Scalable Production of Octadecanedioic Acid via Novel Radical Coupling and Hydrogenation
The pharmaceutical and polymer industries have long sought efficient, scalable routes for long-chain dicarboxylic acids, specifically octadecanedioic acid, a critical monomer for high-performance nylons and specialty coatings. Patent CN112079708A introduces a groundbreaking preparation method that bypasses the limitations of traditional biological fermentation and harsh chemical oxidation. This novel approach utilizes a free radical coupling reaction between 1-hydroxy-2-hydroperoxy-cyclopentane and butadiene, followed by a mild catalytic hydrogenation step. By leveraging inexpensive iron-based catalysts and standard palladium-carbon systems, this technology achieves a total yield exceeding 85 percent with purity levels surpassing 98.5 percent. For R&D directors and procurement specialists, this represents a significant shift towards cost-effective, environmentally compliant manufacturing of high-value fine chemical intermediates without compromising on structural integrity or supply reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of octadecanedioic acid has been plagued by significant technical and economic hurdles. Biological methods, often employing Candida tropicalis strains to oxidize fatty alcohols or acids, suffer from inherently low target yields and generate complex byproduct profiles that make purification arduous and expensive. As illustrated in the biological oxidation pathways, these enzymatic processes are sensitive to substrate inhibition and require stringent control over fermentation conditions, limiting their scalability. Furthermore, existing chemical synthesis routes reported in prior art frequently rely on stoichiometric amounts of toxic oxidants like potassium dichromate or expensive precious metal catalysts such as ruthenium. These conventional chemical methods often necessitate high temperatures and pressures, leading to increased energy consumption, safety risks, and the generation of hazardous heavy metal waste streams that complicate regulatory compliance and disposal logistics.
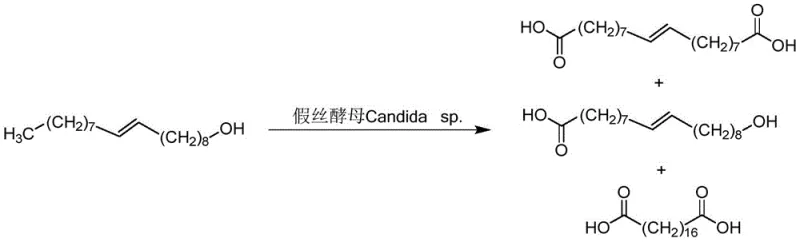
The Novel Approach
In stark contrast, the methodology disclosed in CN112079708A offers a streamlined, two-step chemical synthesis that dramatically simplifies the production landscape. The process initiates with a radical coupling reaction that efficiently constructs the carbon backbone under mild thermal conditions ranging from 30 to 90 degrees Celsius. This is followed by a selective catalytic hydrogenation using palladium-carbon, a standard and recoverable heterogeneous catalyst, to saturate the intermediate double bonds. This route eliminates the need for toxic chromium reagents and expensive ruthenium complexes, thereby reducing raw material costs and environmental liability. The use of common organic solvents like tetrahydrofuran and methanol further enhances the practicality of the process, allowing for straightforward workup procedures involving extraction and crystallization. This robust chemical strategy ensures consistent product quality and high throughput, addressing the supply chain vulnerabilities associated with biologically derived intermediates.
Mechanistic Insights into Fe-Catalyzed Radical Coupling and Hydrogenation
The core innovation of this synthesis lies in the efficient construction of the C18 carbon chain via a free radical mechanism initiated by transition metal salts. In the first step, catalysts such as ferrous ammonium sulfate hexahydrate or ammonium thiocyanate facilitate the homolytic cleavage of the hydroperoxide bond in Compound I. This generates reactive radical species that undergo coupling with butadiene molecules, effectively extending the carbon chain to form octadecyl-7,11-diene diacid. The reaction kinetics are carefully managed by controlling the addition rate of reactants and maintaining temperatures between 30 and 90 degrees Celsius to minimize side reactions such as polymerization or over-oxidation. The choice of solvent, typically tetrahydrofuran or acetone, plays a crucial role in stabilizing the radical intermediates and ensuring high conversion rates. This mechanistic precision allows for the formation of the specific diene intermediate with high regioselectivity, setting the stage for the subsequent reduction step.
