Advanced Synthetic Route for High-Purity Cannabidiol Intermediates and API Manufacturing
Advanced Synthetic Route for High-Purity Cannabidiol Intermediates and API Manufacturing
The global pharmaceutical landscape is witnessing a paradigm shift in the production of Cannabidiol (CBD), moving away from agriculturally dependent extraction methods toward precise, scalable chemical synthesis. Patent CN112592260A introduces a groundbreaking methodology that addresses the critical bottlenecks of purity, regulatory compliance, and supply chain stability inherent in traditional hemp extraction. This technical insight report analyzes the novel synthetic pathway disclosed in the patent, which leverages a sophisticated combination of acid-catalyzed alkylation and palladium-catalyzed Suzuki cross-coupling. By decoupling production from crop cycles and eliminating the risk of tetrahydrocannabinol (THC) contamination, this technology offers a compelling value proposition for R&D directors and procurement leaders seeking a reliable cannabinoid intermediate supplier. The following analysis details the mechanistic advantages and commercial viability of this next-generation manufacturing process.
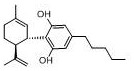
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on extracting Cannabidiol from industrial cannabis varieties, a method fraught with significant regulatory and technical challenges. Although industrial hemp is legally defined by a tetrahydrocannabinol content below 0.3%, the extraction process utilizing tender leaves and buds often results in THC levels that exceed this threshold, triggering stringent drug control regulations. Furthermore, prior art synthetic attempts, such as the one-step method described in US20090036523A1 using olivetol and p-toluenesulfonic acid, have proven inadequate for commercial scale-up due to complex reaction systems that generate numerous isomers and dimers. These legacy processes typically suffer from low yields, often around 24%, and necessitate tedious column chromatography for purification, rendering them economically unfeasible for high-volume API manufacturing. Additionally, alternative routes involving methyl ester intermediates, as seen in U.S. Pat. No. 4,20100298579, fail to provide intermediates that can be effectively purified via recrystallization, leading to purity plateaus that do not meet rigorous pharmaceutical standards.
The Novel Approach
In stark contrast to these legacy limitations, the methodology outlined in Patent CN112592260A presents a streamlined, high-efficiency synthetic strategy that fundamentally resolves the purification and yield issues of previous generations. This novel approach utilizes a controlled acid-catalyzed coupling between a specific resorcinol derivative and a chiral terpene alcohol to form a stable intermediate, which can be further activated into a halide-like compound or sulfonate ester. The key innovation lies in the subsequent Suzuki coupling step, where a pentyl boron species is introduced to construct the final alkyl side chain with high regioselectivity. This two-stage process allows for the isolation of intermediates that are amenable to standard recrystallization techniques using solvents like ethyl acetate, achieving intermediate purities of up to 98.7% and final product purities exceeding 99%. By replacing unpredictable agricultural sourcing with a deterministic chemical synthesis, this route ensures consistent quality and eliminates the legal complexities associated with THC management.
Mechanistic Insights into Acid-Catalyzed Coupling and Suzuki Cross-Coupling
The core of this synthetic breakthrough relies on a meticulously optimized two-step reaction sequence that maximizes atom economy while minimizing byproduct formation. The first stage involves a Friedel-Crafts-type alkylation where a resorcinol derivative (Raw Material A), bearing either a hydroxyl or a halogen substituent at the 5-position, reacts with (1S,4R)-1-methyl-4-(1-methylvinyl)-2-cyclohexene-1-alcohol. This transformation is driven by a robust acid catalyst system, which may include p-toluenesulfonic acid, boron trifluoride diethyl etherate, or Lewis acids like zinc chloride, operating at moderate temperatures between 80°C and 100°C. The stereochemical integrity of the terpene moiety is preserved during this coupling, ensuring the formation of the correct trans-configuration essential for biological activity. Following the initial coupling, the resulting Intermediate II can be activated by converting its phenolic hydroxyl group into a superior leaving group, such as a mesylate (-OMs) or tosylate (-OTs), using methanesulfonyl chloride or p-toluenesulfonyl chloride in the presence of a base like triethylamine. This activation step is crucial for facilitating the subsequent cross-coupling reaction by enhancing the electrophilicity of the aromatic ring.
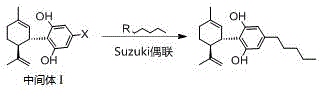
The second mechanistic pillar is the palladium-catalyzed Suzuki-Miyaura cross-coupling, which installs the pentyl side chain with exceptional precision. In this step, the activated intermediate reacts with a pentyl boron reagent, such as pentylboronic acid, pentylboronic acid pinacol ester, or potassium pentyltrifluoroborate. The reaction is mediated by a palladium catalyst system, potentially employing Pd(PPh3)4, Pd(OAc)2, or Pd(dppf)Cl2, in conjunction with a suitable base like potassium phosphate or sodium carbonate. The choice of solvent is equally critical, with toluene, tetrahydrofuran, or dioxane providing the optimal medium for the catalytic cycle to proceed efficiently at temperatures ranging from 80°C to 100°C. This mechanistic design effectively suppresses the formation of homocoupling byproducts and isomers, which are common pitfalls in cannabinoid synthesis. The result is a clean reaction profile that allows for simple workup procedures involving acid washes and recrystallization, rather than expensive and wasteful chromatographic separations, thereby significantly enhancing the overall process mass intensity (PMI).
