacts the bill of materials without compromising product quality. Additionally, the enhanced stability of the intermediates reduces the need for expedited shipping and specialized cold-chain logistics, allowing for more flexible inventory management strategies. The ability to recycle solvents and the reduction in hazardous waste generation also translate into lower disposal costs and simplified regulatory compliance, contributing to a more sustainable and resilient manufacturing operation.
- Cost Reduction in Manufacturing: The substitution of expensive iodine reagents with low-cost brominating agents like N-bromosuccinimide represents a direct material cost saving that scales linearly with production volume. Furthermore, the use of catalytic amounts of ammonium salts eliminates the need for stoichiometric quantities of expensive promoters, reducing the overall reagent consumption per kilogram of product. The high reaction yields reported in the patent minimize the loss of valuable starting materials, ensuring that the theoretical maximum output is approached in practical settings. By avoiding complex protection and deprotection steps required in other routes, the process reduces labor hours and utility consumption, leading to significant operational expenditure optimization.
- Enhanced Supply Chain Reliability: The chemical stability of the 21-bromo intermediate allows manufacturers to produce and stockpile this key building block during periods of low demand, creating a buffer that protects against upstream supply shocks. Since the reagents involved, such as ammonium acetate and common organic solvents, are commodity chemicals available from multiple global vendors, the risk of single-source dependency is drastically minimized. This diversification of the supply base ensures continuous production capability even when specific raw material markets experience volatility. The robustness of the process conditions also means that manufacturing can be transferred between different facilities with minimal requalification effort, enhancing overall network agility.
- Scalability and Environmental Compliance: The mild reaction conditions, operating at temperatures well below the decomposition thresholds of the steroid backbone, facilitate safe scale-up from pilot plants to multi-ton commercial reactors. The process generates significantly less toxic waste compared to traditional iodine-based methods, simplifying effluent treatment and reducing the environmental footprint of the facility. Solvent recovery systems can be easily integrated due to the volatility and compatibility of the chosen solvents, promoting a circular economy approach within the plant. These factors collectively lower the barrier to entry for large-scale production and ensure long-term viability in an increasingly regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide clarity on process capabilities. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this technology into their existing production lines.
Q: Why is the ammonium salt catalyst critical in the bromination step?
A: According to patent CN101418029B, the addition of catalytic amounts of ammonium salts (such as ammonium acetate or formiate) significantly improves reaction yield and selectivity. Without this catalyst, the reaction between the starting material and the brominating reagent shows little to no conversion or produces excessive by-products.
Q: How does this method improve supply chain stability compared to traditional routes?
A: Traditional methods often rely on expensive and unstable iodine reagents. This novel process utilizes low-cost, stable brominating reagents like N-bromosuccinimide. Furthermore, the key intermediate (21-bromo derivative) is structurally stable and can be stored for extended periods, reducing the risk of raw material degradation during logistics.
Q: What represents the primary environmental advantage of this synthesis route?
A: The process avoids the use of toxic iodine reagents and complex protection/deprotection sequences found in older methodologies. Additionally, the solvents used in the reaction steps are recyclable, and the overall 'three wastes' discharge is significantly reduced, facilitating easier compliance with environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methylprednisolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101418029B are fully realized in practice. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of methylprednisolone meets the highest international standards for potency and impurity control. Our commitment to technical excellence allows us to navigate the complexities of steroid chemistry with precision and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this innovative bromination route can be tailored to your specific supply requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your decision-making process and secure a stable supply of high-quality corticosteroid intermediates for your organization.