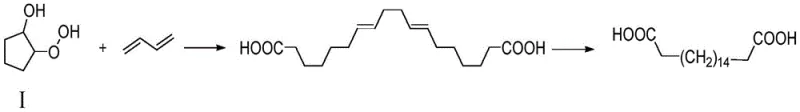
Following the coupling reaction, the resulting unsaturated diacid undergoes catalytic hydrogenation to yield the final saturated octadecanedioic acid. This step utilizes a palladium-carbon catalyst in an alcoholic solvent medium, such as methanol or ethanol, under a hydrogen atmosphere at moderate temperatures of 30 to 50 degrees Celsius. The mild conditions of this hydrogenation step are critical for preserving the integrity of the carboxylic acid groups while selectively reducing the carbon-carbon double bonds. The heterogeneous nature of the Pd/C catalyst allows for easy removal via filtration, preventing metal contamination in the final product and facilitating catalyst recycling. This two-stage mechanism ensures that impurities are minimized throughout the process, resulting in a final product with purity levels consistently above 98.5 percent, which is essential for downstream applications in high-performance polymers and pharmaceutical synthesis where trace impurities can degrade material properties.
How to Synthesize Octadecanedioic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing precise control over reaction parameters to maximize yield. The process begins with the careful dropwise addition of the hydroperoxide precursor to a butadiene solution at low temperatures to manage exothermicity, followed by the introduction of the iron catalyst to initiate coupling. After isolation of the diene intermediate, the subsequent hydrogenation is performed under controlled hydrogen pressure to ensure complete saturation. While the general procedure is robust, specific optimization of catalyst loading and solvent ratios may be required for different batch sizes.
- Perform a radical coupling reaction between Compound I and butadiene using an iron-based catalyst at 30-90°C to form octadecyl-7,11-diene diacid.
- Dissolve the intermediate diacid in an alcohol solvent and introduce hydrogen gas with a palladium-carbon catalyst at 30-50°C.
- Filter the reaction mixture to remove the catalyst, concentrate the filtrate, and crystallize the final octadecanedioic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers tangible strategic benefits centered around cost stability and operational efficiency. By replacing expensive precious metal catalysts like ruthenium with abundant iron salts, the raw material cost structure is significantly optimized, reducing exposure to volatile commodity markets. Furthermore, the elimination of toxic reagents such as potassium dichromate removes the need for specialized hazardous waste disposal contracts and reduces the regulatory burden associated with heavy metal handling. The mild reaction conditions, operating well below the extreme temperatures and pressures required by older chemical methods, translate to lower energy consumption and reduced wear on reactor equipment, thereby extending asset life and minimizing maintenance downtime. These factors collectively contribute to a more resilient and cost-effective supply chain for long-chain diacids.
- Cost Reduction in Manufacturing: The substitution of high-cost ruthenium catalysts and toxic oxidants with economical iron-based systems and hydrogen gas drastically lowers the direct material costs per kilogram of product. Additionally, the simplified downstream processing, which relies on standard extraction and crystallization rather than complex chromatographic purification, reduces labor and solvent recovery expenses. This lean manufacturing approach ensures that the final octadecanedioic acid can be produced at a competitive price point, enhancing margin potential for downstream polymer and pharmaceutical applications.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals like butadiene and common solvents such as methanol and tetrahydrofuran mitigates the risk of supply disruptions often associated with specialized biological substrates or rare earth catalysts. The robustness of the chemical synthesis allows for flexible production scheduling and rapid scale-up capabilities, ensuring consistent delivery timelines even during periods of high market demand. This reliability is crucial for maintaining continuous operation in just-in-time manufacturing environments where raw material availability is a critical bottleneck.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale production, utilizing unit operations that are standard in the fine chemical industry, such as stirred tank reactors and filtration units. The absence of persistent organic pollutants and heavy metal residues simplifies wastewater treatment and aligns with increasingly stringent global environmental regulations. This eco-friendly profile not only reduces compliance costs but also enhances the sustainability credentials of the final product, appealing to end-users in green chemistry and sustainable materials sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or new product development pipelines.
Q: What are the primary advantages of this radical coupling method over biological fermentation?
A: Unlike biological methods which suffer from low yields and complex purification due to enzyme specificity issues, this chemical route offers a total yield exceeding 85% and purity above 98.5% with significantly simpler downstream processing.
Q: Does this process utilize toxic heavy metal catalysts like Ruthenium or Chromium?
A: No, the process explicitly avoids expensive and toxic reagents such as ruthenium catalysts or potassium dichromate, utilizing instead cost-effective iron salts and standard palladium-carbon for hydrogenation.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the reaction conditions are mild (30-90°C) and utilize common organic solvents like tetrahydrofuran and methanol, making the process highly scalable and safe for commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octadecanedioic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity octadecanedioic acid plays in the advancement of next-generation polyamides and pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for high-performance applications. Our commitment to quality assurance means that clients can rely on us for consistent supply of materials that drive innovation in their own formulations.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall production costs.
