Advanced Biocatalytic Synthesis of Pregabalin Chiral Intermediate for Commercial Scale-up
Advanced Biocatalytic Synthesis of Pregabalin Chiral Intermediate for Commercial Scale-up
The pharmaceutical industry continuously seeks more efficient and sustainable pathways for producing high-value active pharmaceutical ingredients (APIs) and their precursors. A pivotal development in this domain is documented in Chinese Patent CN112359036B, which discloses a novel nitrilase mutant with significantly improved catalytic activity and reaction specificity. This biotechnology breakthrough addresses the critical challenges associated with the synthesis of pregabalin, a first-line treatment for neuropathic pain and epilepsy. The patent details the engineering of specific amino acid substitutions, namely V82L and C237S, within the BaNITmut backbone, resulting in biocatalysts capable of operating at high substrate concentrations while minimizing unwanted side reactions. For R&D directors and procurement specialists, this technology represents a paradigm shift towards greener, more cost-effective manufacturing of chiral intermediates, specifically (S)-3-cyano-5-methylhexanoic acid, which is the direct precursor to pregabalin.
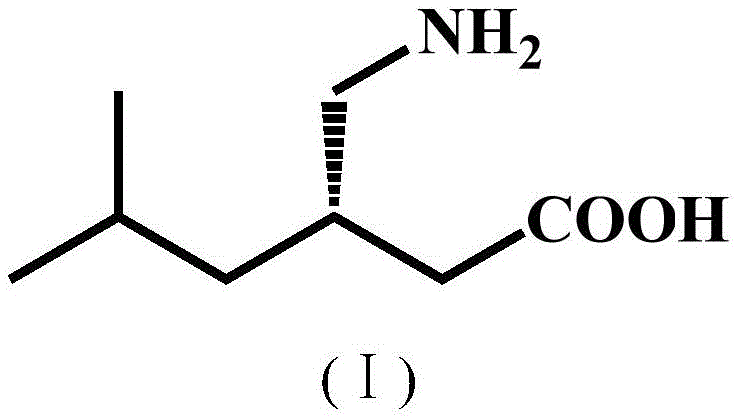
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral intermediates for gabapentinoids like pregabalin has relied on chemical resolution or asymmetric hydrogenation, processes that often suffer from inherent inefficiencies. Chemical routes frequently require harsh reaction conditions, expensive chiral catalysts, and complex purification steps to remove toxic metal residues, which poses significant environmental and regulatory burdens. Furthermore, earlier generations of biocatalysts, while greener, often struggled with low substrate tolerance and poor stereoselectivity. For instance, prior art nitrilases exhibited limited activity when faced with high concentrations of racemic isobutyl succinonitrile (IBSN), leading to low space-time yields that are economically unviable for large-scale production. Additionally, a persistent issue in nitrilase-catalyzed hydrolysis is the competing nitrile hydratase activity, which generates amide by-products instead of the desired carboxylic acid. This side reaction not only consumes the substrate but also complicates the downstream isolation of the pure chiral acid, thereby inflating production costs and reducing overall process yield.
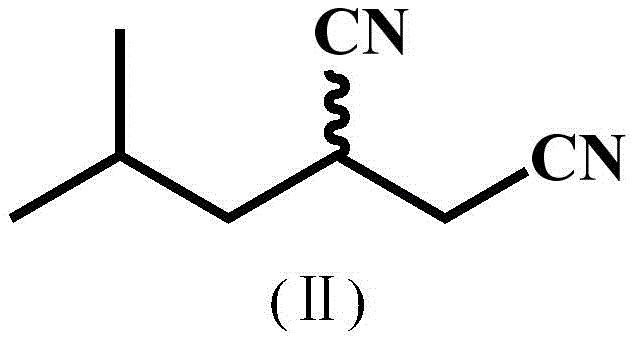
The Novel Approach
The innovative strategy presented in the patent overcomes these historical bottlenecks through precise protein engineering. By utilizing directed evolution techniques, researchers have developed the BaNITmut/V82L single mutant and the BaNITmut/V82L/C237S double mutant. These engineered enzymes display a remarkable ability to hydrolyze racemic IBSN with high stereoselectivity, effectively distinguishing between the enantiomers to produce the desired (S)-configuration. The structural transformation involves the selective hydrolysis of one nitrile group in the dinitrile substrate while leaving the other intact, a delicate chemical feat. As illustrated by the conversion of the substrate structure to the product structure, the process achieves high atom economy. Crucially, the new mutants exhibit suppressed nitrile hydratase activity, drastically lowering the generation of the amide impurity. This improvement means that the reaction mixture contains a much higher proportion of the desired acid product, simplifying the workup procedure and enhancing the overall purity of the final intermediate without the need for extensive chromatographic separation.
