Advanced Synthetic Route for High-Purity Cannabidiol: Scalable Manufacturing Insights
The pharmaceutical landscape for cannabinoid-based therapeutics is rapidly evolving, driven by the critical need for enantiomerically pure active substances that minimize adverse side effects. As highlighted in the background of patent CN113508120B, the historical tragedy of thalidomide underscores the absolute necessity of controlling stereochemistry in drug synthesis, where even minor contamination by undesired enantiomers can lead to catastrophic pharmacological outcomes. This patent introduces a sophisticated and robust methodology for the preparation of cannabidiol (CBD), addressing the longstanding challenges of safety, scalability, and purity that have plagued earlier synthetic routes. By leveraging a novel sequence involving silylation and controlled bromination, the inventors have established a pathway that not only mitigates the risks associated with hazardous reagents but also generates stable, marketable intermediates. This technical breakthrough represents a significant leap forward for manufacturers seeking to establish a reliable supply chain for high-purity cannabinoid APIs, offering a distinct advantage over conventional methods that often struggle with regioselectivity and operational hazards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for producing dibrominated olivetol precursors, such as those described in prior art like WO 2017011210 A1, frequently rely on the direct use of elemental bromine. This approach presents severe limitations when translated from laboratory benchtops to industrial-scale manufacturing environments. The handling of elemental bromine requires stringent safety protocols due to its highly corrosive nature and the potential for generating toxic vapors, which poses significant health risks to personnel and necessitates expensive containment infrastructure. Furthermore, these conventional bromination reactions often mandate cryogenic conditions, typically requiring temperatures well below zero degrees Celsius to control exothermicity and prevent poly-bromination or degradation. Maintaining such low temperatures on a multi-ton scale imposes a substantial energy burden and complicates reactor design, leading to increased capital expenditure and operational costs. Additionally, the lack of protecting groups in these older methods can result in poor regioselectivity, yielding complex mixtures of isomers that are difficult and costly to separate, ultimately compromising the overall purity and yield of the final active pharmaceutical ingredient.
The Novel Approach
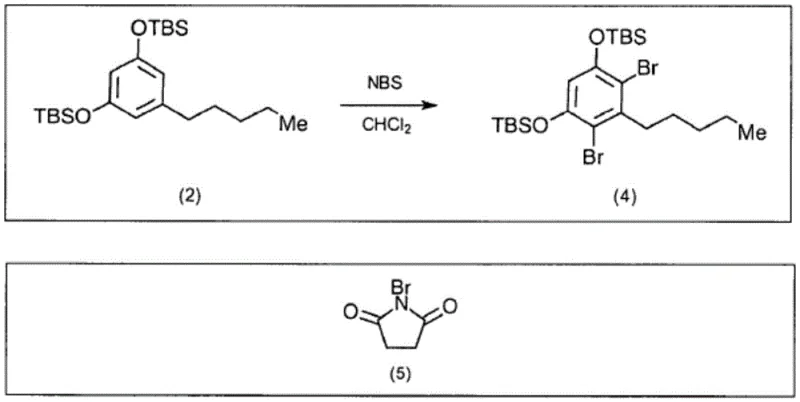
In stark contrast, the methodology disclosed in patent CN113508120B employs a strategic silylation step followed by bromination using N-bromosuccinimide (NBS), effectively circumventing the drawbacks of elemental bromine. The initial protection of the olivetol hydroxyl groups with tert-butyldimethylsilyl chloride (TBSCl) serves a dual purpose: it stabilizes the molecule against oxidation and, crucially, masks the para position of the aromatic ring. This masking effect directs the subsequent electrophilic aromatic substitution exclusively to the desired ortho positions, ensuring high regioselectivity without the need for extreme cooling. The use of NBS as the brominating agent allows the reaction to proceed under much milder conditions, often at or near room temperature, which drastically simplifies the thermal management requirements for large-scale reactors. This shift from hazardous elemental halogens to solid, manageable reagents like NBS not only enhances operator safety but also streamlines the workflow, making the process inherently more scalable and economically viable for commercial production of fine chemical intermediates.
