Advanced Chlorination Strategy for Commercial Scale-up of Complex Pharmaceutical Intermediates
Advanced Chlorination Strategy for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking robust, cost-effective, and environmentally sustainable pathways for the synthesis of critical active pharmaceutical ingredients (APIs) and their precursors. A pivotal advancement in this domain is detailed in patent CN101407469A, which discloses a novel preparation method for Bupropion Hydrochloride, a widely used antidepressant and smoking cessation aid. This technology represents a significant departure from legacy synthetic routes by substituting hazardous liquid bromine with chlorine gas in the presence of a copper catalyst. The structural integrity of the final product, Bupropion Hydrochloride, is maintained with high fidelity, ensuring therapeutic efficacy while drastically altering the economic and ecological footprint of the manufacturing process. 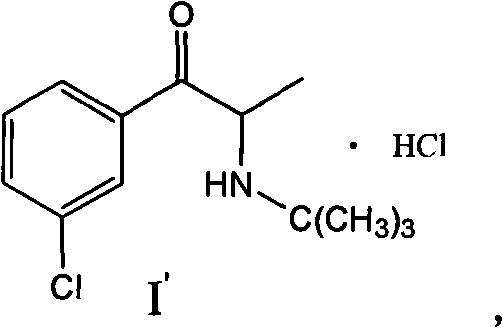 For R&D directors and process chemists, this patent offers a compelling solution to the long-standing challenges associated with halogenation reactions, specifically addressing issues of selectivity, waste management, and operational safety that have historically plagued the production of this key pharmaceutical intermediate.
For R&D directors and process chemists, this patent offers a compelling solution to the long-standing challenges associated with halogenation reactions, specifically addressing issues of selectivity, waste management, and operational safety that have historically plagued the production of this key pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Bupropion has relied heavily on the bromination of 3-chlorophenyl acetone, a pathway elucidated in earlier patents such as US3819706 and US3885046. This conventional approach necessitates the use of elemental liquid bromine (Br2) to generate the alpha-bromo ketone intermediate, which is subsequently reacted with tert-butylamine. While chemically feasible, this legacy method presents severe drawbacks for modern large-scale manufacturing. Liquid bromine is not only exceptionally corrosive and toxic, posing significant occupational health and safety risks, but it is also a costly reagent with volatile market pricing. Furthermore, the bromination process often generates substantial amounts of hazardous waste and requires complex downstream processing to remove brominated impurities, leading to lower overall yields and increased environmental burden. 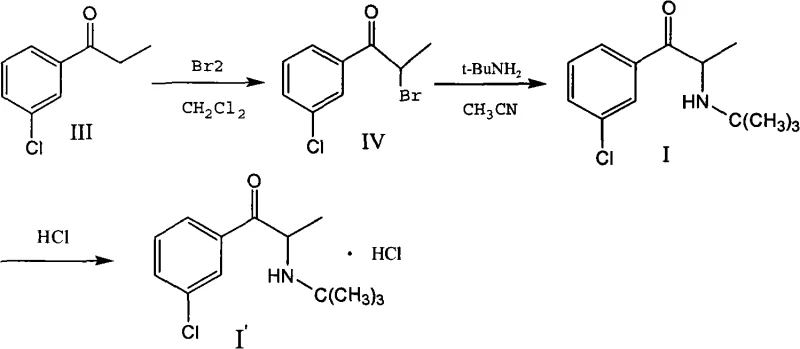 The operational complexity associated with handling liquid bromine, combined with the stringent regulatory pressures regarding halogenated waste disposal, has rendered this traditional pathway increasingly untenable for cost-conscious and sustainability-focused pharmaceutical manufacturers.
The operational complexity associated with handling liquid bromine, combined with the stringent regulatory pressures regarding halogenated waste disposal, has rendered this traditional pathway increasingly untenable for cost-conscious and sustainability-focused pharmaceutical manufacturers.