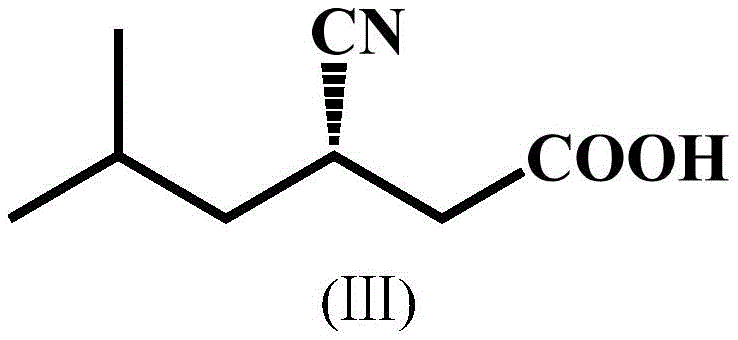
Mechanistic Insights into Nitrilase-Catalyzed Stereoselective Hydrolysis
The enhanced performance of the BaNITmut/V82L/C237S mutant can be attributed to specific conformational changes within the enzyme's active site that favor the binding and hydrolysis of the (S)-enantiomer of IBSN. The mutation of Valine to Leucine at position 82 (V82L) likely alters the hydrophobic pocket surrounding the substrate, optimizing the steric fit for the isobutyl group of the target enantiomer. Furthermore, the additional mutation of Cysteine to Serine at position 237 (C237S) appears to play a critical role in modulating the catalytic triad or the water channel access, thereby accelerating the hydrolysis rate while simultaneously disfavoring the hydration pathway that leads to amide formation. This dual improvement in activity and specificity is rare and highly valuable. The mechanism proceeds through a covalent acyl-enzyme intermediate, where the nitrile carbon is attacked by a catalytic cysteine residue (distinct from the mutation site), followed by hydrolysis to release the carboxylic acid. The high E value (>500) reported for these mutants indicates an exceptional kinetic resolution capability, ensuring that the unreacted (R)-enantiomer remains largely untouched, which prevents contamination of the product stream and maximizes the theoretical yield of the chiral building block.
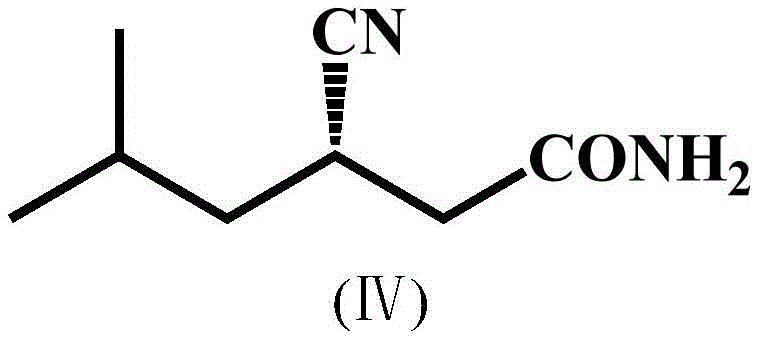
Controlling impurity profiles is paramount in pharmaceutical manufacturing, and the reduction of the amide by-product is a standout feature of this technology. In conventional nitrilase processes, the formation of the amide derivative (Formula IV) is a common side reaction that competes with the desired hydrolysis to the acid. This amide impurity is structurally similar to the product, making it difficult to remove via standard crystallization or extraction techniques, often necessitating costly enzymatic coupling with amidases or rigorous chromatography. The patent data reveals that the double mutant reduces the amide content from 6.2% in the parent strain down to just 2.28%. This substantial decrease in by-product formation is not merely a statistical improvement; it translates directly to operational simplicity. By minimizing the presence of the amide, the downstream processing train can be shortened, solvent usage can be reduced, and the risk of carry-over impurities into the final API is mitigated. This level of purity control is essential for meeting the stringent regulatory standards required for generic and branded drug production.
How to Synthesize (S)-3-cyano-5-methylhexanoic acid Efficiently
The implementation of this biocatalytic route involves a streamlined fermentation and bioconversion process that is amenable to industrial scale-up. The method utilizes recombinant E. coli cells expressing the optimized nitrilase mutants as whole-cell biocatalysts, eliminating the need for expensive enzyme purification steps. The process begins with the cultivation of the engineered strains in a standard LB medium supplemented with kanamycin to maintain plasmid stability. Upon reaching the optimal cell density, protein expression is induced using IPTG, prompting the bacteria to produce high levels of the active nitrilase. The harvested wet cells are then suspended in a buffered aqueous solution containing the racemic IBSN substrate. The reaction proceeds under mild conditions (30°C, pH 8.0), leveraging the enzyme's natural affinity for the substrate to drive the stereoselective hydrolysis. Detailed standardized synthesis steps for replicating this high-efficiency pathway are provided in the guide below.
