Advanced Biological Dehydrogenation for High-Purity Steroid Pharmaceutical Intermediates Manufacturing
Advanced Biological Dehydrogenation for High-Purity Steroid Pharmaceutical Intermediates Manufacturing
The pharmaceutical industry continuously seeks innovative pathways to enhance the efficiency and safety of producing critical corticosteroid intermediates. Patent CN101760495B introduces a groundbreaking biological dehydrogenation preparation method for 6 alpha-methylprednisolone intermediates, utilizing a specialized Arthrobacter simplex strain to achieve superior transformation rates. This technology represents a significant leap forward in steroid synthesis, replacing hazardous chemical oxidants with a sustainable biocatalytic process that ensures high purity and operational safety. By leveraging microbial fermentation, manufacturers can overcome the limitations of traditional chemistry, achieving conversion rates that substantially exceed historical benchmarks while eliminating toxic heavy metal residues from the final product stream. As a reliable steroid pharmaceutical intermediates supplier, understanding these technological shifts is crucial for maintaining competitive advantage in the global market. The structural integrity of the target molecule, as depicted below, is preserved through mild enzymatic action rather than harsh chemical conditions.
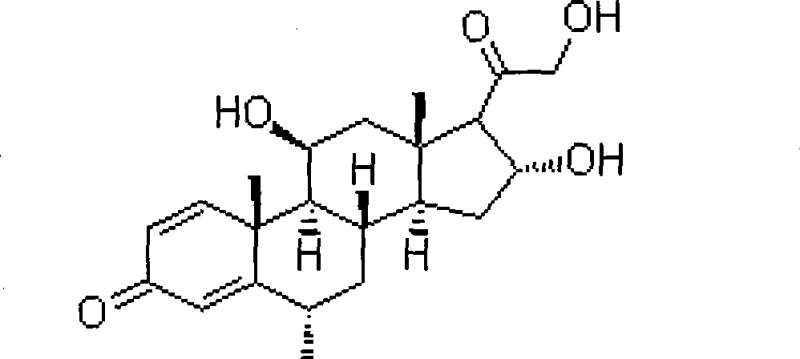
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the 1,2-position dehydrogenation of steroid drugs has relied heavily on Selenium Dioxide (SeO2) as the primary oxidizing agent, a practice fraught with significant safety and efficiency challenges. The use of SeO2 introduces severe toxicity risks, not only to the operating personnel but also to the environment, necessitating complex and costly waste treatment protocols to manage selenium-containing byproducts. Furthermore, the chemical yield associated with this traditional method is notoriously low, typically hovering around 55%, which drastically impacts the overall economics of the manufacturing process. Residual selenium impurities are exceptionally difficult to eliminate completely, often requiring additional purification steps that increase production time and reduce the final quality of the active pharmaceutical ingredient. These factors collectively create a bottleneck in cost reduction in pharmaceutical intermediates manufacturing, forcing producers to seek alternative, greener technologies that do not compromise on output quality or regulatory compliance.
The Novel Approach
In stark contrast to the hazardous chemical oxidation methods, the novel approach detailed in the patent utilizes a simple Arthrobacter biological dehydrogenation method that fundamentally transforms the production landscape. This biocatalytic route operates under mild conditions, typically between 30°C and 34°C, which significantly reduces energy consumption compared to high-temperature chemical reactions. The process achieves a remarkable biotransformation rate ranging from 70% to 90%, representing a substantial improvement in material efficiency and throughput capacity. By employing specific bacterial strains such as AS 1.754 or AS 1.94, the method ensures high specificity for the 1,2-double bond formation without affecting other sensitive functional groups on the steroid nucleus. This shift not only enhances the purity profile of the resulting intermediate but also simplifies the downstream processing requirements, making it an ideal solution for the commercial scale-up of complex steroid pharmaceutical intermediates.
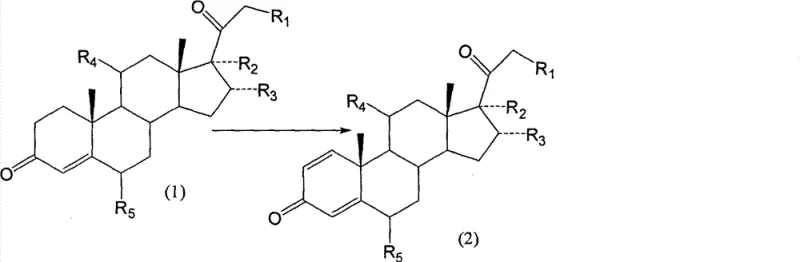
Mechanistic Insights into Arthrobacter Simplex-Catalyzed Dehydrogenation
The core of this technological advancement lies in the specific enzymatic activity of the Arthrobacter simplex strain, which facilitates the selective removal of hydrogen atoms from the steroid backbone. The mechanism involves a highly regulated microbial metabolism where the bacteria utilize the steroid substrate as part of their growth or maintenance cycle, specifically targeting the saturated bonds at the 1 and 2 positions of the A-ring. This enzymatic precision ensures that the stereochemistry of the molecule remains intact, preventing the formation of unwanted isomers that often plague chemical synthesis routes. The fermentation medium, composed of glucose, yeast extract, and corn steep liquor, provides the necessary nutrients to maintain high cell viability and enzymatic activity throughout the 36 to 72-hour reaction window. Understanding this biological mechanism is vital for R&D directors aiming to optimize fermentation parameters for maximum yield and minimal byproduct formation in large-scale bioreactors.
