Advanced Raney Nickel Catalytic Hydrogenation for High-Purity 3-Amino-4-Methoxyacetanilide Production
Advanced Raney Nickel Catalytic Hydrogenation for High-Purity 3-Amino-4-Methoxyacetanilide Production
In the competitive landscape of fine chemical intermediates, particularly for the textile and dye sectors, the efficiency and environmental footprint of synthesis routes are paramount. Patent CN101880242A introduces a transformative methodology for the preparation of 3-amino-4-methoxyacetanilide, a critical building block for high-performance disperse dyes such as C.I. Disperse Blue 79 and Violet 63. This technology leverages Raney Nickel catalytic hydrogenation to achieve unprecedented selectivity and conversion rates, addressing the long-standing industry pain points associated with traditional reduction methods. By optimizing reaction parameters including temperature, pressure, and catalyst loading, this process delivers a product purity profile that meets the rigorous demands of modern pharmaceutical and agrochemical supply chains, positioning it as a cornerstone technology for reliable dye intermediate suppliers seeking to upgrade their manufacturing capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-amino-4-methoxyacetanilide has relied heavily on archaic reduction techniques that are increasingly untenable in a regulated global market. The traditional iron powder reduction method, while chemically straightforward, generates voluminous quantities of iron sludge and acidic wastewater, creating severe environmental liabilities and高昂 disposal costs that erode profit margins. Alternatively, sodium sulfide reduction poses significant safety hazards due to the generation of toxic hydrogen sulfide gas and sulfur-containing waste streams that are difficult to treat. Furthermore, hydrazine hydrate reduction, though cleaner in terms of solid waste, suffers from high raw material costs and potential safety risks associated with handling energetic materials. These conventional pathways often struggle to maintain consistent selectivity, leading to complex impurity profiles that require energy-intensive purification steps, ultimately compromising the economic viability and sustainability of the supply chain for high-purity dye intermediates.
The Novel Approach
The innovative approach detailed in the patent data utilizes Raney Nickel as a heterogeneous catalyst to facilitate the direct hydrogenation of 3-nitro-4-methoxyacetanilide. This method represents a paradigm shift towards green chemistry principles, replacing stoichiometric reductants with molecular hydrogen, the ultimate clean reducing agent. The process operates under moderate conditions, typically between 80-110°C and 0.8-2 MPa pressure, ensuring operational safety while maintaining high reaction kinetics. Crucially, this catalytic system demonstrates exceptional chemoselectivity, reducing the nitro group to an amine without affecting the acetamido or methoxy functionalities, thereby minimizing byproduct formation. The result is a streamlined workflow that not only enhances yield but also drastically simplifies post-reaction workup, as the catalyst can be filtered and the solvent recovered, aligning perfectly with the goals of cost reduction in dye intermediate manufacturing.
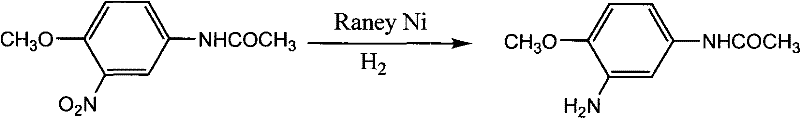
Mechanistic Insights into Raney Nickel Catalytic Hydrogenation
The efficacy of this synthesis lies in the unique surface properties of the Raney Nickel catalyst, which is prepared by leaching aluminum from a Ni-Al alloy using sodium hydroxide. This activation process creates a highly porous, sponge-like structure with an immense surface area, providing abundant active sites for hydrogen adsorption and activation. During the reaction, molecular hydrogen dissociates on the nickel surface into atomic hydrogen species, which are then transferred to the nitro group of the substrate adsorbed on the catalyst. The mechanism proceeds through a series of intermediate reduction steps, likely involving nitroso and hydroxylamine intermediates, before finally yielding the primary amine. The high dispersion of nickel ensures that the reaction rate is not limited by mass transfer, allowing for rapid conversion even at relatively low catalyst loadings, which is a critical factor for commercial scale-up of complex fine chemicals.
From an impurity control perspective, the specificity of the Raney Nickel surface is instrumental in achieving the reported selectivity of 99.0-99.6%. Unlike non-selective reducing agents that might attack the amide bond or demethylate the ether linkage under harsh conditions, the nickel-hydrogen system is tuned to target the nitro functionality exclusively. This selectivity is further enhanced by the choice of alcoholic solvents like methanol or ethanol, which stabilize the transition states and prevent side reactions. The patent data indicates that by carefully controlling the reaction time (2-6 hours) and avoiding excessive temperatures beyond 110°C, the formation of over-reduced products or condensation byproducts is effectively suppressed. This precise control over the reaction trajectory ensures that the final crude product requires minimal purification, directly translating to higher overall process efficiency and reduced solvent consumption.
