Advanced Manufacturing of Bupropion Hydrochloride: A Technical Breakthrough for Global Supply Chains
Advanced Manufacturing of Bupropion Hydrochloride: A Technical Breakthrough for Global Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the production of critical antidepressant agents. Patent CN101088985A introduces a transformative synthesis strategy for Bupropion Hydrochloride, a widely prescribed atypical antidepressant, addressing longstanding inefficiencies in prior art. This novel methodology streamlines the production sequence into three distinct, high-yielding stages: direct alpha-bromination of m-chloropropiophenone, nucleophilic substitution with tert-butylamine, and final salt formation. By circumventing the need for hazardous Grignard reagents or toxic chloromethane, this process establishes a new benchmark for safety and operational simplicity. For global procurement leaders and R&D directors, this technology represents a pivotal opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while drastically reducing the environmental footprint associated with legacy manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Bupropion Hydrochloride has been plagued by significant technical and economic hurdles that impede efficient commercialization. Traditional routes often rely on the chlorination of 3-chloropropiophenone using cupric chloride, a process that, while selective, generates substantial quantities of heavy metal-containing wastewater, imposing severe environmental compliance costs and disposal challenges. Alternative pathways utilizing Grignard reagents demand strictly anhydrous conditions and cryogenic temperatures, escalating energy consumption and introducing safety risks related to exothermic runaway reactions. Furthermore, methods employing toxic alkylating agents like chloromethane pose unacceptable occupational health hazards, necessitating expensive containment infrastructure. These legacy processes are characterized by long reaction cycles, difficult post-treatment purification steps to remove stubborn by-products, and overall yields that fail to maximize raw material utilization, thereby inflating the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in CN101088985A offers a streamlined, direct route that fundamentally reshapes the economic and operational landscape of Bupropion manufacturing. By initiating the synthesis with the direct bromination of m-chloropropiophenone using elemental bromine, the process bypasses the need for complex precursor synthesis or unstable organometallic intermediates. The subsequent amination step leverages excess tert-butylamine not only as a reactant but effectively as a solvent medium, driving the equilibrium towards the desired product without requiring exotic phase transfer catalysts. This approach significantly shortens the production timeline and simplifies the reactor setup, allowing for operation at moderate temperatures between 50°C and 100°C. The elimination of heavy metal catalysts and toxic gases translates directly into reduced capital expenditure on waste treatment facilities and enhanced worker safety, making this route exceptionally attractive for cost reduction in API manufacturing on a multi-ton scale.
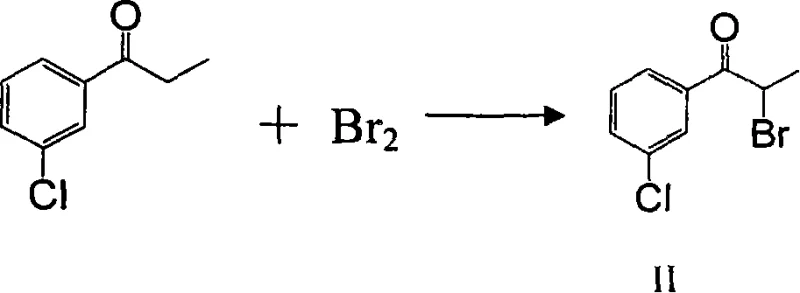
Mechanistic Insights into Direct Alpha-Bromination and Nucleophilic Substitution
The core chemical innovation lies in the controlled electrophilic alpha-bromination of the ketone substrate, a reaction that requires precise thermal management to ensure mono-substitution selectivity. As illustrated in the reaction scheme, m-chloropropiophenone undergoes enolization under the reaction conditions, facilitating the attack of elemental bromine at the alpha-carbon position. Maintaining the reaction temperature within the specified range of 50-100°C is critical; temperatures that are too low may result in incomplete conversion, while excessive heat could promote poly-bromination or degradation of the sensitive ketone functionality. The use of haloalkane solvents such as dichloromethane or 1,2-dichloroethane further stabilizes the reaction mixture, ensuring homogeneous mixing and efficient heat dissipation. This step yields 3'-chloro-alpha-bromopropiophenone (Compound II) with high fidelity, setting the stage for the subsequent substitution without the need for intermediate isolation, thereby minimizing material loss and handling time.
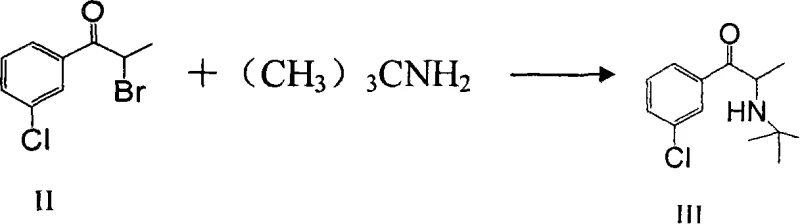
Following bromination, the process advances to a nucleophilic substitution where the bromide leaving group is displaced by tert-butylamine to form the free base of Bupropion (Compound III). This transformation is driven by the use of a significant molar excess of tert-butylamine (4 to 10 equivalents), which serves to push the reaction kinetics forward despite the steric hindrance inherent to the bulky tert-butyl group. The reaction is typically conducted under reflux conditions for 2 to 6 hours to ensure complete consumption of the brominated intermediate. Crucially, the workup procedure involves an aqueous extraction designed specifically to remove the tert-butylamine hydrobromide salt formed as a by-product. This washing step is vital for impurity control, as residual amine salts can interfere with the final crystallization and affect the purity profile of the API. The organic phase, now enriched with the free base, is dried and concentrated, ready for the final salt formation step which locks the molecular structure into its stable hydrochloride form.