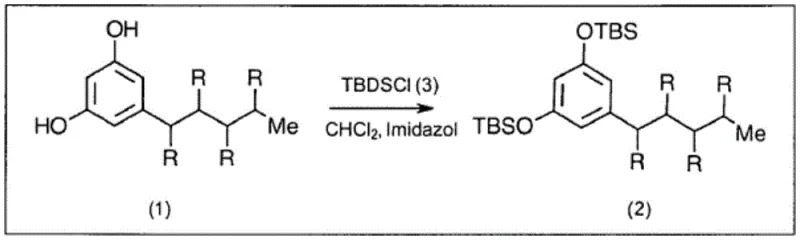
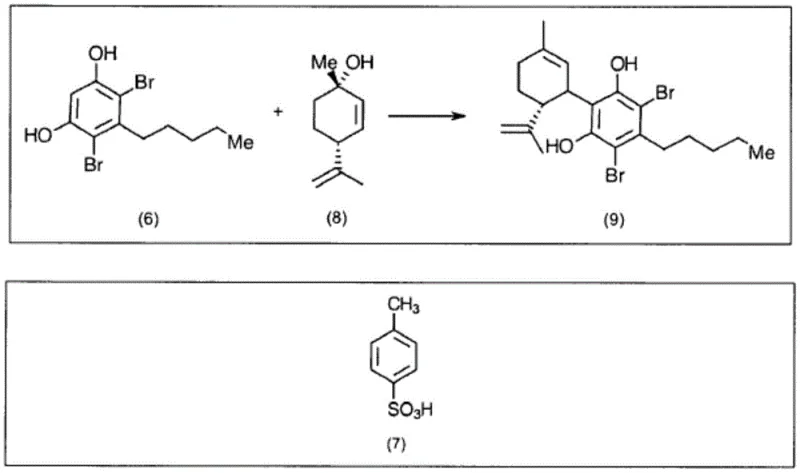
The mechanistic elegance of this synthesis lies in the precise control exerted over the aromatic substitution pattern through temporary functional group protection. The initial silylation of olivetol converts the reactive phenolic hydroxyls into bulky silyl ethers, which electronically and sterically influence the reactivity of the benzene ring. By blocking the para position, the synthesis forces the incoming bromine electrophiles to attack the positions ortho to the pentyl chain, resulting in the formation of 4,6-dibromo derivatives with exceptional fidelity. This level of control is paramount for R&D directors focused on impurity profiles, as it minimizes the formation of regioisomers that could act as genotoxic impurities or complicate downstream purification. Following the bromination, the silyl groups are removed under acidic conditions to regenerate the phenolic functionality required for the subsequent coupling reaction with the terpene moiety. The final coupling step, utilizing an acid catalyst like p-toluenesulfonic acid, joins the resorcinol core with a chiral terpene derivative, establishing the critical stereocenters of the cannabidiol molecule. The entire sequence is designed to preserve the integrity of the chiral centers, avoiding the racemization issues that plague other synthetic strategies, thereby ensuring the production of the pharmacologically active enantiomer.
From a process chemistry perspective, the ability to isolate and store the silylated intermediates offers a unique logistical advantage that transforms the manufacturing paradigm. Unlike unstable intermediates that must be consumed immediately in a telescoped process, the di-OTBS-olivetol and its brominated derivative are described as stable, storable, and transportable. This stability allows manufacturers to decouple the production stages, enabling the synthesis of large batches of intermediates during periods of low demand or favorable raw material pricing, and storing them for later conversion into the final API. Such flexibility is invaluable for supply chain heads who must manage volatility in raw material availability and fluctuating market demands. Furthermore, the high yields reported in the experimental examples, such as the 96.8% yield in the bromination step and the quantitative conversion in the silylation step, indicate a highly efficient atom economy. This efficiency translates directly into reduced waste generation and lower solvent consumption per kilogram of product, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing facility.
