Advanced Low-Temperature Synthetic Route for Erlotinib Intermediates and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, particularly for kinase inhibitors like erlotinib. Patent CN111320586B introduces a transformative methodology for synthesizing erlotinib intermediates, addressing critical bottlenecks found in legacy production techniques. This innovation pivots away from hazardous nitration and noble metal catalysis, opting instead for a controlled oxidative cyclization strategy. By leveraging common raw materials and moderating reaction temperatures, this approach offers a compelling solution for reliable erlotinib intermediate supplier networks aiming to enhance process reliability. The technical breakthrough lies in the strategic replacement of aggressive electrophilic aromatic substitution with a catalytic oxidation protocol, fundamentally altering the impurity profile and thermal demands of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes, such as Route A referenced in patents like CN96102992.7, depend heavily on harsh nitration conditions utilizing fuming nitric acid and concentrated sulfuric acid. These reagents are not only strongly corrosive and volatile but also present severe safety liabilities, including fire risks upon contact with organic substrates and potential for causing severe respiratory and skin burns. Furthermore, the subsequent reduction of the nitro group typically necessitates precious metal catalysts like platinum or palladium. These metals are prone to remaining as trace residues in the final product, creating significant regulatory hurdles regarding heavy metal limits and necessitating costly purification steps to meet stringent pharmaceutical standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN111320586B utilizes a multi-step sequence beginning with etherification followed by a catalytic oxidation using peroxyacetic acid. This route completely bypasses the need for fuming nitric acid and noble metal catalysts, thereby mitigating both safety risks and heavy metal contamination issues. The process operates under significantly milder thermal conditions, particularly during the critical ring-closing step, where temperatures are maintained between 80°C and 100°C rather than the exceeding 160°C required by prior art. This reduction in thermal stress not only lowers energy consumption but also minimizes the formation of thermal degradation byproducts, ensuring a cleaner reaction profile suitable for cost reduction in pharmaceutical intermediates manufacturing.
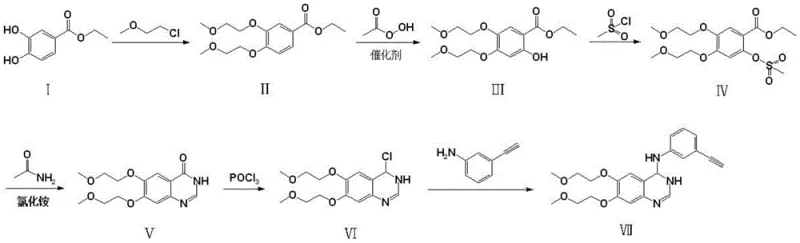
Mechanistic Insights into Catalytic Oxidation and Cyclization
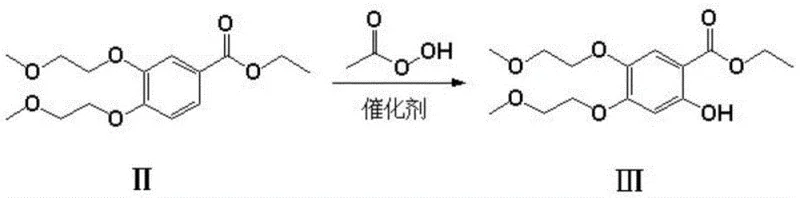
The core of this technological advancement resides in the oxidation step, where compound II is converted to compound III using peroxyacetic acid in the presence of transition metal catalysts such as ferrous sulfate, copper oxide, or sulfonated cobalt phthalocyanine. This reaction is conducted under pressurized conditions (2.0-3.0 MPa) with gases like ozone or oxygen, facilitating a highly selective hydroxylation of the aromatic ring. The mechanism likely involves the generation of reactive oxygen species that attack the electron-rich aromatic system activated by the ether groups, achieving regioselectivity that traditional nitration struggles to match without generating isomeric impurities. This precise control over the oxidation state is pivotal for maintaining high purity throughout the synthesis chain.
Following oxidation, the synthesis proceeds through mesylation and a subsequent cyclization reaction to form the quinazolinone core (Compound V). Unlike conventional methods that require heating ammonium formate in formamide above 160°C, this novel route employs ammonium chloride and formamide at temperatures ranging from 80°C to 100°C. The lower temperature regime reduces the kinetic energy available for side reactions, effectively suppressing the formation of polymeric tars and other high-molecular-weight impurities. This mechanistic refinement ensures that the resulting heterocyclic scaffold possesses the structural integrity required for the final substitution with m-aminophenylacetylene, ultimately yielding high-purity erlotinib with minimal downstream processing.