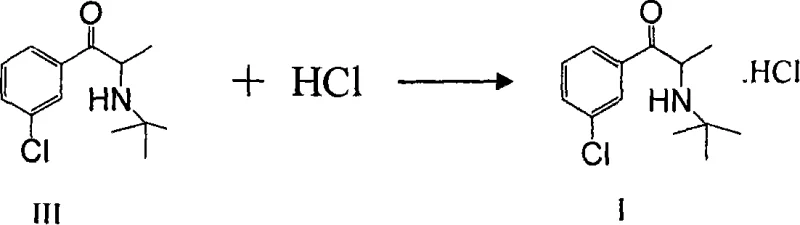
How to Synthesize Bupropion Hydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and thermal profiles outlined in the patent to achieve the reported 70-80% total yield and ≥99.9% purity. The process begins with the dropwise addition of bromine to the ketone substrate, followed by the direct introduction of tert-butylamine into the same vessel or after solvent swap, minimizing unit operations. The final salting-out step utilizes hydrogen chloride gas or saturated solutions in compatible solvents like ethyl acetate or alcohols, precipitating the product which is then subjected to activated carbon treatment and recrystallization. For detailed operational parameters, solvent choices, and specific workup instructions required for GMP compliance, please refer to the standardized synthesis guide below.
- Brominate m-chloropropiophenone with elemental bromine at 50-100°C to form 3'-chloro-alpha-bromopropiophenone.
- React the brominated intermediate with excess tert-butylamine under reflux to perform nucleophilic substitution.
- Treat the resulting free base with hydrogen chloride gas or solution to precipitate pure Bupropion Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers compelling advantages that directly address the pain points of modern pharmaceutical supply chains, particularly regarding cost stability and regulatory compliance. By eliminating the reliance on cupric chloride and Grignard reagents, manufacturers can avoid the volatile pricing and supply constraints associated with specialized metal catalysts and pyrophoric reagents. The simplified workflow reduces the number of discrete processing steps, which correlates to lower labor costs, reduced utility consumption, and decreased equipment occupancy time. Furthermore, the ability to recycle solvents and recover tert-butylamine hydrobromide for neutralization and reuse creates a closed-loop system that minimizes raw material waste. These factors collectively contribute to a more resilient supply chain capable of withstanding market fluctuations while delivering high-purity pharmaceutical intermediates at a competitive price point.
- Cost Reduction in Manufacturing: The exclusion of expensive transition metal catalysts and the avoidance of complex anhydrous conditions significantly lowers the operational expenditure per kilogram of product. The process utilizes commodity chemicals like elemental bromine and tert-butylamine, which are readily available in the global market at stable prices, shielding buyers from the cost volatility often seen with niche reagents. Additionally, the high atom economy of the direct bromination route ensures that raw material inputs are efficiently converted into the final product, reducing the cost burden of waste disposal and unreacted starting materials.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward, as m-chloropropiophenone and tert-butylamine are bulk chemicals produced by multiple vendors worldwide, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which do not require cryogenic cooling or ultra-high vacuum systems, means that production can be maintained across diverse manufacturing sites without specialized infrastructure. This flexibility ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, allowing downstream formulators to maintain optimal inventory levels without fear of disruption.
- Scalability and Environmental Compliance: The process is inherently scalable, having been validated for industrial production with yields reaching up to 80% on large batches. The absence of heavy metal effluents simplifies wastewater treatment protocols, ensuring compliance with increasingly stringent environmental regulations in key manufacturing hubs like China and India. The use of standard solvents such as toluene, ethyl acetate, and alcohols facilitates easy recovery and recycling, aligning with green chemistry principles and reducing the overall environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and comparative analysis with prior art. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or capacity expansion. The answers provided reflect the specific advantages of the direct bromination route over legacy methods, focusing on purity, safety, and economic efficiency.
Q: How does this synthesis method improve upon traditional copper-catalyzed chlorination?
A: Unlike traditional methods using cupric chloride which generate heavy metal waste and require complex disposal, this patent utilizes direct bromination. This eliminates the environmental burden of heavy metal effluents and simplifies the purification workflow, significantly reducing operational costs and environmental compliance risks.
Q: What measures ensure the high purity (≥99.9%) of the final product?
A: The process incorporates a rigorous purification protocol including aqueous washing to remove tert-butylamine hydrobromide salts, followed by activated carbon decolorization and recrystallization from alcohol or ester solvents. This multi-stage refinement ensures the removal of colored impurities and residual solvents, meeting stringent pharmacopeial standards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for scalability. It avoids hazardous reagents like chloromethane and sensitive Grignard reactions. The use of common solvents like toluene or ethyl acetate, combined with robust temperature ranges (50-100°C), allows for safe and efficient scale-up from pilot plants to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bupropion Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of CN101088985A are fully realized in practice. We operate stringent purity specifications through our rigorous QC labs, utilizing advanced HPLC and GC-MS instrumentation to guarantee that every batch of Bupropion Hydrochloride meets or exceeds the ≥99.9% purity benchmark required by global pharmacopeias. Our commitment to quality assurance extends beyond the final product, encompassing full traceability of raw materials and comprehensive documentation to support your regulatory filings.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and environmentally superior synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing process can enhance your supply chain resilience and drive down the total cost of ownership for this critical antidepressant ingredient.