How to Synthesize Cannabidiol Efficiently
The synthesis of cannabidiol via this patented route involves a series of carefully controlled organic transformations that prioritize safety and selectivity. The process begins with the protection of olivetol, followed by regioselective bromination, desilylation, and finally, the coupling with a chiral terpene. Each step has been optimized to maximize yield while minimizing the formation of byproducts, ensuring that the final product meets the stringent purity requirements of the pharmaceutical industry. The detailed standardized synthesis steps for implementing this route are provided in the guide below.
- Protect olivetol hydroxyl groups using TBSCl and imidazole to form stable di-OTBS-olivetol.
- Perform regioselective bromination using NBS on the silylated intermediate to introduce bromine atoms at the 4 and 6 positions.
- Execute desilylation followed by acid-catalyzed coupling with trans-menthol and subsequent debromination to yield final CBD.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this synthetic route offers compelling economic and operational benefits that extend beyond simple yield metrics. The elimination of elemental bromine removes the need for specialized corrosion-resistant equipment and complex scrubbing systems, leading to significant capital expenditure savings and reduced maintenance costs over the lifecycle of the production facility. Moreover, the use of stable intermediates allows for a more resilient supply chain model, where inventory can be strategically held to buffer against disruptions in the supply of starting materials or sudden spikes in demand for the final API. This decoupling of production stages reduces the risk of batch failures affecting the entire value chain, as intermediates can be quality-controlled and released independently before committing to the final, more valuable synthesis steps.
- Cost Reduction in Manufacturing: The transition from hazardous elemental bromine to solid N-bromosuccinimide eliminates the high costs associated with handling, storage, and disposal of dangerous liquids. This substitution simplifies the regulatory compliance burden and reduces insurance premiums related to chemical storage. Additionally, the ability to run reactions at ambient or near-ambient temperatures significantly lowers energy consumption compared to cryogenic processes, resulting in substantial operational cost savings. The high regioselectivity achieved through silylation minimizes the need for expensive chromatographic purifications, further driving down the cost of goods sold by reducing solvent usage and processing time.
- Enhanced Supply Chain Reliability: The stability of the silylated intermediates means that production does not need to be continuous or linear. Manufacturers can produce and stockpile key intermediates when raw material costs are low, creating a strategic buffer that insulates the supply chain from market volatility. This capability ensures consistent delivery schedules to downstream customers, even in the face of upstream disruptions. The robustness of the chemistry also implies fewer batch failures and higher overall equipment effectiveness, contributing to a more predictable and reliable supply of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reagents and conditions that are manageable in large reactors without exotic engineering controls. The reduction in hazardous waste streams, particularly the avoidance of bromine-contaminated effluents, simplifies wastewater treatment and ensures compliance with increasingly strict environmental regulations. This environmental compatibility not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a critical factor in vendor selection for major pharmaceutical companies committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this patented synthesis method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the feasibility and advantages of the process for potential partners and stakeholders.
Q: Why is silylation used in this CBD synthesis route?
A: Silylation protects the hydroxyl groups and masks the para position of the aromatic system. This ensures that subsequent bromination occurs only at the desired ortho positions relative to the alkyl chain, preventing unwanted side reactions and improving regioselectivity.
Q: Does this process use elemental bromine?
A: No, this method utilizes N-bromosuccinimide (NBS) instead of elemental bromine. This substitution significantly enhances operational safety by avoiding the handling of corrosive and hazardous elemental bromine, especially at low temperatures.
Q: Are the intermediates stable for storage?
A: Yes, the patent explicitly states that the silylated olivetol and silylated brominated olivetol intermediates are stable, storable, and transportable. This allows for flexible production scheduling and inventory management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cannabidiol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of cannabidiol intermediate or API we produce meets the highest international standards. Our expertise in handling complex organic syntheses, particularly those involving sensitive functional groups and chiral centers, positions us as an ideal partner for bringing this patented technology to full commercial fruition.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the specific economic benefits tailored to your production volume and quality requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive efficiency and reliability in your cannabinoid manufacturing operations.
