Advanced Purification of Ursodeoxycholic Acid via Diisopropylethylamine Salt Crystallization for Commercial Scale-Up
Advanced Purification of Ursodeoxycholic Acid via Diisopropylethylamine Salt Crystallization for Commercial Scale-Up
The pharmaceutical landscape for hepatobiliary therapeutics continues to evolve, driven by the critical need for high-purity active pharmaceutical ingredients (APIs) that meet rigorous global safety standards. A pivotal advancement in this domain is detailed in Chinese Patent CN110669091A, which discloses a sophisticated purification method for ursodeoxycholic acid (UDCA), a key therapeutic agent approved by the FDA for treating primary biliary cirrhosis and cholesterol gallstones. This technology represents a significant departure from traditional extraction and purification methodologies, addressing long-standing inefficiencies in yield and purity. By leveraging a novel salt formation strategy using diisopropylethylamine, the process achieves exceptional selectivity, effectively separating UDCA from structurally similar impurities such as chenodeoxycholic acid (CDCA) and 7-ketolithocholic acid (7K-LCA). For R&D directors and procurement specialists seeking a reliable ursodeoxycholic acid supplier, understanding the mechanistic underpinnings and commercial viability of this patented route is essential for securing a robust supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial purification of ursodeoxycholic acid has been plagued by significant technical and economic bottlenecks that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. Conventional approaches often rely on organic solvent recrystallization, typically utilizing ethyl acetate, to selectively dissolve and remove impurities. However, the solubility differences between ursodeoxycholic acid and its analogues in ethyl acetate are marginal, necessitating the use of excessive solvent volumes and multiple recrystallization cycles. This not only drives up energy consumption due to the need for extensive solvent recovery and distillation but also results in substantial product loss, negatively impacting overall yield. Furthermore, alternative pathways described in prior art, such as US Patent 4282161, involve complex multi-step sequences including esterification with methanol, followed by saponification and acidification. These lengthy processes extend production lead times and introduce additional opportunities for product degradation and impurity generation, making them less attractive for modern, lean manufacturing environments.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology disclosed in CN110669091A introduces a streamlined, high-efficiency purification protocol centered on the formation of a diisopropylethylammonium salt. This innovative approach capitalizes on the distinct solubility profile of the UDCA-DIPEA salt in tetrahydrofuran (THF), which differs markedly from that of the impurities. By converting the crude acid into its corresponding ammonium salt, the process transforms a difficult separation problem based on subtle polarity differences into a robust crystallization event driven by ionic interactions and temperature control. This method eliminates the need for hazardous esterification reagents and the associated hydrolysis steps, significantly simplifying the workflow. The result is a process that is not only operationally simpler but also delivers high-purity ursodeoxycholic acid with purity levels exceeding 99.5%, directly addressing the stringent quality requirements of downstream formulation teams.
Mechanistic Insights into Diisopropylethylamine-Mediated Salt Crystallization
The core of this purification technology lies in the precise acid-base reaction between ursodeoxycholic acid and diisopropylethylamine (DIPEA) within a tetrahydrofuran medium. When the crude mixture containing UDCA is dissolved in THF and treated with DIPEA at elevated temperatures ranging from 50°C to 65°C, a rapid proton transfer occurs, generating the diisopropylethylammonium ursodeoxycholate salt in situ. This salt formation is critical because it alters the molecular lattice energy and solvation shell of the bile acid derivative. Unlike the free acid, which may co-crystallize with impurities like CDCA due to structural homology, the bulky diisopropylethylammonium cation imposes steric constraints that favor the formation of a highly ordered, pure crystal lattice specific to the UDCA salt. This selectivity is the fundamental driver behind the method's ability to exclude contaminants that would otherwise persist through standard recrystallization.
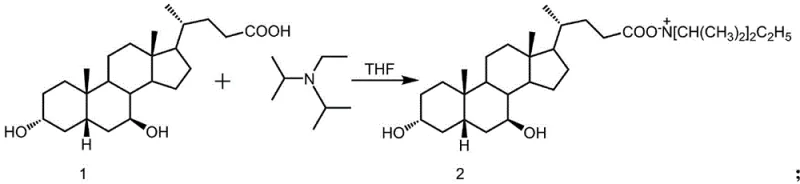
Following the salt formation, the solution undergoes a controlled cooling phase to 0-5°C, inducing supersaturation and subsequent crystallization of the pure salt. The impurities, remaining more soluble in the THF mother liquor due to their inability to form stable salts with the same crystallization kinetics or their different ionic characteristics, are effectively rejected from the growing crystal lattice. The isolated salt is then subjected to a hydrolysis step where it is suspended in ultrapure water and treated with sodium hydroxide. This regenerates the free acid form of UDCA while keeping the DIPEA in the aqueous phase or allowing it to be removed during the dichloromethane extraction step. Finally, careful pH adjustment with dilute hydrochloric acid precipitates the purified UDCA. This mechanism ensures that residual amines and organic impurities are washed away, resulting in a final product that meets rigorous pharmacopeial standards for residual solvents and related substances.