How to Synthesize 3-Amino-4-Methoxyacetanilide Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing the importance of catalyst preparation and reaction parameter optimization. To achieve the highest yields, operators must first ensure the Raney Nickel is freshly activated or properly stored in ethanol to prevent oxidation and loss of activity. The reaction is typically conducted in a stirred autoclave, where the substrate, solvent, and catalyst are charged under an inert atmosphere before introducing hydrogen.
- Preparation of Raney Nickel Catalyst: Treat Ni-Al alloy with sodium hydroxide solution at 50-80°C to leach aluminum, then wash to neutral pH and store in ethanol.
- Hydrogenation Reaction: Load 3-nitro-4-methoxyacetanilide, solvent (methanol/ethanol), and catalyst into an autoclave. Replace air with nitrogen, then pressurize with hydrogen to 0.8-2 MPa.
- Reaction Control & Isolation: Heat to 80-110°C for 2-6 hours with stirring. Upon completion, cool the mixture, filter off the catalyst, and purify the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Raney Nickel hydrogenation technology offers compelling strategic advantages that extend beyond simple yield improvements. The transition from iron powder or sulfide reduction to catalytic hydrogenation fundamentally alters the cost structure of production by eliminating the massive waste disposal fees associated with heavy metal sludge. This shift not only reduces the direct operational expenditure but also mitigates regulatory risks, ensuring uninterrupted production schedules in regions with stringent environmental enforcement. Furthermore, the use of commodity chemicals like hydrogen and ethanol as reagents enhances supply chain resilience, as these materials are globally available and less prone to the price volatility seen with specialized reducing agents like hydrazine.
- Cost Reduction in Manufacturing: The implementation of this catalytic route drives significant cost savings primarily through the elimination of expensive waste treatment processes and the reduction of raw material consumption. By achieving near-quantitative conversion and high selectivity, the process minimizes the loss of valuable starting materials and reduces the volume of solvents required for purification. The ability to recover and reuse the Raney Nickel catalyst, or at least minimize its consumption due to high activity, further contributes to a lower cost of goods sold, making the final intermediate more competitive in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: Adopting this cleaner technology diversifies the supply base by enabling production in facilities that may have previously been restricted due to environmental capacity limits. The simplified process flow, characterized by fewer unit operations and shorter cycle times compared to iron reduction, allows for faster turnaround and increased throughput. This agility is crucial for meeting the just-in-time delivery requirements of downstream dye manufacturers, ensuring that lead times for high-purity dye intermediates are consistently met even during periods of high market demand.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard high-pressure reactor equipment found in most fine chemical plants, which facilitates easy technology transfer from pilot to commercial scale. From an environmental standpoint, the absence of heavy metal waste and toxic gas emissions aligns with global sustainability goals, such as Responsible Care® initiatives. This compliance not only protects the corporate reputation but also future-proofs the manufacturing asset against tightening environmental regulations, ensuring long-term operational continuity and social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Raney Nickel catalytic hydrogenation process. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making.
Q: What are the primary advantages of using Raney Nickel over iron powder reduction for this intermediate?
A: Raney Nickel catalytic hydrogenation offers significantly higher selectivity (99.0-99.6%) compared to traditional iron reduction, which generates massive amounts of iron sludge waste. The Raney Ni process is cleaner, eliminates heavy metal contamination risks, and simplifies downstream purification, leading to superior product quality for sensitive dye applications.
Q: What are the optimal reaction conditions for maximizing conversion rates?
A: According to patent data, optimal conversion (up to 100%) is achieved at temperatures between 80-110°C and hydrogen pressures ranging from 0.8 to 2.0 MPa. Using ethanol as a solvent with a catalyst-to-substrate ratio of roughly 3:10 often yields the highest efficiency, completing the reaction within 2 to 6 hours.
Q: Is this process scalable for industrial production of disperse dye intermediates?
A: Yes, the process is designed for industrial scalability. It utilizes standard batch autoclave equipment common in fine chemical plants. The use of recoverable solvents like methanol or ethanol and the robust nature of the Raney Nickel catalyst make it highly suitable for large-scale manufacturing of intermediates like C.I. Disperse Blue precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-4-Methoxyacetanilide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the value chain of the global dye and specialty chemical industries. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. Our facility is equipped to handle the specific safety and environmental requirements of catalytic hydrogenation, guaranteeing a secure and sustainable supply source for your critical raw materials.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced Raney Nickel technology can enhance your product quality and operational efficiency. Let us be your partner in driving innovation and reliability in the fine chemical sector.
