Advanced Radical Coupling Synthesis of Octadecanedioic Acid for Commercial Scale-Up
Advanced Radical Coupling Synthesis of Octadecanedioic Acid for Commercial Scale-Up
The global demand for long-chain dicarboxylic acids, particularly octadecanedioic acid, has surged due to their critical role in manufacturing high-performance nylon materials like Nylon 1818, advanced hot melt adhesives, and specialized coatings. Historically, the supply chain for this valuable intermediate has been constrained by inefficient production methods that struggle to balance yield, purity, and environmental compliance. A groundbreaking preparation method detailed in patent CN112079708A introduces a robust two-step synthetic route that fundamentally shifts the production paradigm from complex biological fermentation or harsh chemical oxidation to a streamlined free radical coupling strategy. This innovation delivers a target product with a total yield exceeding 85 percent and a purity surpassing 98.5 percent, addressing the chronic supply shortages that have kept market prices artificially high. By leveraging mild reaction conditions and avoiding toxic reagents, this technology offers a compelling value proposition for manufacturers seeking a reliable octadecanedioic acid supplier capable of meeting stringent quality standards without compromising on sustainability or cost-efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of octadecanedioic acid has relied heavily on biological fermentation processes or aggressive chemical oxidation techniques, both of which present significant bottlenecks for industrial scalability. Biological methods, often utilizing mutant strains of Candida tropicalis or cytochrome P450 enzymes to oxidize fatty acids or alcohols, are plagued by inherent enzymatic limitations such as low catalytic efficiency, susceptibility to substrate inhibition, and the generation of complex byproduct profiles that complicate purification. As illustrated in the biological oxidation pathways, these processes frequently result in low target yields and require energy-intensive downstream processing to isolate the desired diacid from a mixture of mono-acids and unreacted starting materials. Furthermore, alternative chemical routes reported in prior art often depend on expensive and hazardous oxidizing agents like potassium dichromate or noble metal catalysts such as ruthenium, which necessitate rigorous removal steps to meet pharmaceutical and polymer-grade specifications, thereby inflating production costs and environmental liabilities.
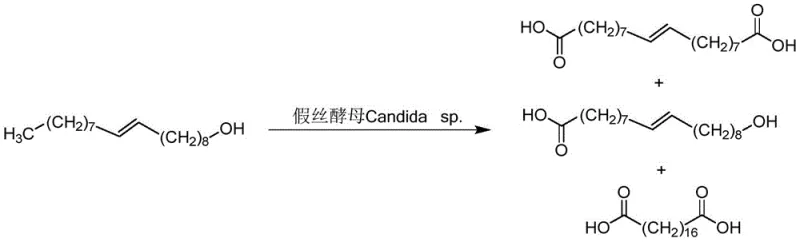
The Novel Approach
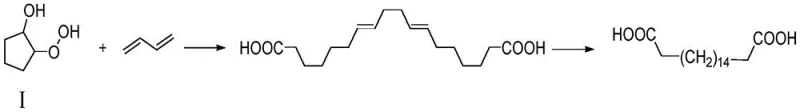
In stark contrast to these legacy technologies, the novel approach disclosed in the patent utilizes a clever free radical coupling reaction between 1-hydroxy-2-hydroperoxy-cyclopentane (Compound I) and butadiene to construct the carbon backbone efficiently. This method bypasses the slow, step-wise oxidation of long-chain fatty acids by directly assembling the C18 chain through a rapid coupling mechanism catalyzed by inexpensive iron salts. The subsequent hydrogenation step is equally optimized, operating at mild temperatures between 30 to 50 degrees Celsius using a standard palladium-carbon catalyst, which ensures complete saturation of the double bonds without degrading the carboxylic acid functionalities. This strategic shift not only drastically reduces the number of synthetic steps but also eliminates the need for extreme reaction conditions, such as the high pressures and temperatures seen in older hydrolysis methods, making the process inherently safer and more adaptable to continuous flow manufacturing setups for cost reduction in polymer additive manufacturing.
Mechanistic Insights into Fe-Catalyzed Radical Coupling and Hydrogenation
The core of this technological breakthrough lies in the precise control of the free radical coupling mechanism, where an iron-based catalyst, such as ferrous ammonium sulfate hexahydrate or ammonium thiocyanate, facilitates the homolytic cleavage of the peroxide bond in Compound I. Upon protonation and activation by the catalyst, the cyclopentane derivative generates reactive radical species that readily attack the electron-rich double bonds of butadiene, initiating a chain propagation sequence that extends the carbon skeleton to the desired C18 length. The reaction is meticulously controlled within a temperature window of 30 to 90 degrees Celsius to balance reaction kinetics with selectivity; temperatures that are too low result in incomplete conversion, while excessive heat promotes side reactions that degrade yield. The use of polar aprotic solvents like tetrahydrofuran or acetonitrile further stabilizes the transition states, ensuring that the coupling proceeds with high regioselectivity to form octadecyl-7,11-diene diacid as the predominant intermediate, minimizing the formation of oligomeric impurities that typically plague radical reactions.