How to Synthesize Cannabidiol Efficiently
The practical implementation of this synthetic route is designed for seamless translation from laboratory benchtop to pilot plant and full-scale commercial production. The process begins with the preparation of the key coupled intermediate, followed by the final Suzuki coupling to yield the target API. Detailed operational parameters, including specific molar ratios, temperature ramps, and quenching protocols, are critical for maintaining the high purity profiles reported in the patent examples. For R&D teams looking to replicate or license this technology, understanding the nuances of the recrystallization steps is paramount, as this is where the bulk of impurity rejection occurs. The standardized synthesis steps below outline the critical path for producing high-quality Cannabidiol in accordance with the patented methodology.
- Perform an acid-catalyzed coupling reaction between a resorcinol derivative (Raw Material A) and (1S,4R)-1-methyl-4-(1-methylvinyl)-2-cyclohexene-1-alcohol to form Intermediate I or II.
- Optionally convert the hydroxyl group of Intermediate II into a leaving group (mesylate or tosylate) to form Intermediate III for enhanced reactivity.
- Execute a Suzuki coupling reaction between the intermediate and a pentyl boron species (such as pentylboronic acid or potassium pentyltrifluoroborate) using a palladium catalyst to yield Cannabidiol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction to this advanced synthetic route represents a strategic opportunity to de-risk the supply of critical cannabinoid ingredients. Traditional extraction models are inherently vulnerable to agricultural variables such as weather patterns, pest infestations, and seasonal harvest cycles, all of which can lead to volatile pricing and inconsistent availability. In contrast, this chemical synthesis method utilizes readily available petrochemical-derived starting materials and standard reagents, creating a supply chain that is immune to crop failures. The ability to produce the API on demand, independent of growing seasons, ensures a continuous and reliable flow of materials for downstream formulation, which is essential for maintaining market share in the fast-moving nutraceutical and pharmaceutical sectors. Furthermore, the synthetic route eliminates the need for costly and time-consuming regulatory approvals associated with handling controlled cannabis biomass, streamlining the logistics of international shipping and warehousing.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of column chromatography, which is a major cost center in traditional cannabinoid purification. By designing intermediates that can be purified via simple recrystallization, the process drastically reduces solvent consumption, silica gel waste, and labor hours associated with fraction collection. Additionally, the high yields reported for both the intermediate and final steps mean that less raw material is required to produce a kilogram of finished goods, directly lowering the cost of goods sold (COGS). The use of common industrial solvents like toluene and ethyl acetate, which are easily recovered and recycled, further contributes to substantial cost savings compared to specialized extraction solvents.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the synthetic nature of this pathway, which decouples production from the geopolitical and environmental risks associated with hemp cultivation. Manufacturers can secure long-term contracts for chemical raw materials with greater stability than agricultural commodities, allowing for more accurate forecasting and inventory planning. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require cryogenic temperatures or ultra-high pressures, ensures that production can be maintained across multiple geographic locations without the need for specialized infrastructure. This flexibility allows for a diversified manufacturing footprint, reducing the risk of single-point failures in the supply network.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with green chemistry principles by minimizing waste generation and energy consumption. The absence of THC byproducts simplifies wastewater treatment and hazardous waste disposal, as there is no need to manage controlled substances or complex botanical residues. The process is inherently scalable, as demonstrated by the successful execution of reactions in multi-liter vessels in the patent examples, indicating a clear path to metric-ton production without the need for re-optimization. This scalability ensures that the technology can meet surging global demand without the lead times associated with expanding agricultural acreage, making it an ideal solution for rapid market expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic Cannabidiol process. These answers are derived directly from the experimental data and beneficial effects described in Patent CN112592260A, providing clarity on purity standards, regulatory implications, and process robustness. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does this synthetic method compare to plant extraction in terms of regulatory compliance?
A: Unlike extraction from industrial hemp, which risks exceeding the 0.3% THC limit and requires strict drug control licenses, this synthetic route produces Cannabidiol without generating tetrahydrocannabinol byproducts, significantly simplifying regulatory approval and supply chain logistics.
Q: What are the purity advantages of the intermediates produced in Patent CN112592260A?
A: The intermediates generated in this process, specifically Intermediate I and Intermediate III, possess physical properties that allow for recrystallization using conventional solvents like ethyl acetate. This enables the achievement of purities up to 98.7% for intermediates and over 99% for the final API, overcoming the purification difficulties associated with prior art methyl ester intermediates.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes robust reaction conditions with temperatures below 100°C and common industrial solvents such as toluene and ethyl acetate. The elimination of complex column chromatography in favor of recrystallization makes the process highly scalable and cost-effective for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cannabidiol Supplier
As the global demand for high-purity cannabinoids continues to surge, partnering with a technically proficient CDMO is essential for navigating the complexities of modern API manufacturing. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Cannabidiol meets the highest international pharmacopeial standards. We understand the critical importance of impurity control and are fully prepared to implement the advanced Suzuki coupling technologies described in recent patents to deliver superior product quality.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can optimize your supply chain and reduce manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching from extraction to synthesis for your specific application. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven technical capability.