The Novel Approach
In stark contrast to the bromine-dependent legacy methods, the innovative process disclosed in CN101407469A utilizes chlorine gas (Cl2) mediated by a catalytic amount of copper(II) chloride (CuCl2) to achieve the critical alpha-halogenation step. This strategic substitution transforms the synthesis landscape by leveraging chlorine, a reagent that is significantly more abundant, economical, and easier to handle in continuous flow or large batch reactors compared to liquid bromine. The new route proceeds by dissolving 3-chlorophenyl acetone in a solvent such as ethanol or dichloromethane, introducing the copper catalyst, and feeding chlorine gas under controlled low-temperature conditions to selectively produce 2-chloro-(3-chlorophenyl)-1-acetone. 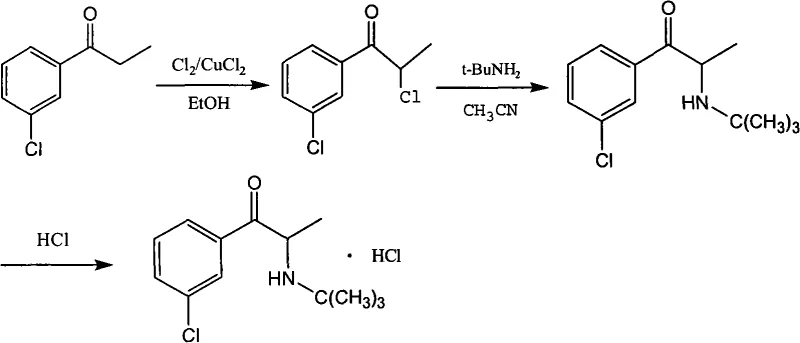 This chloro-intermediate is then seamlessly converted to Bupropion via reaction with tert-butylamine in acetonitrile, followed by salt formation with hydrochloric acid. The elimination of liquid bromine not only simplifies the operational workflow but also aligns the manufacturing process with green chemistry principles, offering a cleaner, safer, and more economically viable alternative for the global supply of this essential antidepressant medication.
This chloro-intermediate is then seamlessly converted to Bupropion via reaction with tert-butylamine in acetonitrile, followed by salt formation with hydrochloric acid. The elimination of liquid bromine not only simplifies the operational workflow but also aligns the manufacturing process with green chemistry principles, offering a cleaner, safer, and more economically viable alternative for the global supply of this essential antidepressant medication.
Mechanistic Insights into CuCl2-Catalyzed Alpha-Chlorination
The core chemical innovation of this patent lies in the efficient alpha-chlorination of the ketone functionality using a transition metal catalyst. Mechanistically, the copper(II) chloride acts as a Lewis acid and potentially facilitates the generation of chlorinating species that selectively target the alpha-carbon adjacent to the carbonyl group. The reaction is typically conducted at temperatures ranging from -10°C to 20°C, with a preferred window of 0°C to 10°C, which is critical for suppressing poly-chlorination and ensuring high regioselectivity for the mono-chlorinated product. The use of solvents like ethanol or dichloromethane provides a suitable medium for the dissolution of the organic substrate and the dispersion of the catalyst, allowing for effective mass transfer of the chlorine gas into the liquid phase. This catalytic system demonstrates remarkable efficiency, with the patent specifying that only 1% to 10% (by weight relative to the substrate) of CuCl2 is required to drive the reaction to completion, thereby minimizing metal contamination in the final product.
From an impurity control perspective, this mechanism offers distinct advantages over radical bromination. The controlled introduction of chlorine gas, monitored by techniques such as TLC, allows operators to halt the reaction precisely at the mono-substitution stage, preventing the formation of di-chloro or tri-chloro byproducts that are difficult to separate. The subsequent workup involves simple aqueous washing with dilute alkali to neutralize acidic byproducts, followed by drying and solvent removal, yielding a buttery oil of the chloro-ketone intermediate that is sufficiently pure for the next step without rigorous distillation. This streamlined purification profile is crucial for maintaining high overall yields and reducing the accumulation of genotoxic impurities, a key concern for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Bupropion Hydrochloride Efficiently
The synthesis of Bupropion Hydrochloride via this chlorination route is designed for operational simplicity and high throughput, making it an ideal candidate for technology transfer and commercial adoption. The process begins with the catalytic chlorination of 3-chlorophenyl acetone, followed by a straightforward nucleophilic substitution with tert-butylamine and final salt formation. The detailed standardized synthesis steps, including specific molar ratios, agitation speeds, and quenching protocols, are outlined in the structured guide below to ensure reproducibility and safety during scale-up operations.
- Dissolve 3-chlorophenyl acetone in a suitable solvent such as ethanol or dichloromethane, add catalytic CuCl2, and introduce chlorine gas at controlled low temperatures (-10°C to 20°C) to obtain 2-chloro-(3-chlorophenyl)-1-acetone.
- React the resulting chloro-ketone intermediate with tert-butylamine in acetonitrile at room temperature to form the free base bupropion.
