Advanced Carbonylation Strategy for Commercial Production of 2-Nitro-4-Methylsulfonylbenzoic Acid
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical intermediates such as 2-nitro-4-methylsulfonylbenzoic acid (NMSBA), a key precursor for the widely used triketone herbicide Mesotrione. Recent intellectual property developments, specifically patent CN113801045A published in late 2021, have introduced a transformative approach to manufacturing this compound. Unlike legacy processes that rely on aggressive oxidation, this new methodology employs a sophisticated palladium-catalyzed carbonylation strategy. This shift represents a significant technological leap for the agrochemical industry, addressing long-standing challenges related to environmental compliance, operational safety, and product purity. For R&D directors and procurement specialists alike, understanding the nuances of this carbonylation pathway is essential for securing a robust and sustainable supply chain for next-generation crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-nitro-4-methylsulfonylbenzoic acid has been fraught with significant chemical and environmental hurdles. The traditional synthetic route typically involves the direct oxidation of 2-nitro-4-methylsulfonyltoluene (NMST). As illustrated in the reaction scheme below, this transformation is chemically demanding because both the nitro group and the methylsulfonyl group attached to the benzene ring act as strong electron-withdrawing substituents. These groups deactivate the aromatic ring and the methyl side chain, making the oxidation of the methyl group to a carboxylic acid exceptionally difficult. Consequently, conventional processes often necessitate the use of harsh oxidizing agents, such as potassium permanganate or sodium dichromate, or corrosive mixed acids comprising nitric and sulfuric acid.
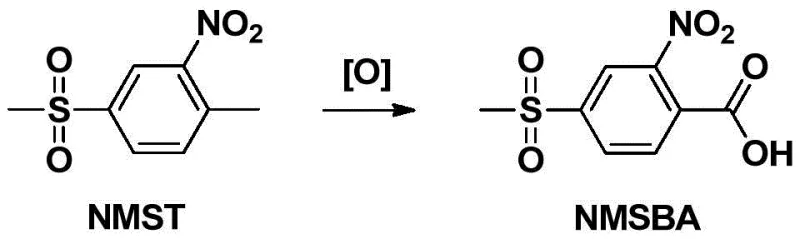
The reliance on these aggressive reagents creates severe downstream consequences for manufacturing facilities. The use of heavy metal oxidants generates substantial quantities of toxic wastewater containing chromium or manganese residues, imposing a heavy burden on waste treatment infrastructure and increasing the risk of environmental contamination. Furthermore, the harsh reaction conditions required to overcome the electronic deactivation of the substrate can lead to safety hazards, including thermal runaways and equipment corrosion. From a supply chain perspective, the variability in yield and the complexity of purifying the product from heavy metal impurities often result in inconsistent quality, which is unacceptable for the stringent specifications required in modern agrochemical formulations.
The Novel Approach
In stark contrast to the oxidative degradation of toluene derivatives, the method disclosed in patent CN113801045A utilizes a constructive carbonylation strategy. This innovative route begins with 2-nitro-4-methylsulfonyl chlorobenzene, a readily available starting material, and subjects it to a coupling reaction with carbon monoxide and a lower alcohol (such as methanol or ethanol). This transformation is catalyzed by a palladium complex in the presence of a specific phosphine ligand and an organic base. The reaction proceeds under controlled pressure and temperature to form an ester intermediate, which is subsequently hydrolyzed to yield the target carboxylic acid. This approach fundamentally bypasses the difficulties associated with oxidizing deactivated methyl groups.