How to Synthesize Erlotinib Intermediates Efficiently
The execution of this synthetic pathway requires precise control over reaction parameters, particularly pressure and temperature during the oxidation phase. Operators must ensure the complete dissolution of reactants in supercritical or solvent systems before initiating the oxidation to prevent localized hot spots. The subsequent cyclization step demands careful monitoring of pH and water content to drive the equilibrium towards the desired quinazolinone product. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis steps outlined below which encapsulate the critical process controls identified in the patent documentation.
- Etherification of the starting phenol with 2-chloroethyl methyl ether under supercritical or basic conditions to form the bis-ether intermediate.
- Catalytic oxidation of the ether intermediate using peroxyacetic acid and transition metal catalysts (e.g., ferrous sulfate) to introduce the hydroxyl group.
- Mesylation followed by cyclization with ammonium chloride and formamide at reduced temperatures (80-100°C) to form the quinazolinone core.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial strategic benefits by decoupling production from the volatility of precious metal markets and the logistical complexities of handling hazardous acids. The reliance on common industrial chemicals like peroxyacetic acid and ammonium chloride ensures a stable supply chain, reducing the risk of production stoppages due to raw material shortages. Furthermore, the elimination of heavy metal catalysts removes the need for expensive scavenging resins or complex filtration systems, streamlining the manufacturing workflow and reducing the overall cost of goods sold. This efficiency makes the process highly attractive for partners seeking long-term stability in their API supply chains.
- Cost Reduction in Manufacturing: The exclusion of platinum and palladium catalysts eliminates a significant variable cost component associated with noble metals. Additionally, the avoidance of aggressive nitration reagents reduces the expenditure on specialized corrosion-resistant equipment and hazardous waste disposal. The lower reaction temperatures further contribute to energy savings, as less thermal input is required to drive the cyclization and substitution reactions to completion. These cumulative factors result in a leaner cost structure that enhances competitiveness in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents such as 2-chloroethyl methyl ether and formamide, the process mitigates the risk of supply disruptions often associated with specialty chemicals. The robustness of the oxidation step, which tolerates various transition metal catalysts, provides flexibility in sourcing, allowing manufacturers to switch suppliers based on availability without compromising reaction efficacy. This adaptability ensures consistent production schedules and reliable delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: The milder reaction conditions facilitate easier scale-up from pilot batches to commercial production volumes, as the thermal management requirements are less demanding than those of high-temperature nitration routes. Moreover, the reduction in hazardous waste generation, specifically the absence of spent mixed acids and heavy metal sludge, simplifies environmental compliance and wastewater treatment. This alignment with green chemistry principles supports sustainable manufacturing practices and reduces the regulatory burden on production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel erlotinib synthesis route. These insights are derived directly from the comparative data and experimental results presented in the patent literature, providing a clear understanding of the operational benefits. Stakeholders can use this information to evaluate the feasibility of adopting this technology for their own production lines or procurement strategies.
Q: Why is the elimination of noble metal catalysts critical for API production?
A: Conventional routes often utilize platinum or palladium for nitro reduction, which poses a significant risk of heavy metal residue in the final active pharmaceutical ingredient. Removing these traces requires expensive purification steps. The novel route avoids these metals entirely, simplifying downstream processing and ensuring stricter compliance with ICH Q3D guidelines.
Q: How does the new oxidative method improve process safety compared to traditional nitration?
A: Traditional methods rely on fuming nitric acid and concentrated sulfuric acid, which are highly corrosive, volatile, and pose fire risks when contacting organic matter. The new method employs peroxyacetic acid oxidation under controlled pressure, significantly reducing the hazard profile and eliminating the need for handling aggressive mixed acids.
Q: What are the yield advantages of this synthetic pathway?
A: Data indicates that the total yield from the intermediate compound II to compound V exceeds 50%, whereas existing synthesis methods typically achieve less than 35%. This substantial increase in efficiency directly translates to lower material consumption and improved cost-effectiveness for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Erlotinib Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex laboratory innovations into commercial reality, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the oxidative cyclization processes described in CN111320586B to meet stringent purity specifications required by global regulatory bodies. With rigorous QC labs and a commitment to process safety, we ensure that every batch of erlotinib intermediate delivered meets the highest standards of quality and consistency, supporting our partners in bringing life-saving medications to market faster.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition to this superior manufacturing technology.
