Advanced Chromium-Free Synthesis of Prednisolone for Commercial Scale-Up
Advanced Chromium-Free Synthesis of Prednisolone for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally compliant synthetic routes for critical corticosteroids like prednisolone. Patent CN103387595A introduces a transformative methodology for preparing prednisolone that addresses longstanding challenges in steroid functionalization. This technical insight report analyzes the proprietary four-step sequence involving double bond bromination, reductive debromination, secondary bromination, and hydrolysis. Unlike traditional biotransformation or chromium-dependent chemical routes, this novel approach leverages zinc-mediated reduction to achieve high purity and yield while eliminating heavy metal contamination risks. For global procurement and R&D teams, understanding this pathway is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering high-quality active ingredients with minimized regulatory friction.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of prednisolone has relied on methods fraught with inefficiencies and regulatory hurdles. One prevalent route involves the biological dehydrogenation of hydrocortisone using Arthrobacter simplex; however, this biotransformation method suffers from low transformation efficiency, typically capped at no more than 80%, and requires complex extraction and purification protocols that extend production cycles. Another chemical route utilizes prednisone as a raw material, involving condensation with aminoguanidine hydrochloride and reduction with lithium aluminum hydride, but the high cost of prednisone raw materials makes this economically unviable for cost-sensitive markets. Furthermore, prior art such as Chinese patent CN200710061258.2 employs chromium reagents for reduction steps, which inevitably leads to hazardous heavy-metal residues that are difficult to remove and pose significant environmental and safety compliance issues for modern manufacturing facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN103387595A offers a streamlined chemical solution that bypasses these critical bottlenecks. By selecting 17α-hydroxy-1,4,9(11)-pregnatriene-3,20-dione (Compound I) as the starting material, the process executes a precise sequence of bromination and zinc-mediated reduction. This strategy effectively avoids the use of expensive iodine reagents or unstable intermediates that require nitrogen protection and dark storage. The elimination of chromium reagents is a pivotal advancement, as it removes the need for expensive heavy metal scavenging steps and simplifies the waste treatment profile. Consequently, this route provides a foundation for cost reduction in pharmaceutical intermediates manufacturing by utilizing abundant reagents like zinc powder and N-bromosuccinimide (NBS) while ensuring the final product meets stringent purity specifications without complex chromatographic purification.
Mechanistic Insights into Zinc-Mediated Reductive Debromination
The core innovation of this synthesis lies in the selective reductive debromination step, which restores the critical delta-1 double bond without affecting the sensitive carbonyl functionalities at positions 3 and 20. In the first stage, Compound I undergoes bromination using NBS in a mixed solvent system of acetone and acetic acid at controlled temperatures between 0°C and 10°C. This generates the dibromo intermediate (Compound II) with high regioselectivity. Subsequently, the addition of activated zinc powder in an ether solvent facilitates the reductive elimination of the bromine atoms. The mechanism relies on the single-electron transfer capability of zinc, which cleaves the carbon-bromine bonds to reform the alkene. Crucially, the reaction conditions are tuned to ensure that the ketone groups remain intact, preventing the formation of alcohol by-products that would complicate downstream purification.
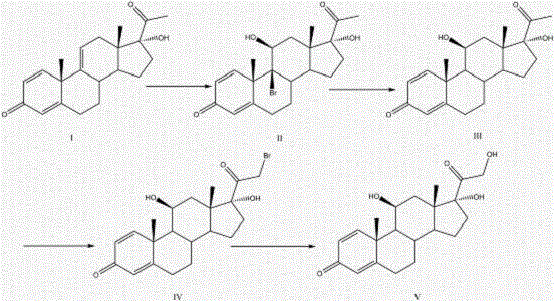
Following the reduction, the resulting Compound III undergoes a second bromination under acidic conditions using elemental bromine and a catalytic amount of aluminum chloride (AlCl3). This step introduces a bromine atom at the alpha-position relative to the carbonyl, setting up the molecule for the final hydrolysis. The use of a Lewis acid catalyst accelerates the reaction at low temperatures (-5°C to 0°C), minimizing side reactions such as polysubstitution. The final hydrolysis step, conducted in a mixture of sodium hydroxide, acetone, and methanol, displaces the bromine with a hydroxyl group to yield the target prednisolone (Compound V). This mechanistic precision ensures that impurity profiles are tightly controlled, making the process highly suitable for the commercial scale-up of complex hormonal intermediates where structural integrity is paramount.