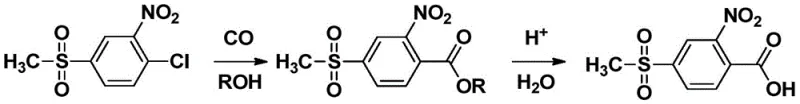
The advantages of this novel approach are manifold and address the core pain points of the legacy technology. By avoiding heavy metal oxidants and mixed acids, the process significantly reduces the generation of hazardous waste, aligning with increasingly strict global environmental regulations. The reaction conditions, while requiring pressure equipment for carbon monoxide, are generally milder and more controllable than high-temperature oxidations. Moreover, the selectivity of the palladium-catalyzed carbonylation ensures that the sensitive nitro and sulfonyl functionalities remain intact, leading to a cleaner reaction profile. This results in a final product with exceptional purity, often exceeding 98.5% without the need for extensive and costly purification steps, thereby streamlining the entire manufacturing workflow.
Mechanistic Insights into Palladium-Catalyzed Alkoxycarbonylation
The core of this technological advancement lies in the precise orchestration of the palladium catalytic cycle. The reaction initiates with the oxidative addition of the aryl chloride bond of 2-nitro-4-methylsulfonyl chlorobenzene to the active palladium(0) species generated in situ from precursors like palladium acetate or palladium chloride. The choice of ligand is critical here; the patent highlights bis-diphenylphosphinopropane (DPPP) as a superior ligand compared to triphenylphosphine or bidentate ligands like DPPE. DPPP likely provides an optimal bite angle and electronic environment that stabilizes the palladium center against the strongly electron-withdrawing nitro and sulfonyl groups on the substrate, facilitating the oxidative addition step which is often the rate-determining step in such deactivated systems.
Following oxidative addition, carbon monoxide inserts into the palladium-carbon bond to form an acyl-palladium complex. This step is driven by the partial pressure of CO, typically maintained between 2 MPa and 3 MPa in the described examples. Subsequently, the alcohol nucleophile (methanol or ethanol), which also serves as the solvent, attacks the acyl complex, leading to the formation of the ester product and regenerating the palladium catalyst. The presence of an organic base, such as triethylamine or pyridine, is essential to neutralize the hydrochloric acid byproduct formed during the cycle, preventing catalyst deactivation and driving the equilibrium forward. This mechanistic pathway ensures high atom economy and minimizes side reactions, which is crucial for maintaining the integrity of the sensitive functional groups on the aromatic ring.
Impurity control in this process is inherently superior due to the specificity of the transition metal catalysis. Unlike free-radical oxidations which can attack various positions on the molecule or lead to over-oxidation, the carbonylation is highly regioselective for the carbon-chlorine bond. The subsequent hydrolysis step is performed under mild acidic conditions (pH 1-3) at moderate temperatures (40-80°C), which further prevents the decomposition of the nitro group or the sulfone moiety. This gentle work-up allows for the isolation of the product simply by filtration and washing, avoiding the complex extraction and chromatography often needed to remove heavy metal salts from oxidation reactions. The result is a process that delivers high-purity material suitable for direct use in the synthesis of Mesotrione.
How to Synthesize 2-Nitro-4-Methylsulfonylbenzoic Acid Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the catalyst system and the management of gas pressures. The patent outlines a robust protocol where the molar ratio of the substrate to the palladium catalyst is optimized between 1:0.01 and 1:0.2, with a preference for lower catalyst loadings to enhance economic viability. The ligand-to-metal ratio is equally important, with a range of 1:1 to 1:1.5 proving most effective for maximizing turnover numbers. The reaction is typically conducted in a stainless steel autoclave capable of withstanding the requisite CO pressure and temperature up to 180°C. Following the carbonylation, the solvent is removed, and the residue is subjected to aqueous acid hydrolysis to cleave the ester and precipitate the final acid product.
- Perform coupling reaction of 2-nitro-4-methylsulfonyl chlorobenzene with carbon monoxide and alcohol (e.g., methanol) using a palladium catalyst and phosphine ligand at 120-180°C under 2-3 MPa pressure.
