Scaling High-Concentration Biocatalytic Synthesis of Chiral Pharma Intermediates
Scaling High-Concentration Biocatalytic Synthesis of Chiral Pharma Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable routes for the production of high-value chiral intermediates. A pivotal advancement in this domain is documented in Chinese Patent CN111172124B, which discloses a novel carbonyl reductase mutant and its application in the preparation of (R)-4-chloro-3-hydroxy-butyrate. This specific chiral building block is a critical precursor for the synthesis of L-carnitine and other bioactive molecules, making its efficient production a strategic priority for supply chain stability. The patent introduces a genetically engineered solution that overcomes the historical limitations of low substrate tolerance in enzymatic reductions, thereby unlocking new potential for cost reduction in pharmaceutical intermediate manufacturing. By leveraging site-directed mutagenesis on the YOL151W enzyme derived from Saccharomyces cerevisiae, the inventors have created a biocatalyst capable of operating at industrially relevant concentrations.
This technological breakthrough addresses the fundamental bottleneck of biocatalysis: the trade-off between selectivity and productivity. Traditional wild-type enzymes often exhibit exquisite selectivity but fail under the high substrate loads required for commercial viability. The innovation described in CN111172124B shifts this paradigm by enhancing the catalytic efficiency without compromising stereocontrol. For R&D directors and process chemists, this represents a significant opportunity to transition from stoichiometric chemical reductions, which often require harsh conditions and generate heavy metal waste, to a greener enzymatic alternative. The ability to synthesize high-purity pharmaceutical intermediates using a renewable biological system aligns perfectly with modern green chemistry principles and regulatory pressures to reduce environmental footprints in API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the biocatalytic reduction of chloroacetoacetate was severely constrained by the low substrate tolerance of available enzymes. Literature reports, such as those by Kim and Sakai, utilized the wild-type carbonyl reductase YOL151W, which demonstrated excellent enantioselectivity but could only function effectively at substrate concentrations of ≤10 g/L, corresponding to a mass percentage of ≤1%. From a process engineering perspective, this limitation is catastrophic for commercial scale-up. Operating at such dilute concentrations implies that vast volumes of water must be processed to produce a relatively small amount of product, leading to enormous energy costs for heating, cooling, and downstream separation. Furthermore, the low space-time yield means that reactor volumes must be impractically large to meet market demand, rendering the process economically unfeasible for bulk chemical production. This inefficiency has historically forced manufacturers to rely on traditional chemical synthesis routes involving borohydrides or other reducing agents, which introduce issues of waste disposal and safety hazards.
The Novel Approach
The novel approach presented in the patent data fundamentally alters the economic equation by enabling high-concentration biocatalysis. Through rational protein engineering, specifically the substitution of phenylalanine with methionine at position 85 (F85M), the modified enzyme exhibits a dramatically improved reducing ability towards chloroacetoacetate. This mutation allows the reaction to proceed efficiently at substrate mass percentages ranging from 10% to 30% (w/v), representing a tenfold to thirtyfold increase over previous methods. This leap in performance transforms the process from a laboratory curiosity into a viable industrial operation. The new method employs a whole-cell biocatalyst system where the engineered E. coli strain co-expresses the mutant carbonyl reductase and a glucose dehydrogenase (GDH). This dual-enzyme system facilitates in situ cofactor regeneration, eliminating the need for expensive external addition of NADPH. The result is a streamlined, cost-effective process that maintains the high stereoselectivity of the wild type while achieving the throughput necessary for commercial success.
Mechanistic Insights into YOL151W-Catalyzed Asymmetric Reduction
The core of this technology lies in the precise structural modification of the YOL151W enzyme. The wild-type enzyme, while selective, possesses an active site architecture that likely restricts the binding or turnover of the substrate at higher concentrations, possibly due to steric hindrance or suboptimal hydrophobic interactions. The F85M mutation replaces a bulky aromatic phenylalanine residue with a sulfur-containing methionine residue. This subtle change is hypothesized to enlarge the substrate binding pocket or alter the local electronic environment, thereby facilitating better accommodation of the chloroacetoacetate molecule. This enhanced fit allows the enzyme to maintain high catalytic turnover rates even when the substrate is present in significant excess. The mechanism involves the hydride transfer from the reduced nicotinamide adenine dinucleotide phosphate (NADPH) to the ketone group of the substrate, generating the chiral alcohol and oxidized NADP+.