How to Synthesize Prednisolone Efficiently
The operational simplicity of this route is a key driver for its industrial adoption. The synthesis begins with the dissolution of the starting triene in acetone and acetic acid, followed by the slow addition of NBS to manage exothermicity. After the initial bromination, the solvent is recovered, and the crude intermediate is subjected to zinc reduction in ether. The reaction progress is monitored via TLC, and upon completion, the mixture is treated with acid to consume excess zinc. This streamlined workflow minimizes unit operations and reduces the overall processing time compared to multi-step biotransformation sequences.
- Bromination of the starting triene compound using N-bromosuccinimide (NBS) in acetone and acetic acid at 0-10°C to form the dibromo intermediate.
- Reductive debromination using activated zinc powder in ether at room temperature to restore the double bond without reducing carbonyl groups.
- Secondary bromination using elemental bromine with catalytic aluminum chloride in ether at -5 to 0°C, followed by alkaline hydrolysis to yield prednisolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this zinc-based chemistry represents a strategic opportunity to optimize both cost structures and supply reliability. The replacement of chromium reagents with zinc powder not only mitigates environmental liability but also drastically simplifies the supply chain for raw materials, as zinc is a globally abundant and inexpensive commodity. Furthermore, the avoidance of unstable iodine intermediates eliminates the need for specialized storage conditions such as nitrogen blanketing and light protection, thereby reducing infrastructure costs and inventory risks. The high atom economy and quantitative nature of the transformation steps mean that less raw material is wasted, directly contributing to substantial cost savings in the final API production.
- Cost Reduction in Manufacturing: The elimination of heavy metal catalysts removes the necessity for expensive purification technologies like ion exchange or specialized scavenger resins, which are typically required to meet residual metal limits in pharmaceuticals. Additionally, the use of common solvents like acetone and ether allows for efficient solvent recovery and recycling loops, further driving down the variable cost per kilogram. By simplifying the workup to a basic pH adjustment and filtration, the process reduces labor hours and energy consumption associated with distillation and chromatography, leading to a more competitive pricing model for bulk pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Dependence on biotransformation often introduces variability due to biological factors, whereas this purely chemical route offers consistent batch-to-b reproducibility. The robustness of the zinc reduction step ensures that production schedules are not disrupted by fermentation failures or enzyme stability issues. Moreover, the starting materials and reagents are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. This reliability is critical for maintaining continuous supply lines for high-purity hormones required in just-in-time manufacturing environments.
- Scalability and Environmental Compliance: The process is designed for scalability, with reaction conditions that can be easily managed in standard stainless steel reactors without requiring exotic materials of construction. The waste stream is significantly cleaner due to the absence of chromium, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The ability to perform the synthesis in fewer steps with high yields means that facility throughput can be maximized, allowing manufacturers to respond rapidly to market demand fluctuations without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, impurity control, and operational parameters. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer. This transparency ensures that all stakeholders have a clear understanding of the process capabilities before initiating partnership discussions.
Q: Why is the zinc-based reduction method preferred over chromium-based methods for prednisolone?
A: The zinc-based method eliminates the risk of toxic heavy metal residues (chromium) which require complex and costly removal steps to meet pharmaceutical regulatory standards. Additionally, zinc powder is significantly cheaper and easier to handle than chromium reagents.
Q: What is the overall yield advantage of this synthetic route?
A: The patent data indicates a total recovery rate of approximately 85.2% with a purity of 97.8%, which is superior to biotransformation methods that often struggle with yields below 80% and difficult purification processes.
Q: How does this process simplify downstream processing?
A: The process utilizes solvents like acetone and ether where by-products are water-soluble while the product is water-insoluble. This allows for simple pH adjustment and filtration rather than complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prednisolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN103387595A to maintain competitiveness in the global pharmaceutical market. Our CDMO division possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. We are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of prednisolone meets the highest international standards. Our commitment to technological excellence allows us to offer clients a secure and efficient supply of high-value hormonal intermediates.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most effective and compliant chemical strategy available.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