- Remove solvent from the resulting ester intermediate and subject it to hydrolysis at pH 1-3 and 40-80°C to yield the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this carbonylation-based process offers tangible strategic benefits beyond mere technical elegance. The elimination of hazardous oxidants translates directly into reduced costs associated with waste disposal and environmental compliance. Facilities no longer need to invest in specialized treatment systems for chromium or manganese-laden effluents, nor do they face the regulatory risks associated with storing and handling large quantities of concentrated nitric acid. This simplification of the EHS (Environment, Health, and Safety) profile makes the manufacturing process more resilient to regulatory changes and community scrutiny, ensuring long-term operational continuity.
- Cost Reduction in Manufacturing: The economic implications of switching to this catalytic route are profound. By removing the need for expensive and stoichiometric heavy metal oxidants, the raw material costs are significantly lowered. Furthermore, the high selectivity of the reaction minimizes the formation of by-products, which reduces the loss of valuable starting materials and lowers the cost of goods sold (COGS). The ability to use the alcohol reactant as both a reagent and a solvent simplifies the material balance and reduces solvent recovery costs. Additionally, the simplified post-reaction work-up, which avoids complex purification steps to remove metal residues, leads to substantial savings in labor and utility consumption, effectively driving down the overall manufacturing cost per kilogram of the intermediate.
- Enhanced Supply Chain Reliability: Supply chain stability is often compromised by the availability of specialized reagents and the capacity of waste treatment facilities. This new method relies on commodity chemicals such as carbon monoxide, methanol, and standard palladium catalysts, which are widely available in the global chemical market. The robustness of the reaction conditions means that production is less susceptible to batch failures caused by sensitivity to moisture or temperature fluctuations, common issues in traditional oxidation processes. This reliability ensures a consistent flow of high-quality intermediate to downstream herbicide manufacturers, reducing the risk of production stoppages and enabling better inventory planning for finished agrochemical products.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is always a challenge, but this carbonylation route is inherently designed for industrial application. The use of continuous or semi-batch pressurized reactors is a standard unit operation in the fine chemical industry, facilitating straightforward scale-up from pilot plants to commercial manufacturing lines. The process generates minimal three wastes (waste water, waste gas, waste residue), particularly avoiding the heavy metal sludge that plagues oxidation routes. This green chemistry profile not only meets current environmental standards but also future-proofs the supply chain against tightening global regulations on industrial emissions, making it a sustainable choice for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical disclosures found in the patent literature, providing a clear picture of what potential partners and licensees can expect regarding performance and operational requirements.
Q: Why is the traditional oxidation method for NMSBA considered problematic?
A: Traditional methods involve oxidizing 2-nitro-4-methylsulfonyltoluene using heavy metal oxidants like dichromate or permanganate, which generate significant toxic waste and require harsh reaction conditions due to the electron-withdrawing nature of the nitro and sulfonyl groups.
Q: What are the key advantages of the carbonylation route described in CN113801045A?
A: The novel route utilizes carbon monoxide and alcohol in the presence of a palladium catalyst, eliminating the need for toxic heavy metal oxidants and mixed acids, resulting in a safer, more environmentally friendly process with high product purity (up to 98.5%).
Q: Which catalyst system yields the best results for this synthesis?
A: Experimental data indicates that using palladium acetate or palladium chloride combined with bis-diphenylphosphinopropane (DPPP) as the ligand provides superior yields compared to other phosphine ligands like triphenylphosphine or Xantphos.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-4-Methylsulfonylbenzoic Acid Supplier
As the agrochemical sector evolves towards more sustainable and efficient manufacturing practices, the ability to produce key intermediates like 2-nitro-4-methylsulfonylbenzoic acid via advanced catalytic methods becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging deep expertise in transition metal catalysis and process optimization. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of heavy metal contaminants and ensure that every batch conforms to the high standards required for herbicide synthesis.
We invite global agrochemical manufacturers and trading companies to collaborate with us to optimize their supply chains. By adopting this cleaner, more efficient synthesis route, we can jointly achieve significant operational improvements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your production goals for Mesotrione and related crop protection solutions.
