Advanced Raney Nickel Catalytic Hydrogenation for High-Purity 3-Amino-4-Methoxyacetanilide Production
The global demand for high-performance disperse dyes, specifically C.I. Disperse Blue 79 and Disperse Violet 58, has necessitated a rigorous re-evaluation of the synthetic routes for their key precursors. Patent CN101880242B introduces a transformative methodology for the preparation of 3-amino-4-methoxyacetanilide, a critical coupling component in the textile chemistry sector. This intellectual property delineates a sophisticated catalytic hydrogenation protocol utilizing Raney Nickel, marking a significant departure from archaic reduction techniques that have long plagued the fine chemical industry with environmental inefficiencies. By leveraging the unique surface properties of Raney Nickel, this process achieves a remarkable balance between kinetic efficiency and thermodynamic selectivity, ensuring that the nitro group is reduced to the amine functionality without compromising the integrity of the adjacent methoxy or acetamido groups. For R&D directors and process engineers, this patent represents a blueprint for modernizing production lines to meet increasingly stringent global environmental standards while maintaining economic viability through superior yield profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-amino-4-methoxyacetanilide has relied heavily on stoichiometric reduction methods, most notably the iron filings reduction process and the alkali sulfide reduction method. While these traditional pathways are chemically straightforward, they suffer from profound logistical and ecological drawbacks that render them unsustainable for modern large-scale manufacturing. The iron filings method, in particular, generates voluminous quantities of iron oxide sludge, creating a severe waste disposal burden that escalates operational costs and complicates regulatory compliance. Furthermore, the post-reaction workup in these conventional processes is notoriously labor-intensive, requiring extensive filtration and washing steps to remove inorganic residues, which invariably leads to product loss and reduced overall throughput. The use of sulfide reagents introduces additional hazards, including the potential release of toxic hydrogen sulfide gas and the formation of sulfur-containing impurities that are difficult to separate from the final product, thereby compromising the purity required for high-end dye applications.
The Novel Approach
In stark contrast, the novel approach detailed in the patent employs a catalytic hydrogenation strategy that fundamentally alters the reaction landscape by replacing stoichiometric reductants with molecular hydrogen. This shift eliminates the generation of inorganic solid waste, effectively converting the reduction byproduct into water, which aligns perfectly with the principles of green chemistry. The utilization of Raney Nickel as the heterogeneous catalyst provides a highly active surface for hydrogen adsorption and activation, facilitating the reduction under relatively mild conditions compared to other catalytic systems. This method not only streamlines the downstream processing by removing the need for sludge filtration but also enhances the safety profile of the operation by avoiding the handling of pyrophoric iron powders or toxic sulfides. The result is a cleaner, more atom-economical process that delivers a product with exceptional purity, directly addressing the quality constraints faced by procurement managers in the dye synthesis supply chain.
Mechanistic Insights into Raney Nickel-Catalyzed Nitro Reduction
The efficacy of this synthesis hinges on the specific interaction between the nitro substrate and the activated nickel surface. Raney Nickel, prepared by the leaching of aluminum from a Ni-Al alloy using sodium hydroxide, possesses a highly porous structure with a vast surface area, providing abundant active sites for the dissociative adsorption of hydrogen molecules. During the reaction, hydrogen atoms spill over onto the nickel surface and subsequently transfer to the nitrogen-oxygen bonds of the 3-nitro-4-methoxyacetanilide molecule. The mechanism proceeds through a series of sequential hydrogenation steps, typically passing through nitroso and hydroxylamine intermediates before reaching the final amine state. The high selectivity observed, ranging from 99.0% to 99.6%, suggests that the catalyst surface geometry favors the reduction of the nitro group while sterically or electronically inhibiting the hydrogenolysis of the methoxy ether bond or the deacetylation of the amide group, which are common side reactions in less selective catalytic systems.
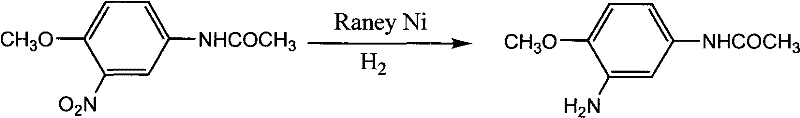
Furthermore, the control of impurities is intrinsically linked to the precise modulation of reaction parameters such as temperature and pressure. The patent data indicates that operating within the 80-110°C window prevents the thermal degradation of the sensitive acetamido moiety, which could otherwise lead to the formation of deacetylated byproducts that are difficult to remove. Similarly, maintaining hydrogen pressure between 0.8-2 MPa ensures a sufficient concentration of dissolved hydrogen to drive the reaction to completion without promoting over-reduction or ring saturation. This mechanistic precision allows for a robust impurity profile, where the primary organic impurity is typically the unreacted starting material, which can be easily managed through recrystallization, rather than complex structural analogs that require chromatographic separation. For quality assurance teams, this predictability in the impurity spectrum simplifies the validation of analytical methods and ensures consistent batch-to-batch reproducibility.