How to Synthesize Ursodeoxycholic Acid Efficiently
The implementation of this purification route requires careful attention to temperature gradients and stoichiometric ratios to maximize yield and purity. The process begins with the dissolution of the crude starting material, typically obtained from the reduction of 7-ketolithocholic acid, in a substantial volume of tetrahydrofuran to ensure complete solubilization before the addition of the amine base. The reaction temperature must be maintained strictly within the 50-65°C window to facilitate complete salt formation without degrading the thermally sensitive bile acid structure. Once the salt is formed and crystallized at low temperatures, the subsequent hydrolysis and pH control steps are critical for recovering the free acid in its most stable polymorphic form. For detailed operational parameters and specific equipment recommendations, please refer to the standardized synthesis guide below.
- Dissolve crude ursodeoxycholic acid mixture in tetrahydrofuran (THF) and react with diisopropylethylamine at 50-65°C to form the ammonium salt.
- Cool the resulting salt solution to 0-5°C to induce selective crystallization, followed by filtration and drying to isolate the pure salt intermediate.
- Hydrolyze the isolated salt in ultrapure water using sodium hydroxide and dichloromethane extraction, followed by acidification with hydrochloric acid to precipitate pure ursodeoxycholic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DIPEA-mediated purification technology offers transformative benefits that extend far beyond mere technical superiority. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly translates to enhanced operational efficiency and reduced overhead costs. By eliminating the multi-step esterification and saponification sequences required in older patents, manufacturers can significantly reduce the number of unit operations, thereby lowering labor costs, minimizing equipment occupancy time, and reducing the potential for human error. This streamlined approach allows for faster batch turnover rates, enabling suppliers to respond more agilely to market demand fluctuations and ensuring a more consistent flow of materials to downstream API producers.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the reduction in solvent consumption and energy usage. Unlike traditional methods that require vast quantities of ethyl acetate and repeated distillation cycles to achieve marginal purity improvements, this novel route utilizes a highly selective crystallization mechanism that requires significantly less solvent volume per kilogram of product. Furthermore, the avoidance of esterification reagents like methanol and the associated alkaline hydrolysis steps removes the need for handling and disposing of large volumes of reactive chemicals. This reduction in raw material intensity, combined with lower energy demands for heating and cooling cycles, results in a substantially lower cost of goods sold (COGS), providing a competitive pricing advantage in the global marketplace.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method enhances the reliability of the commercial scale-up of complex bile acid derivatives. The reagents employed, such as tetrahydrofuran and diisopropylethylamine, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that can plague specialized reagent-dependent processes. Additionally, the high selectivity of the crystallization step means that the process is more tolerant to variations in the quality of the crude starting material. This tolerance reduces the pressure on upstream suppliers to deliver ultra-high purity intermediates, thereby diversifying the potential sourcing options for raw materials and mitigating the risk of supply disruptions caused by single-source dependencies.
- Scalability and Environmental Compliance: The environmental footprint of pharmaceutical manufacturing is an increasingly critical factor for supply chain sustainability. This purification method aligns well with green chemistry principles by minimizing waste generation and solvent usage. The simplified workup procedure, which avoids the generation of large volumes of saline wastewater typical of saponification processes, eases the burden on wastewater treatment facilities. Moreover, the ease of scaling crystallization processes from laboratory to pilot and eventually to full commercial production ensures that the technology can be rapidly deployed to meet increasing global demand for ursodeoxycholic acid without requiring massive capital expenditure on new, specialized reactor infrastructure.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented purification technology, we have compiled a set of frequently asked questions based on the specific details outlined in the patent documentation. These inquiries address common concerns regarding impurity profiles, regulatory compliance, and process scalability, providing decision-makers with the clarity needed to evaluate this technology for their supply chains. The answers provided are derived directly from the experimental data and beneficial effects reported in the patent, ensuring accuracy and relevance for technical assessment.
Q: How does the DIPEA salt method improve purity compared to traditional solvent recrystallization?
A: Traditional methods using ethyl acetate often struggle with similar solubility profiles between ursodeoxycholic acid and impurities like chenodeoxycholic acid. The DIPEA salt formation creates a distinct chemical species with vastly different solubility characteristics in THF, allowing for highly selective crystallization that effectively separates the target molecule from structurally similar bile acid impurities, achieving purity levels exceeding 99.5%.
Q: What are the regulatory advantages of using diisopropylethylamine over triethylamine?
A: While triethylamine has been used in prior art, it presents significant challenges in meeting pharmaceutically acceptable residue limits due to its high volatility and difficulty in complete removal. Diisopropylethylamine, being more sterically hindered and having different physicochemical properties, facilitates easier removal during the hydrolysis and extraction phases, ensuring the final API meets stringent residual solvent and amine specifications required by global regulatory bodies.
Q: Is this purification process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It eliminates the multi-step esterification and saponification sequences found in older patents, reducing the overall unit operations. The reliance on standard crystallization techniques (cooling crystallization) and common solvents like THF and water makes it highly adaptable to existing reactor infrastructure without requiring specialized high-pressure or high-temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursodeoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO specializing in complex pharmaceutical intermediates, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this advanced purification technology to life. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of ursodeoxycholic acid meets the exacting standards required for FDA-approved drug products. We understand the critical nature of hepatobiliary therapeutics and are committed to delivering materials that support patient safety and therapeutic efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge purification method for their ursodeoxycholic acid supply needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments that can optimize your supply chain for the future. Let us help you secure a sustainable, high-quality source of this vital API intermediate.
