Advanced Palladium-Catalyzed Synthesis of Unsaturated Nitriles for Commercial Scale Manufacturing
The pharmaceutical and fine chemical industries constantly seek robust methodologies for constructing carbon-carbon bonds, particularly for generating versatile building blocks like unsaturated nitriles. Patent CN108586288B discloses a groundbreaking synthetic route for compounds of formula (3), utilizing a sophisticated palladium-catalyzed cross-coupling strategy. This innovation addresses the longstanding challenge of efficiently functionalizing allyl cyanide derivatives with diverse aryl groups. By employing a unique comprehensive reaction system comprising palladium acetate, 2,2'-bipyridine, silver acetate, and trifluoroacetic acid in tetrahydrofuran, the method achieves exceptional yields exceeding 80% under optimized conditions. This technical breakthrough offers a reliable pathway for producing high-purity pharmaceutical intermediates, ensuring that downstream synthesis of active pharmaceutical ingredients (APIs) can proceed with minimal impurity burdens. The strategic selection of reagents not only enhances reaction efficiency but also simplifies the purification process, making it an attractive candidate for commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl butenenitrile compounds has relied on a variety of disparate methods, each carrying significant drawbacks that hinder large-scale application. Prior art techniques, such as those involving Vilsmeier reagents or Mitsunobu reactions, often necessitate harsh reaction conditions, toxic reagents, or complex multi-step sequences that erode overall atom economy. For instance, traditional approaches might require the pre-functionalization of substrates into unstable halides or the use of stoichiometric amounts of expensive metal promoters, leading to substantial waste generation and increased production costs. Furthermore, many conventional routes struggle with stereoselectivity, producing mixtures of E/Z isomers that require difficult and costly separation processes. These inefficiencies create bottlenecks in the supply chain for key organic building blocks, limiting the ability of manufacturers to respond rapidly to market demands for novel drug candidates. The reliance on less predictable catalytic systems often results in batch-to-batch variability, posing a significant risk for quality control in regulated industries.
The Novel Approach
In stark contrast, the methodology described in CN108586288B introduces a streamlined, direct coupling protocol that bypasses many of these historical hurdles. The core innovation lies in the direct reaction between allyl cyanide derivatives (formula 1) and aryl boronic acids (formula 2), leveraging the stability and commercial availability of boronic acid reagents. 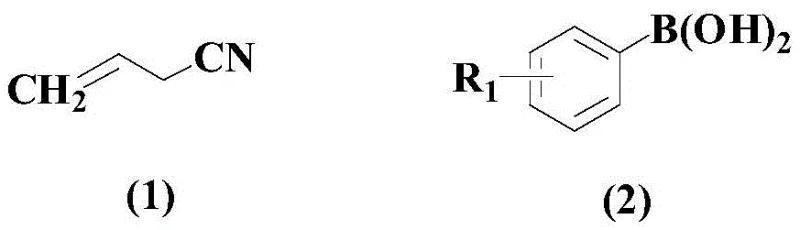 This approach eliminates the need for pre-activating the cyanide component into reactive halides, thereby reducing raw material costs and handling hazards. The use of a specific palladium catalyst system ensures high conversion rates while maintaining excellent functional group tolerance, allowing for the introduction of diverse substituents such as halogens, alkyls, and alkoxy groups without side reactions. By operating in a homogeneous phase with THF as the solvent, the process facilitates efficient heat and mass transfer, which is critical for maintaining consistent product quality during scale-up. This novel route represents a paradigm shift towards greener, more economical synthesis of unsaturated nitriles.
This approach eliminates the need for pre-activating the cyanide component into reactive halides, thereby reducing raw material costs and handling hazards. The use of a specific palladium catalyst system ensures high conversion rates while maintaining excellent functional group tolerance, allowing for the introduction of diverse substituents such as halogens, alkyls, and alkoxy groups without side reactions. By operating in a homogeneous phase with THF as the solvent, the process facilitates efficient heat and mass transfer, which is critical for maintaining consistent product quality during scale-up. This novel route represents a paradigm shift towards greener, more economical synthesis of unsaturated nitriles.
Mechanistic Insights into Pd-Catalyzed Oxidative Coupling
The success of this synthesis hinges on the intricate interplay between the palladium catalyst, the bipyridine ligand, and the silver oxidant within an acidic environment. The catalytic cycle likely initiates with the coordination of the palladium species to the olefinic bond of the allyl cyanide, followed by oxidative addition or activation of the C-H bond adjacent to the nitrile group. The presence of 2,2'-bipyridine stabilizes the palladium center, preventing aggregation into inactive metallic palladium black, which is a common deactivation pathway in such coupling reactions. Subsequently, transmetallation occurs with the aryl boronic acid, transferring the aryl group to the palladium center. 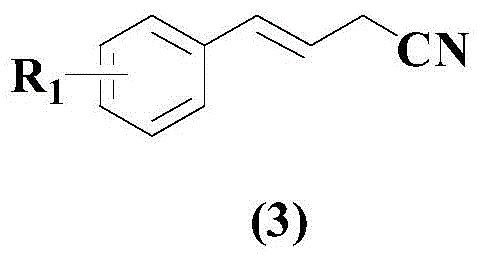 The silver acetate plays a dual role: it acts as an oxidant to regenerate the active Pd(II) species from Pd(0) and potentially assists in the activation of the boronic acid. The acidic additive, specifically trifluoroacetic acid, is crucial for protonating intermediate species and facilitating the final reductive elimination step to release the product. This precise tuning of the reaction environment ensures that the catalytic turnover number remains high, minimizing the required catalyst loading and reducing residual metal contamination in the final product.