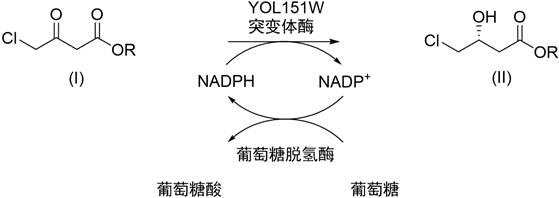
To sustain this reaction without prohibitive costs, the system integrates a cofactor regeneration cycle driven by glucose dehydrogenase. As the carbonyl reductase consumes NADPH to reduce the ketone, the GDH simultaneously oxidizes glucose to gluconolactone, regenerating NADPH from NADP+. This closed-loop system ensures that only a catalytic amount of the expensive cofactor is needed, while the inexpensive glucose serves as the terminal reductant. The reaction is typically conducted in a biphasic system involving an aqueous buffer and a water-immiscible organic solvent like toluene. The organic phase serves to solubilize the hydrophobic substrate and product, reducing substrate inhibition and facilitating product extraction. This mechanistic design not only ensures high conversion rates but also simplifies downstream processing, as the product can be easily separated from the aqueous enzymatic broth. The consistency of this mechanism across different ester variants (ethyl, methyl, tert-butyl) demonstrates the robustness of the mutant enzyme.
Impurity control is another critical aspect of this mechanistic framework. In chemical reductions, over-reduction or dehalogenation can occur, leading to difficult-to-remove impurities. The enzymatic route, governed by the specific geometry of the mutant active site, exhibits high chemoselectivity. The enzyme specifically targets the pro-chiral ketone without affecting the chloro-group or the ester functionality. This intrinsic selectivity minimizes the formation of side products such as dechlorinated alcohols or diols, which are common impurities in metal-catalyzed hydrogenations. Consequently, the crude product obtained from the biocatalytic reaction possesses a much cleaner impurity profile. This reduces the burden on purification steps such as distillation or crystallization, directly contributing to higher overall yields and lower production costs. For quality control teams, this means a more predictable and stable impurity spectrum, which is essential for regulatory filings and consistent batch-to-batch quality.
How to Synthesize (R)-4-Chloro-3-Hydroxy-Butyrate Efficiently
The implementation of this biocatalytic route requires a structured approach to strain construction and process optimization. The patent outlines a clear pathway starting from the genetic modification of the enzyme to the final isolation of the product. The process begins with the construction of the recombinant plasmid carrying the F85M mutation, followed by its transformation into a suitable host organism alongside the cofactor regeneration machinery. Once the whole-cell biocatalyst is prepared, the reaction conditions must be carefully controlled to maximize efficiency. Key parameters include maintaining the pH within a narrow range (6.5-7.5) using a base like potassium carbonate, controlling the temperature between 25-37°C, and ensuring adequate mixing in the biphasic system. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Construct the recombinant expression vector pET28b-YOL151W F85M via site-directed mutagenesis replacing phenylalanine with methionine at position 85.
- Co-transform the mutant vector and a glucose dehydrogenase vector (pACYC-GDH) into E. coli BL21(DE3) to create the whole-cell biocatalyst.
