Advanced Purification Technology For Latamoxef Sodium Ensuring Commercial Scalability And High Purity
The pharmaceutical industry continuously demands higher purity standards for beta-lactam antibiotics to ensure patient safety and regulatory compliance. Patent CN101857601B introduces a groundbreaking purification methodology specifically designed for Latamoxef Sodium, a broad-spectrum oxacephem antibiotic critical for treating various microbial infections. Traditional manufacturing routes often struggle with residual impurities, color discrepancies, and inconsistent content levels, which directly impact the quality of the final drug formulation. This novel approach leverages a sophisticated acid-base transformation coupled with activated carbon adsorption to overcome these historical limitations. By meticulously controlling the pH during the conversion between the sodium salt and the free acid form, manufacturers can effectively isolate the target compound from soluble impurities. The integration of this technology represents a significant leap forward for any reliable Latamoxef Sodium supplier aiming to deliver premium-grade active pharmaceutical ingredients. The method not only enhances the chemical profile of the substance but also streamlines the downstream processing requirements, making it an essential reference for modern antibiotic production facilities seeking to optimize their quality control protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Latamoxef Sodium has relied heavily on direct crystallization or simple recrystallization techniques from various solvent systems. While these methods are straightforward, they frequently fail to remove trace organic impurities and colored by-products that co-precipitate with the target molecule. The resulting bulk drug often exhibits suboptimal HPLC purity profiles and undesirable coloration, which necessitates additional, costly reprocessing steps to meet pharmacopeial standards. Furthermore, conventional methods often suffer from inconsistent yields due to the high solubility of the sodium salt in aqueous environments, leading to significant product loss during mother liquor separation. The inability to effectively separate structurally similar degradation products limits the overall quality of the API, posing risks for clinical safety and efficacy. For procurement managers, these inefficiencies translate into higher production costs and potential supply chain disruptions caused by batch failures. The lack of a robust decolorization step in traditional protocols means that the final product may require extensive washing or secondary purification, further extending the manufacturing lead time and reducing the overall throughput of the production facility.
The Novel Approach
The innovative process described in the patent fundamentally shifts the purification paradigm by exploiting the differential solubility properties of the Latamoxef free acid versus its sodium salt. Instead of attempting to purify the salt directly, the method first converts the crude Latamoxef Sodium into its insoluble free acid form through precise acidification. This phase separation allows for the removal of water-soluble impurities that would otherwise remain trapped in the crystal lattice of the salt. Following filtration, the free acid is dissolved in an organic solvent, creating an ideal environment for activated carbon adsorption to remove hydrophobic impurities and color bodies without significant product loss. The final regeneration of the sodium salt via alkaline adjustment ensures that the product precipitates in a highly pure, crystalline form. This strategic sequence not only drastically simplifies the purification workflow but also significantly enhances the consistency of the final product quality. For stakeholders focused on cost reduction in antibiotic manufacturing, this approach minimizes solvent consumption and reduces the need for complex chromatographic separations, offering a more economically viable path to high-purity API production.
Mechanistic Insights into Acid-Base Transformation and Adsorption
The core chemical mechanism driving this purification success lies in the reversible ionization of the carboxylic acid groups present in the Latamoxef structure. At a controlled acidic pH range of 2 to 2.5, the carboxylate anions are protonated to form the neutral carboxylic acid, which exhibits markedly lower solubility in water compared to the ionic sodium salt. This physicochemical shift forces the Latamoxef molecule out of the aqueous solution, leaving behind polar impurities and inorganic salts in the supernatant. The subsequent dissolution of this precipitate in an organic solvent like ether or ethyl acetate prepares the molecule for the critical decolorization step. Activated carbon, with its extensive surface area and porous structure, selectively adsorbs conjugated organic impurities responsible for coloration through pi-pi interactions and van der Waals forces. This step is crucial for meeting the stringent visual and spectral requirements of modern pharmaceutical standards. The final addition of a base, such as sodium hydroxide, deprotonates the carboxylic acid, regenerating the water-soluble sodium salt which then crystallizes out of the organic-aqueous interface or upon concentration. This cycle effectively acts as a chemical filter, purging the material of both polar and non-polar contaminants.
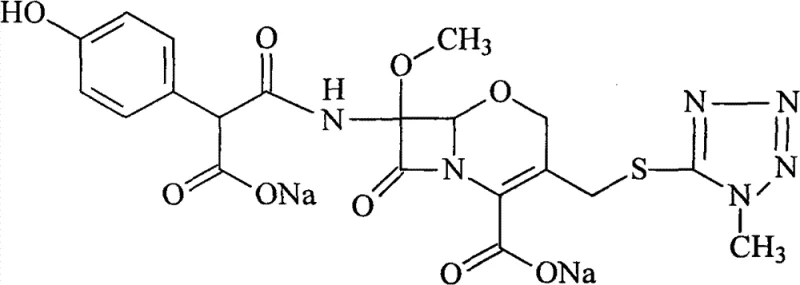
Controlling the impurity profile is paramount for R&D directors concerned with the stability and safety of the final drug product. The acid-base cycle specifically targets degradation products that may have formed during the initial synthesis, such as open-ring beta-lactam derivatives or isomeric by-products. By adjusting the pH to the precise window of 2-2.5, the process ensures maximum precipitation of the target compound while keeping degradation products in solution. The activated carbon treatment further polishes the profile by removing trace catalysts or organic reagents carried over from previous synthetic steps. The result is a product with an HPLC purity consistently exceeding 99.4%, as demonstrated in the patent embodiments. This level of purity reduces the burden on formulation scientists, who no longer need to account for variable impurity loads during drug product development. Moreover, the mild conditions employed during the adsorption phase, typically at room temperature for 20 to 30 minutes, prevent thermal degradation of the sensitive beta-lactam ring, preserving the biological activity of the molecule throughout the purification process.