Impurity control is inherently superior in this biological system due to the absence of heavy metal catalysts and the specificity of the microbial enzymes. In chemical dehydrogenation, side reactions often lead to over-oxidation or degradation of the steroid skeleton, creating a complex impurity谱 that requires extensive chromatographic separation. However, the biological route produces a cleaner reaction profile, where the primary impurity is simply the unreacted starting material, which can be easily recycled or separated. This inherent selectivity contributes directly to achieving high-purity steroid pharmaceutical intermediates, reducing the burden on quality control laboratories and accelerating the release of batches for further synthesis. The elimination of selenium residues also means that the final product meets stricter international pharmacopoeia standards without the need for aggressive scavenging agents that might introduce new contaminants.
How to Synthesize 6 Alpha-Methylprednisolone Intermediate Efficiently
Implementing this biological dehydrogenation process requires a structured approach to fermentation management, starting from strain activation to final product isolation. The protocol begins with the preparation of the Arthrobacter simplex seed culture, progressing through slant, primary, and secondary stages to ensure a robust inoculum for the main fermentation tank. Once the biomass reaches the optimal density, the steroid substrate, dissolved in a compatible solvent like ethanol or DMF, is introduced into the fermenter under controlled aeration and agitation. The detailed standardized synthesis steps see the guide below, which outlines the precise temperature controls, feeding concentrations, and extraction procedures necessary to replicate the patent's success. Adhering to these parameters is essential for maintaining the consistency and reproducibility required for GMP-compliant manufacturing environments.
- Prepare Arthrobacter simplex strains via slant, primary, and secondary culture in specific glucose-yeast extract media at 30-34°C.
- Introduce the steroid substrate (Compound 1) into the fermenter at a concentration of 1-3% and maintain biotransformation for 36-72 hours.
- Terminate the reaction by heating to 70-90°C, extract the product using ethyl acetate, and refine to obtain the dehydrogenated Compound 2.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this biological method offers profound strategic benefits that extend beyond simple yield improvements. The elimination of toxic selenium reagents removes a major regulatory hurdle, simplifying the disposal of chemical waste and reducing the liability associated with handling hazardous materials. This shift aligns perfectly with global trends towards green chemistry and sustainable manufacturing, enhancing the corporate social responsibility profile of the production facility. Moreover, the higher conversion rates mean that less raw material is required to produce the same amount of finished intermediate, leading to substantial cost savings in raw material procurement. The milder reaction conditions also reduce energy consumption, contributing to a lower carbon footprint and reduced operational expenditures over the lifecycle of the product.
- Cost Reduction in Manufacturing: The removal of expensive and toxic selenium dioxide from the process equation eliminates the need for specialized containment equipment and complex residue removal steps. This simplification of the workflow reduces both capital expenditure on safety infrastructure and operational costs related to waste treatment and compliance monitoring. Additionally, the higher yield directly translates to better asset utilization, allowing existing fermentation tanks to produce more output without requiring physical expansion. These factors combine to create a more economically resilient supply chain capable of withstanding market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: Biological processes often offer greater scalability and flexibility compared to batch chemical reactions that rely on specific hazardous reagents which may face supply constraints. The use of common fermentation nutrients like glucose and yeast extract ensures that the supply chain is not vulnerable to the volatility of specialty chemical markets. Furthermore, the robustness of the Arthrobacter strains allows for consistent production cycles, reducing the lead time for high-purity steroid pharmaceutical intermediates and ensuring timely delivery to downstream API manufacturers. This reliability is critical for maintaining continuous production schedules in the fast-paced pharmaceutical industry.
- Scalability and Environmental Compliance: Scaling up a fermentation process is a well-understood engineering challenge, allowing for seamless transition from pilot scale to multi-ton commercial production. The aqueous nature of the fermentation broth simplifies the handling of large volumes compared to organic solvent-heavy chemical processes, reducing fire risks and ventilation requirements. From an environmental perspective, the biodegradable nature of the waste stream significantly lowers the ecological impact, ensuring compliance with increasingly stringent environmental protection regulations. This future-proofs the manufacturing site against potential regulatory crackdowns on chemical pollutants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biological dehydrogenation technology. These insights are derived directly from the patent specifications and are designed to clarify the operational feasibility and strategic value of adopting this method. Stakeholders often seek confirmation on the versatility of the strain and the specific quality advantages over traditional chemistry. The answers provided here reflect the documented capabilities of the Arthrobacter simplex system in producing high-quality steroid intermediates.
Q: How does this biological method improve safety compared to traditional SeO2 dehydrogenation?
A: Traditional methods use highly toxic Selenium Dioxide (SeO2), leaving hazardous residues that are difficult to remove. This biological process eliminates selenium entirely, ensuring a safer product profile and reducing environmental compliance burdens.
Q: What conversion rates can be expected using the Arthrobacter simplex strain?
A: According to patent data, the biotransformation rate consistently reaches between 70% and 90%, significantly outperforming the conventional chemical yield of approximately 55%.
Q: Is this process applicable to various steroid substrates beyond methylprednisolone?
A: Yes, the patent specifies versatility across multiple steroid intermediates, including various hydroxy-pregna-diones and epoxy derivatives, offering flexibility for diverse synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6 Alpha-Methylprednisolone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalytic processes in modern pharmaceutical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory techniques are successfully translated into robust industrial operations. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards. We understand the complexities involved in steroid synthesis and are equipped to handle the nuanced requirements of biological fermentation, from strain maintenance to downstream purification.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your project's efficiency and profitability. Let us be your partner in navigating the future of sustainable and high-performance chemical manufacturing.