- Design site-directed mutagenesis primers targeting specific amino acid positions (e.g., V82L, C237S) on the parent nitrilase gene sequence.
- Perform overlap extension PCR using the parent plasmid as a template to introduce the desired mutations, followed by DpnI digestion to remove methylated template DNA.
- Transform the mutated plasmids into competent E. coli BL21(DE3) cells, screen for positive clones via sequencing, and induce expression for biocatalytic application.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nitrilase mutant technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the manufacturing workflow. By utilizing a biocatalyst that operates with high specificity and low by-product formation, manufacturers can eliminate several unit operations typically required for impurity removal. This reduction in process complexity directly correlates to lower capital expenditure (CAPEX) for equipment and reduced operational expenditure (OPEX) regarding solvents, energy, and labor. Furthermore, the ability of the mutant enzymes to tolerate high substrate concentrations (up to 175 g/L) means that reactors can produce more product per batch, significantly improving the volumetric productivity of the facility. This efficiency gain allows for smaller reactor footprints to achieve the same output, optimizing factory space utilization.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction of downstream purification steps lead to substantial cost savings. Since the enzymatic process avoids the use of heavy metals, there is no need for expensive metal scavenging resins or rigorous testing for residual metals, which are costly compliance requirements. Additionally, the high conversion rates and reduced amide by-product formation mean that less raw material is wasted, improving the overall mass balance of the synthesis. The qualitative reduction in waste generation also lowers the costs associated with environmental compliance and waste disposal, contributing to a leaner and more profitable production model for the chiral intermediate.
- Enhanced Supply Chain Reliability: The robustness of the recombinant E. coli expression system ensures a consistent and reliable supply of the biocatalyst. Unlike chemical catalysts that may suffer from batch-to-batch variability or supply chain disruptions for precious metals, the biological catalyst can be reproduced indefinitely through fermentation. The high stability of the mutants under reaction conditions further ensures that the process is reproducible on a multi-ton scale. This reliability is crucial for securing long-term supply contracts for API manufacturers, as it minimizes the risk of production delays caused by catalyst failure or inconsistent quality. The use of readily available fermentation substrates also insulates the supply chain from the volatility of petrochemical feedstock prices.
- Scalability and Environmental Compliance: The process aligns perfectly with green chemistry principles, utilizing water as the primary solvent and operating at ambient temperatures and pressures. This mild operational profile significantly reduces the energy consumption compared to high-temperature chemical syntheses. The biodegradable nature of the biocatalyst and the aqueous waste streams simplifies effluent treatment, making it easier for facilities to meet increasingly strict environmental regulations. The scalability of the fermentation process is well-established in the industry, allowing for seamless transition from pilot scale to commercial production volumes of 100 MT or more without the need for specialized high-pressure equipment, thereby de-risking the scale-up phase for new product launches.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrilase technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the mutants in an industrial setting. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing infrastructure.
Q: How does the BaNITmut/V82L/C237S double mutant improve upon previous nitrilase variants?
A: The double mutant exhibits a 2.72-fold increase in catalytic activity compared to the parent strain and significantly reduces the formation of the amide by-product from 6.2% down to 2.28%, ensuring higher optical purity and simplified downstream processing.
Q: What substrate concentrations can this enzymatic process tolerate?
A: The engineered nitrilase mutants demonstrate robust performance at high substrate loadings, effectively catalyzing the hydrolysis of racemic isobutyl succinonitrile (IBSN) at concentrations up to 175 g/L while maintaining conversion rates above 45%.
Q: Is the enantiomeric excess (ee) sufficient for direct pharmaceutical use?
A: Yes, the process yields the chiral intermediate (S)-3-cyano-5-methylhexanoic acid with an enantiomeric excess (ee) greater than 99% and an E value exceeding 500, meeting stringent requirements for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-cyano-5-methylhexanoic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the BaNITmut/V82L/C237S system can be seamlessly translated into robust manufacturing processes. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (S)-3-cyano-5-methylhexanoic acid meets the exacting standards required for global API registration. We are committed to delivering not just a product, but a comprehensive solution that optimizes your supply chain and reduces your time to market.
We invite you to collaborate with our technical procurement team to explore how this technology can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our proven track record in delivering high-purity pharmaceutical intermediates.