How to Synthesize Latamoxef Sodium Efficiently
Implementing this purification protocol requires precise control over reaction parameters to ensure reproducibility and high yield. The process begins with the dissolution of crude material in water, followed by the careful addition of acid to reach the critical precipitation point. Operators must monitor the pH closely to avoid over-acidification, which could lead to hydrolysis of the beta-lactam ring. Once the free acid is isolated and dried, the choice of solvent for the adsorption step is critical; ethers and esters have shown superior performance in balancing solubility and impurity removal. The detailed standardized synthesis steps, including specific reagent concentrations, stirring times, and drying temperatures, are outlined in the technical guide below to ensure seamless technology transfer. Adhering to these parameters allows manufacturing teams to achieve yields upwards of 92% while maintaining the high-purity specifications required for global markets. This structured approach minimizes batch-to-batch variability and ensures that the commercial scale-up of complex antibiotics remains robust and predictable.
- Dissolve crude Latamoxef Sodium in water and adjust pH to 2-2.5 using hydrochloric acid to precipitate the insoluble free acid form (latamoxef).
- Dissolve the filtered latamoxef in an organic solvent such as ether, add 0.1-0.2% activated carbon, and stir for 20-30 minutes to adsorb impurities and color bodies.
- Filter the solution to remove carbon, then add sodium hydroxide solution to adjust pH to 8.5-9, precipitating the purified Latamoxef Sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology offers substantial strategic benefits beyond mere chemical purity. The simplification of the process flow reduces the number of unit operations required, which directly correlates to lower operational expenditures and reduced energy consumption. By eliminating the need for complex chromatographic columns or exotic solvents, the facility can rely on standard stainless-steel reactors and filtration equipment, enhancing the overall reliability of the production line. The high yield reported in the patent embodiments suggests a significant reduction in raw material waste, contributing to a more sustainable and cost-effective manufacturing model. Furthermore, the robustness of the method against variations in crude feedstock quality ensures a more stable supply of the finished API, mitigating the risk of production delays. These factors collectively strengthen the supply chain resilience, allowing partners to secure a reliable Latamoxef Sodium supplier capable of meeting demanding volume requirements without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive chromatographic purification steps and the use of common, low-cost reagents like hydrochloric acid and sodium hydroxide significantly lower the direct material costs. The high recovery rate of the product during the precipitation phases minimizes yield loss, ensuring that more of the valuable intermediate is converted into saleable API. Additionally, the reduced processing time and lower energy requirements for drying and solvent recovery contribute to a leaner cost structure. This economic efficiency allows for more competitive pricing strategies in the global market while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial chemicals and standard equipment reduces the risk of supply bottlenecks associated with specialized reagents. The process tolerance to minor variations in operating conditions ensures consistent batch success rates, preventing the stop-start production cycles that often plague complex antibiotic manufacturing. This stability is crucial for maintaining continuous supply to downstream formulation partners, ensuring that market demand is met without interruption. The ability to scale this process from laboratory to commercial production without significant re-engineering further secures the long-term availability of the product.
- Scalability and Environmental Compliance: The process generates less hazardous waste compared to traditional methods that rely on heavy metal catalysts or large volumes of toxic solvents. The aqueous waste streams from the acid-base steps are easier to treat and neutralize, facilitating compliance with increasingly stringent environmental regulations. The use of activated carbon, a regenerable or disposable solid, simplifies waste management protocols. This environmental friendliness not only reduces disposal costs but also aligns with the sustainability goals of modern pharmaceutical companies, enhancing the brand value of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. They are derived from the specific operational parameters and benefits detailed in the patent documentation. Understanding these aspects is vital for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams assessing the quality credentials of their suppliers. The answers provide clarity on the critical control points and the expected outcomes of the process.
Q: What is the primary advantage of the acid-base transformation method for Latamoxef Sodium?
A: The primary advantage is the significant improvement in purity and color. By converting the sodium salt to the free acid form, impurities that remain soluble in the acidic aqueous phase are separated. Subsequent activated carbon adsorption removes trace organic impurities and color bodies, resulting in a final product with HPLC purity exceeding 99.4%.
Q: Which solvents and reagents are optimal for this purification process?
A: According to patent data, hydrochloric acid is the preferred acid for precipitation (pH 2-2.5), and ether is the preferred solvent for dissolving the free acid during the decolorization step. Sodium hydroxide is the preferred base for regenerating the sodium salt at a pH of 8.5-9.
Q: How does this process impact industrial scalability?
A: The process utilizes common industrial reagents and standard unit operations such as filtration, stirring, and pH adjustment. The mild conditions (room temperature stirring for adsorption, 40-50°C drying) and high yields (over 92%) indicate strong suitability for large-scale commercial production without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Latamoxef Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-purity standards demonstrated in the laboratory are maintained at an industrial scale. We understand the critical nature of beta-lactam stability and employ stringent purity specifications and rigorous QC labs to verify every batch against the highest global pharmacopeial standards. Our commitment to quality ensures that the Latamoxef Sodium we supply meets the exacting requirements of R&D directors and regulatory bodies alike, providing a solid foundation for your drug development projects.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance data. By partnering with us, you gain access to a supply chain that prioritizes both technical excellence and commercial reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