The silver acetate plays a dual role: it acts as an oxidant to regenerate the active Pd(II) species from Pd(0) and potentially assists in the activation of the boronic acid. The acidic additive, specifically trifluoroacetic acid, is crucial for protonating intermediate species and facilitating the final reductive elimination step to release the product. This precise tuning of the reaction environment ensures that the catalytic turnover number remains high, minimizing the required catalyst loading and reducing residual metal contamination in the final product.
Impurity control is another critical aspect managed by this specific mechanistic design. The choice of trifluoroacetic acid over weaker acids like acetic acid significantly suppresses the formation of homocoupling byproducts of the boronic acid, which are common contaminants in Suzuki-type reactions. Furthermore, the specific molar ratios defined in the patent, such as a 1:1.5 to 2.5 ratio of substrate to oxidant, ensure that the oxidation potential is sufficient to drive the reaction to completion without causing over-oxidation of the sensitive nitrile or olefin functionalities. The reaction temperature window of 70 to 100°C is optimized to balance reaction kinetics with thermal stability, preventing decomposition of the product or polymerization of the olefinic starting materials. By strictly adhering to these parameters, manufacturers can achieve a clean impurity profile, reducing the burden on downstream purification steps like crystallization or chromatography. This level of control is essential for meeting the stringent purity specifications required for pharmaceutical grade intermediates.
How to Synthesize Unsaturated Nitrile Efficiently
To implement this synthesis effectively, operators must carefully prepare the reaction mixture by combining the allyl cyanide derivative and the aryl boronic acid in the specified molar ratios. The addition of the palladium acetate catalyst and 2,2'-bipyridine ligand should be performed under inert atmosphere conditions to prevent premature oxidation of the catalyst before the reaction begins. Silver acetate and trifluoroacetic acid are then introduced to the THF solvent system, creating the active catalytic environment necessary for the transformation. The detailed standardized synthesis steps see the guide below.
- Combine allyl cyanide derivative, aryl boronic acid, palladium acetate catalyst, 2,2'-bipyridine ligand, silver acetate oxidant, and trifluoroacetic acid in THF solvent.
- Heat the reaction mixture to 70-100°C with stirring and maintain for 20-40 hours to ensure complete conversion.
- Perform post-treatment by pouring into ethyl acetate, washing with brine, drying over sodium sulfate, and purifying via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by utilizing widely available and cost-effective raw materials. Aryl boronic acids are commodity chemicals produced on a massive scale for Suzuki couplings, ensuring a stable and competitive supply market that mitigates the risk of raw material shortages. The elimination of exotic or highly specialized reagents means that purchasing managers can leverage existing vendor relationships to secure favorable pricing, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. Additionally, the use of tetrahydrofuran (THF) as the primary solvent aligns with standard industry practices, allowing facilities to utilize existing solvent recovery infrastructure without requiring significant capital investment in new equipment. This compatibility with standard operational protocols simplifies the technology transfer process from R&D to pilot and commercial scales.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by employing a low loading of palladium catalyst relative to the substrate, which minimizes the expense associated with precious metal consumption. Since the catalyst system is highly efficient, the need for expensive ligand additives is reduced to a simple bipyridine derivative, further lowering the bill of materials. The high yield obtained under these conditions means that less starting material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final product. Moreover, the simplified workup procedure involving standard extraction and chromatography reduces labor hours and solvent consumption during purification, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: By relying on stable solid reagents like boronic acids and silver salts rather than sensitive organometallic reagents like Grignards or organolithiums, the process enhances safety and storage stability. This stability translates to longer shelf lives for raw materials, reducing waste due to expiration and allowing for bulk purchasing strategies that buffer against market volatility. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by minor fluctuations in environmental parameters, ensuring consistent on-time delivery to customers. This reliability is critical for maintaining the continuity of supply chains for downstream API manufacturers who operate on just-in-time inventory models.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and atmospheric pressure, removing the need for high-pressure autoclaves or cryogenic cooling systems, which simplifies reactor design and maintenance. The waste stream generated is primarily composed of silver salts and organic solvents, both of which can be effectively treated or recycled using established industrial waste management protocols. The absence of heavy metal contaminants like chromium or mercury in the reagent list simplifies regulatory compliance and environmental reporting. As production scales from kilograms to tons, the linear scalability of this batch process ensures that yield and quality remain consistent, facilitating rapid capacity expansion to meet growing market demand without compromising on environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed synthesis. These answers are derived directly from the experimental data and comparative studies presented in the patent documentation to provide clarity on process parameters. Understanding these nuances is vital for process chemists aiming to adapt this method for specific substrate scopes or larger production batches.
Q: Why is palladium acetate preferred over other palladium sources in this synthesis?
A: Experimental data indicates that palladium acetate provides significantly higher yields compared to alternatives like palladium chloride or palladium trifluoroacetate, likely due to optimal solubility and activation kinetics in the specific acidic medium.
Q: What role does silver acetate play in the reaction mechanism?
A: Silver acetate acts as a critical oxidant to regenerate the active palladium species. Substituting it with other oxidants like ammonium persulfate results in a marked decrease in product yield, highlighting its unique compatibility with the catalytic cycle.
Q: Can this method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the use of common solvents like THF and stable reagents like boronic acids suggests high scalability. The robust reaction conditions (70-100°C) are well-suited for standard stainless steel reactors used in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Unsaturated Nitrile Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced catalytic technologies like the one described in CN108586288B to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to full-scale manufacturing. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify the identity and quality of every batch. Our commitment to technical excellence means we can rapidly troubleshoot and optimize reaction conditions to maximize yield and minimize impurities for your specific unsaturated nitrile requirements.
We invite you to collaborate with us to explore how this innovative synthesis can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to be your long-term strategic partner in the global supply chain.