Following the coupling stage, the mechanistic focus shifts to the catalytic hydrogenation of the diene intermediate, a critical step for achieving the final saturated dicarboxylic acid structure required for nylon polymerization. The process employs a heterogeneous palladium-carbon catalyst dispersed in an alcoholic solvent, where molecular hydrogen is activated on the metal surface to sequentially reduce the internal double bonds of the octadecyl-7,11-diene diacid. Operating at a mild 30 to 50 degrees Celsius prevents the over-reduction or hydrogenolysis of the carboxyl groups, a common side reaction in harsher hydrogenation protocols. This gentle yet effective reduction ensures that the stereochemical integrity of the chain is maintained while delivering a product with exceptional purity levels above 99 percent after simple crystallization. The robustness of this mechanism allows for the use of technical grade raw materials without sacrificing the quality of the final output, providing a significant advantage in terms of raw material sourcing flexibility and overall process economics.
How to Synthesize Octadecanedioic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for producing high-purity octadecanedioic acid suitable for immediate industrial adoption. The process begins with the careful addition of Compound I to a cooled solution of butadiene and solvent, followed by the introduction of the aqueous catalyst solution to initiate the exothermic coupling reaction. Strict temperature control during the addition phase, keeping the system below 10 degrees Celsius, is crucial to suppress premature decomposition and maximize the yield of the diene intermediate. Once the coupling is complete, the intermediate is isolated via extraction and concentration before being subjected to the hydrogenation step in methanol or ethanol.
- Perform a free radical coupling reaction between Compound I (1-hydroxy-2-hydroperoxy-cyclopentane) and butadiene using an iron-based catalyst (e.g., ferrous ammonium sulfate) in an organic solvent at 30-90°C to form octadecyl-7,11-diene diacid.
- Dissolve the resulting diene diacid in an alcohol solvent and introduce hydrogen gas in the presence of a palladium-carbon catalyst at 30-50°C to effectuate catalytic hydrogenation.
- Filter the reaction mixture to remove the catalyst, concentrate the filtrate at low temperature, and induce crystallization using diethyl ether to isolate high-purity octadecanedioic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend far beyond simple unit price negotiations. The elimination of expensive noble metal catalysts like ruthenium and toxic oxidants like chromium significantly lowers the raw material cost base, while the mild reaction conditions reduce energy consumption and the wear and tear on reactor vessels. This process optimization leads to substantial cost savings in octadecanedioic acid manufacturing by removing the need for complex heavy metal scavenging steps and extensive wastewater treatment associated with toxic effluents. Furthermore, the high selectivity of the radical coupling reaction minimizes the generation of difficult-to-separate byproducts, streamlining the purification workflow and reducing the loss of valuable material during isolation, which directly improves the overall mass balance and profitability of the production line.
- Cost Reduction in Manufacturing: The replacement of proprietary enzymatic systems and expensive transition metal catalysts with commodity iron salts and standard palladium-on-carbon creates a dramatically lower cost structure for production. By avoiding the high capital expenditure associated with high-pressure reactors required for traditional chemical hydrolysis, manufacturers can utilize existing standard glass-lined or stainless steel equipment, accelerating the time to market and reducing depreciation costs. The high yield of over 85 percent ensures that raw material utilization is maximized, reducing the cost per kilogram of the final API intermediate and allowing for more competitive pricing in the global market without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals such as butadiene and simple cyclopentane derivatives mitigates the risk of supply disruptions often associated with specialized biological feedstocks or rare earth catalysts. The robustness of the chemical process ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed fermentations or inconsistent enzymatic activity. This stability allows supply chain planners to forecast inventory levels with greater accuracy and commit to longer-term delivery schedules, ensuring a continuous flow of high-purity octadecanedioic acid to downstream polymer and pharmaceutical customers.
- Scalability and Environmental Compliance: The mild operating temperatures and absence of hazardous heavy metals simplify the regulatory compliance landscape, making it easier to obtain environmental permits for facility expansion. The process generates significantly less toxic waste compared to chromium-based oxidation routes, lowering disposal costs and aligning with the increasingly strict ESG (Environmental, Social, and Governance) mandates of multinational corporations. This environmental compatibility facilitates the commercial scale-up of complex polymer additives, enabling producers to ramp up capacity from pilot plants to multi-ton annual production lines with minimal regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on the feasibility and advantages of the method.
Q: What are the advantages of the radical coupling method over biological fermentation for octadecanedioic acid?
A: Unlike biological methods which suffer from low yields, complex purification, and enzyme instability, the radical coupling method described in CN112079708A offers a total yield exceeding 85% and purity greater than 98.5% under mild chemical conditions, significantly simplifying downstream processing.
Q: Does this synthesis route utilize toxic heavy metal catalysts?
A: No, this novel process avoids the use of expensive and toxic heavy metals like Ruthenium or Chromium often found in traditional oxidation methods. Instead, it utilizes inexpensive iron-based catalysts for the coupling step and standard Palladium-Carbon for hydrogenation, aligning with green chemistry principles.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (30-90°C for coupling, 30-50°C for hydrogenation) and do not require extreme high-pressure equipment typical of other chemical routes. This makes the process highly scalable and safer for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octadecanedioic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this radical coupling technology in securing a stable and cost-effective supply of octadecanedioic acid for the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of octadecanedioic acid meets the exacting requirements for high-performance nylon synthesis and pharmaceutical applications. We are committed to leveraging this innovative patent to deliver a product that combines superior quality with unmatched supply chain resilience.
We invite forward-thinking R&D and procurement leaders to collaborate with us to evaluate the feasibility of integrating this advanced route into their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project benefits from the latest advancements in fine chemical synthesis while maintaining the highest standards of quality and reliability.