- Treat the bupropion free base with hydrochloric acid to precipitate and isolate the final high-purity bupropion hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the chlorine-based synthesis route described in CN101407469A presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. The primary advantage stems from the fundamental shift in raw material sourcing; replacing expensive and logistically challenging liquid bromine with commodity-grade chlorine gas results in a drastic reduction in direct material costs. Chlorine is produced on a massive industrial scale globally, ensuring a stable and reliable supply chain that is less susceptible to the price volatility often seen with specialized halogenating agents like bromine. Furthermore, the simplified reaction conditions and reduced hazard profile lower the barriers for manufacturing, allowing for broader supplier qualification and reduced dependency on single-source vendors who possess specialized bromine handling infrastructure.
- Cost Reduction in Manufacturing: The economic implications of switching from bromine to chlorine are profound, driven by both the lower unit cost of the halogenating agent and the reduced expenses associated with waste treatment. Liquid bromine requires specialized storage tanks, corrosion-resistant piping, and extensive safety protocols, all of which add significant capital and operational expenditures to a facility. By eliminating these requirements, manufacturers can achieve substantial cost savings in both CAPEX and OPEX. Additionally, the catalytic nature of the reaction means that expensive metal reagents are used in minimal quantities, further optimizing the cost structure. The qualitative improvement in process efficiency, characterized by fewer purification steps and higher throughput, translates directly into a more competitive cost position for the final API, enabling better margin management in a price-sensitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for life-saving medications like antidepressants, and this new method significantly de-risks the production pipeline. Liquid bromine is classified as a hazardous material with strict transportation regulations, which can lead to delays and logistical bottlenecks, especially across international borders. In contrast, chlorine gas, while requiring care, is a staple of the chemical industry with well-established logistics networks and on-site generation capabilities at many large chemical parks. This accessibility ensures that production schedules are less likely to be disrupted by raw material shortages. Moreover, the robustness of the chlorination process allows for flexible manufacturing campaigns, enabling suppliers to respond more agilely to fluctuations in market demand without the lead time penalties associated with sourcing niche reagents.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent globally, the ability to demonstrate a "green" manufacturing process is a significant commercial asset. The replacement of bromine with chlorine inherently reduces the toxicity and persistence of halogenated waste streams, simplifying effluent treatment and lowering disposal costs. The process described avoids the generation of heavy brominated organic waste, which is often difficult and expensive to incinerate or treat. This environmental compatibility facilitates easier permitting for plant expansions and ensures long-term regulatory compliance. The scalability of the process is further enhanced by the ease of controlling gas-liquid reactions in large reactors compared to the exothermic and hazardous addition of liquid bromine, making the commercial scale-up of complex pharmaceutical intermediates safer and more predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chlorination technology, derived directly from the patent specifications and industry best practices for pharmaceutical intermediate production. These insights are intended to clarify the operational feasibility and strategic value of adopting this novel synthetic route for Bupropion Hydrochloride.
Q: Why is the chlorine-based route superior to the traditional bromine method for Bupropion synthesis?
A: The chlorine-based route described in patent CN101407469A eliminates the use of liquid bromine, which is highly polluting, corrosive, and expensive. By substituting bromine with chlorine gas and using a copper catalyst, the process significantly reduces raw material costs, simplifies operational complexity, and minimizes hazardous waste generation, aligning better with modern environmental regulations.
Q: What are the critical reaction conditions for the alpha-chlorination step?
A: The alpha-chlorination of 3-chlorophenyl acetone requires precise temperature control between -10°C and 20°C, preferably 0°C to 10°C, to prevent over-chlorination or side reactions. The reaction utilizes a catalytic amount of CuCl2 (1-10% by weight) and can be performed in solvents like ethanol, methanol, or dichloromethane, ensuring high selectivity for the mono-chlorinated intermediate.
Q: How does this method impact the scalability of Bupropion production?
A: This method enhances scalability by replacing the handling of difficult liquid bromine with gaseous chlorine, which is easier to meter and control in large-scale reactors. The simplified workup procedure, which avoids complex purification steps associated with brominated byproducts, allows for more efficient commercial scale-up of complex pharmaceutical intermediates with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bupropion Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chlorination-based synthesis route for Bupropion Hydrochloride and are fully equipped to leverage this technology for our global partners. As a premier CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are designed to handle gaseous reagents like chlorine with the utmost safety and precision, supported by rigorous QC labs that enforce stringent purity specifications to meet the highest pharmacopoeial standards. We understand that consistency and quality are non-negotiable in the pharmaceutical supply chain, and our commitment to process excellence guarantees a reliable supply of high-purity intermediates.
We invite procurement directors and R&D leaders to collaborate with us to optimize their supply chains through this advanced manufacturing approach. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and comprehensive quotations that demonstrate the tangible economic and operational benefits of our Bupropion Hydrochloride production capabilities.