- Perform the asymmetric reduction in a biphasic system containing substrate, glucose, phosphate buffer, and toluene at 30°C with pH control.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this high-concentration biocatalytic process offers substantial strategic benefits beyond mere technical novelty. The primary driver is the drastic improvement in process efficiency, which translates directly into cost competitiveness. By increasing the substrate loading from 1% to over 20%, the volume of water that needs to be heated, cooled, and treated is significantly reduced. This reduction in utility consumption lowers the variable cost per kilogram of the product. Additionally, the elimination of stoichiometric chemical reducing agents removes the volatility associated with the pricing of inorganic reagents and the costs of disposing of their corresponding waste streams. The reliance on glucose, a commodity chemical with a stable global supply, further insulates the production cost from market fluctuations typical of specialized synthetic reagents.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the intensification of the reaction process. Operating at high substrate concentrations means that the same amount of product can be manufactured in a much smaller reactor volume, or conversely, existing infrastructure can produce significantly more output. This capital efficiency reduces the depreciation cost allocated to each unit of product. Furthermore, the use of a whole-cell biocatalyst avoids the expensive and complex purification steps required for isolated enzymes. The cells can be used directly after cultivation, streamlining the upstream process. The biphasic system also aids in product recovery; since the product partitions into the organic phase, simple phase separation can replace energy-intensive extraction or distillation steps initially. These factors combine to create a leaner, more cost-effective manufacturing model that enhances margin potential.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes and the availability of key reagents. This biocatalytic route simplifies the supply chain by relying on fermentation-derived catalysts and bulk sugars. The engineered E. coli strain is robust and can be stored and propagated reliably, ensuring a consistent source of catalytic activity. Unlike precious metal catalysts, which are subject to geopolitical supply risks and mining constraints, the biological catalyst is renewable and can be produced on-demand. The high yield and selectivity reported in the patent (up to 91% yield and 97% ee) mean that less raw material is wasted, improving the overall material balance. This efficiency reduces the pressure on raw material sourcing and ensures that production targets can be met consistently without unexpected shortages or yield failures.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is often challenging due to oxygen transfer or mixing issues, but the biphasic nature of this specific reaction mitigates many of these risks. The use of toluene as an extractive solvent helps manage substrate inhibition and toxicity, making the scale-up from gram to ton scale more predictable. From an environmental perspective, this process is inherently greener. It operates at mild temperatures and neutral pH, avoiding the extreme conditions that require specialized, corrosion-resistant equipment. The waste stream is primarily biological biomass and aqueous salts, which are easier to treat than the heavy metal residues from chemical reductions. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking to reduce their carbon footprint and comply with stringent environmental regulations in major markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of adopting this route for your specific supply chain needs. The high enantioselectivity and yield data confirm the maturity of the technology for industrial application.
Q: What is the primary advantage of the YOL151W F85M mutant over the wild-type enzyme?
A: The YOL151W F85M mutant exhibits significantly improved catalytic activity, allowing for substrate concentrations of 10%-30% (w/v), whereas the wild-type enzyme is limited to ≤1% (10 g/L), making the mutant far more viable for industrial scale-up.
Q: What level of enantioselectivity can be achieved with this biocatalytic process?
A: The process maintains excellent stereoselectivity, consistently achieving enantiomeric excess (ee) values of 97% to 98% across various ester substrates including ethyl, methyl, and tert-butyl esters.
Q: Is this biocatalytic route suitable for large-scale commercial production?
A: Yes, the use of a biphasic system with toluene and high substrate loading significantly enhances space-time yield, while the cofactor regeneration system using glucose ensures economic feasibility for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-4-Chloro-3-Hydroxy-Butyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic routes described in CN111172124B for the production of high-value chiral intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this laboratory innovation to full industrial reality. Our facilities are equipped with state-of-the-art fermentation and biocatalysis suites capable of handling the specific requirements of whole-cell catalysis, including precise pH control and biphasic reaction management. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of (R)-4-chloro-3-hydroxy-butyrate meets the highest standards of enantiomeric excess and chemical purity demanded by the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical team can provide a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this enzymatic route compared to your current chemical synthesis. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project timelines. By leveraging our expertise in enzyme engineering and process development, we can help you secure a reliable, cost-effective, and sustainable supply of this essential building block, ensuring your downstream drug development programs proceed without interruption.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