How to Synthesize 3-Amino-4-Methoxyacetanilide Efficiently
The practical implementation of this technology requires strict adherence to the optimized parameters defined in the patent to maximize both conversion and selectivity. The process begins with the in-situ or ex-situ preparation of the Raney Nickel catalyst, followed by the charging of the autoclave with the nitro precursor and a protic solvent such as methanol or ethanol. The reaction is conducted in a liquid-phase batch mode, which is highly compatible with existing infrastructure in most fine chemical plants. Critical to the success of the operation is the maintenance of vigorous stirring to ensure effective mass transfer of hydrogen from the gas phase to the catalyst surface, preventing diffusion limitations that could stall the reaction kinetics.
- Prepare the Raney Nickel catalyst by reacting Ni-Al alloy with 20wt% NaOH solution at 50-80°C, followed by washing to neutral pH.
- Load the autoclave with 3-nitro-4-methoxyacetanilide, solvent (methanol or ethanol), and the prepared catalyst at a mass ratio of 1: 10 to 3:10.
- Conduct hydrogenation at 80-110°C and 0.8-2 MPa pressure for 2-6 hours, then cool and analyze product purity via HPLC.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Raney Nickel catalytic process translates into tangible strategic advantages beyond mere technical superiority. The transition from a waste-generating stoichiometric process to a clean catalytic one fundamentally reshapes the cost structure of manufacturing this key dye intermediate. By eliminating the need for expensive waste treatment protocols associated with iron sludge or sulfide residues, manufacturers can realize substantial cost savings in environmental compliance and disposal fees. Additionally, the high selectivity of the reaction minimizes the loss of valuable raw materials to side products, effectively increasing the yield per ton of input and optimizing the utilization of capital-intensive feedstock. This efficiency gain is crucial in a volatile market where raw material price fluctuations can significantly impact margins.
- Cost Reduction in Manufacturing: The elimination of heavy metal waste and the simplification of the workup procedure drastically reduce the operational expenditure associated with downstream processing. Without the need for extensive filtration of iron mud or neutralization of sulfide waste, the labor and utility costs per kilogram of product are significantly lowered. Moreover, the ability to recover and potentially recycle the solvent further contributes to a leaner manufacturing cost base, allowing suppliers to offer more competitive pricing structures to their downstream dye manufacturing clients without sacrificing profitability.
- Enhanced Supply Chain Reliability: The robustness of the catalytic hydrogenation process ensures a more stable and predictable production schedule. Unlike traditional methods that might be susceptible to variations in raw material quality leading to inconsistent reaction rates, the catalytic system described demonstrates high tolerance and consistent performance across multiple batches. This reliability reduces the risk of production delays and ensures a continuous flow of high-purity intermediates to the market. For supply chain planners, this means reduced safety stock requirements and a lower probability of stockouts, thereby strengthening the overall resilience of the dye production supply network.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in batch reactors that can be readily expanded to industrial volumes. The clean nature of the reaction, producing water as the primary byproduct, aligns with increasingly rigorous global environmental regulations, mitigating the risk of regulatory shutdowns or fines. This environmental compatibility not only future-proofs the manufacturing asset but also enhances the brand reputation of the supplier as a responsible partner in the sustainable chemistry value chain, a factor that is becoming increasingly important for multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Raney Nickel catalytic hydrogenation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the advantages of using Raney Nickel over iron filings for nitro reduction?
A: Raney Nickel catalytic hydrogenation offers significantly higher selectivity (99.0-99.6%) and conversion rates compared to traditional iron filings reduction, while eliminating the massive amounts of iron sludge waste associated with the older method.
Q: What are the optimal reaction conditions for this hydrogenation process?
A: The patent specifies a reaction temperature range of 80-110°C and a hydrogen pressure between 0.8-2 MPa. The reaction typically completes within 2-6 hours using methanol or ethanol as the solvent.
Q: Is this process suitable for large-scale industrial production of dye intermediates?
A: Yes, the process is designed for industrial scalability. It utilizes standard batch autoclave equipment and achieves raw material conversion rates up to 100%, making it highly efficient for commercial manufacturing of disperse dye intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-4-Methoxyacetanilide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes is key to maintaining competitiveness in the global dye intermediate market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the laboratory success of the Raney Nickel hydrogenation route is seamlessly translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation, guaranteeing that every batch of 3-amino-4-methoxyacetanilide meets the exacting standards required for high-performance disperse dye synthesis. Our commitment to quality assurance ensures that our clients receive a product with consistent physical and chemical properties, minimizing variability in their final dye formulations.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and quality requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener catalytic technology. We encourage you to request specific COA data and route feasibility assessments to verify how our optimized manufacturing capabilities can support your long-term supply chain goals and drive value for your organization.
